Advanced Manufacturing of 2-Fluoro-4-Hydroxybenzaldehyde for Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways for fluorinated building blocks, particularly those containing sensitive functional groups like aldehydes and phenols. A recent technological breakthrough detailed in patent CN115124410A presents a highly efficient preparation method for 2-fluoro-4-hydroxybenzaldehyde (CAS 348-27-6), a critical intermediate for physiologically active compounds including melanin concentrating hormone receptor antagonists. This novel synthesis strategy fundamentally shifts the paradigm from hazardous, cryogenic lithiation chemistry to a milder, cost-effective Grignard-based protocol utilizing isopropyl protection. By systematically screening phenolic hydroxyl protecting groups and bromination reagents, the inventors have identified a route that eliminates the need for ultra-low temperature equipment while delivering product purity exceeding 99.5 percent. For global procurement teams and R&D directors, this represents a significant opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering high-quality materials with reduced supply chain volatility.
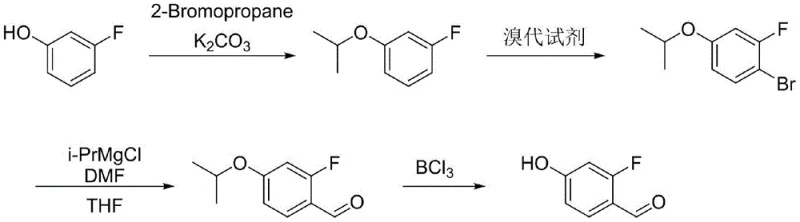
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-fluoro-4-hydroxybenzaldehyde has relied heavily on methodologies that are technically demanding and economically inefficient for large-scale manufacturing. Traditional literature routes, such as those described in the Journal of Fluorine Chemistry, typically employ tert-butyldimethylsilyl chloride for hydroxyl protection followed by metalation with sec-butyllithium. This approach necessitates reaction temperatures as low as minus 78 degrees Celsius to control the highly reactive organolithium species and prevent side reactions. Such cryogenic conditions impose severe constraints on industrial reactors, requiring specialized cooling infrastructure that drastically increases capital expenditure and operational energy costs. Furthermore, the use of sec-butyllithium introduces significant safety hazards due to its pyrophoric nature, complicating handling procedures and increasing insurance liabilities. The cumulative effect of these factors is a process with limited scalability, where the total yield often struggles to justify the high input costs of exotic reagents and extreme thermal management.
The Novel Approach
In stark contrast, the methodology disclosed in CN115124410A introduces a streamlined four-step sequence that prioritizes operational simplicity and economic viability without compromising chemical integrity. The core innovation lies in the strategic selection of an isopropyl protecting group, derived from the inexpensive and readily available 2-bromopropane, rather than bulky silyl ethers. This modification allows the subsequent bromination and Grignard exchange steps to proceed at much more moderate temperatures, specifically between minus 10 and 0 degrees Celsius. By replacing the dangerous lithiation step with a Grignard reagent exchange using isopropyl magnesium chloride, the process mitigates thermal risks and simplifies the engineering requirements for production facilities. This shift not only enhances the safety profile but also facilitates cost reduction in pharmaceutical intermediates manufacturing by utilizing commodity chemicals and standard reactor configurations, making it an ideal candidate for commercial scale-up of complex fluorinated aromatics.
Mechanistic Insights into Isopropyl Protection and Selective Bromination
The success of this synthetic route hinges on the precise control of regioselectivity during the electrophilic aromatic substitution phase, which is directly influenced by the steric and electronic properties of the protecting group. When 3-fluorophenol is protected with smaller groups like methyl or ethyl, the resulting anisole derivatives exhibit poor regiocontrol during bromination, leading to a complex mixture of isomers including unwanted dibrominated byproducts and ortho-brominated impurities. However, the introduction of the isopropyl group creates sufficient steric hindrance around the oxygen atom to effectively block substitution at the ortho-position relative to the ether linkage. This steric bulk forces the incoming bromine electrophile to attack the position ortho to the fluorine atom (para to the oxygen), which is the desired site for the subsequent formylation. Experimental data within the patent demonstrates that while tert-butyl groups offer even greater selectivity, their removal requires harsher conditions and the starting material is cost-prohibitive. The isopropyl group strikes the perfect balance, directing the reaction pathway efficiently while remaining labile enough for mild acidic cleavage later in the sequence.
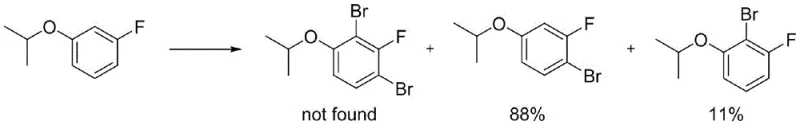
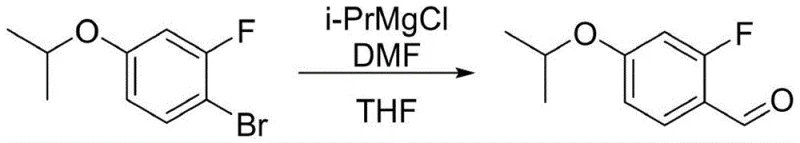
Furthermore, the choice of brominating agent plays a pivotal role in minimizing impurity formation. Traditional elemental bromine can be difficult to handle and often leads to over-bromination. The patent highlights the superiority of solid bromine sources such as tetrabutyl ammonium tribromide or pyridinium tribromide. These reagents release bromine in a controlled manner, maintaining a low concentration of the active electrophile in the solution, which kinetically favors mono-bromination over poly-bromination. Following the installation of the bromine atom, the transformation to the aldehyde is achieved through a halogen-metal exchange. Unlike the irreversible and violent nature of lithium-halogen exchange at cryogenic temperatures, the magnesium-halogen exchange using i-PrMgCl is controllable at minus 10 degrees Celsius. The resulting aryl magnesium species is then quenched with dimethylformamide (DMF) to install the formyl group, completing the construction of the carbon skeleton with high fidelity.
How to Synthesize 2-Fluoro-4-Hydroxybenzaldehyde Efficiently
The implementation of this synthesis requires careful attention to stoichiometry and temperature control to maximize yield and purity. The process begins with the alkylation of 3-fluorophenol using potassium carbonate and 2-bromopropane in a polar aprotic solvent like acetonitrile, heated to 80-82 degrees Celsius to ensure complete conversion. Following isolation, the protected intermediate undergoes bromination in dichloromethane at 0-10 degrees Celsius using tetrabutyl ammonium tribromide, yielding the mono-brominated species with minimal isomeric contamination. The critical formylation step involves dissolving the bromo-intermediate in tetrahydrofuran and treating it with isopropyl magnesium chloride at sub-zero temperatures before adding DMF. Finally, the isopropyl group is removed using boron trichloride in dichloromethane, followed by an aqueous workup and recrystallization from isopropyl ether to achieve the final high-purity specification.
- Protect 3-fluorophenol with 2-bromopropane and potassium carbonate to form 1-fluoro-3-isopropoxybenzene.
- Perform selective bromination using tetrabutyl ammonium tribromide to obtain 1-bromo-2-fluoro-4-isopropoxybenzene.
- Execute Grignard exchange with i-PrMgCl followed by DMF quenching to introduce the aldehyde group.
- Deprotect the isopropyl group using boron trichloride and purify via recrystallization to yield >99.5% pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented methodology offers tangible benefits that extend beyond simple yield improvements. The transition away from cryogenic lithiation chemistry fundamentally alters the cost structure of production by eliminating the need for specialized low-temperature reactors and the associated high energy consumption for cooling. This reduction in utility demand translates directly into lower manufacturing overheads, allowing for more competitive pricing structures in long-term supply agreements. Additionally, the reliance on commodity reagents such as 2-bromopropane and potassium carbonate, rather than expensive silyl chlorides or pyrophoric organolithiums, insulates the supply chain from the volatility often seen in the market for specialty fine chemicals. This stability ensures consistent availability of raw materials, reducing the risk of production stoppages due to supplier shortages.
- Cost Reduction in Manufacturing: The elimination of ultra-low temperature requirements and expensive silyl protecting groups significantly lowers the operational expenditure per kilogram of product. By utilizing standard cooling media instead of cryogenic fluids and replacing high-cost reagents with bulk commodities, the overall production cost is drastically simplified and optimized. This efficiency allows manufacturers to absorb fluctuations in raw material pricing while maintaining healthy margins, ultimately providing customers with a more stable and cost-effective sourcing option for this critical intermediate.
- Enhanced Supply Chain Reliability: The use of robust, non-pyrophoric reagents like Grignard solutions and solid tribromide salts simplifies logistics and storage requirements. Unlike sec-butyllithium, which requires stringent safety protocols and specialized transport, the reagents in this process are widely available from multiple global suppliers. This diversification of the supply base reduces dependency on single-source vendors and minimizes lead times for high-purity pharmaceutical intermediates, ensuring that downstream drug development timelines are not compromised by upstream material delays.
- Scalability and Environmental Compliance: The mild reaction conditions and high selectivity of this process result in a cleaner crude product profile, which simplifies downstream purification and reduces the volume of solvent and waste generated per unit of product. The ability to achieve high purity through simple recrystallization rather than complex chromatographic separations makes the process inherently scalable from pilot plant to multi-ton commercial production. Furthermore, the avoidance of heavy metal catalysts and the use of recyclable solvents align with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance in strict jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-fluoro-4-hydroxybenzaldehyde using this advanced methodology. These insights are derived directly from the experimental data and process optimizations detailed in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their own manufacturing workflows.
Q: Why is the isopropyl protecting group preferred over tert-butyl or methyl groups?
A: The isopropyl group offers an optimal balance of steric hindrance and cost. Unlike methyl or ethyl groups which produce excessive bromination isomers, and tert-butyl groups which are expensive and difficult to remove, the isopropyl group directs bromination selectively to the desired position while remaining easy to cleave under mild conditions.
Q: How does this method improve safety compared to traditional lithiation routes?
A: Traditional methods often require sec-butyllithium and ultra-low temperatures (-78°C), posing significant thermal runaway risks. This patented process utilizes Grignard reagents at moderate temperatures (-10°C to 0°C), drastically reducing energy consumption and operational hazards associated with cryogenic cooling.
Q: What is the achievable purity of the final 2-fluoro-4-hydroxybenzaldehyde?
A: Through optimized recrystallization using isopropyl ether after the deprotection step, the process consistently achieves a chemical purity exceeding 99.5%, meeting the stringent specifications required for advanced pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-4-Hydroxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of new therapeutic agents depends on the consistent supply of high-quality building blocks. Our technical team has extensively analyzed the pathway described in CN115124410A and possesses the expertise to implement this superior synthesis route at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive material that meets stringent purity specifications regardless of order volume. Our rigorous QC labs are equipped to verify the absence of critical isomers and residual solvents, guaranteeing that every batch of 2-fluoro-4-hydroxybenzaldehyde performs reliably in your downstream coupling reactions.
We invite you to collaborate with us to leverage these process efficiencies for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your annual volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your R&D timeline and your bottom line.
