Advanced Chiral Synthesis of Tetrahydrobenzyl Isoquinoline Compounds for Scalable Pharmaceutical Manufacturing
Advanced Chiral Synthesis of Tetrahydrobenzyl Isoquinoline Compounds for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes for neuromuscular blocking agents, where stereochemical integrity is paramount for patient safety. Patent CN114716375A introduces a groundbreaking preparation method for tetrahydrobenzyl isoquinoline compounds, specifically targeting the key intermediates for Mivacurium Chloride and Cisatracurium Besylate. This technology addresses the critical challenge of controlling chiral centers during the synthesis of short-acting non-depolarizing muscle relaxants. The structural complexity of these molecules, particularly the four chiral centers found in Mivacurium Chloride, demands absolute precision to avoid cardiovascular side effects associated with the S-configuration isomers.
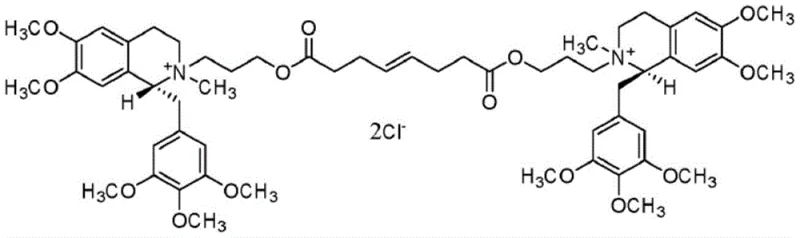
By integrating a specific chiral organic acid resolution step immediately following the hydrogenation reaction, this novel process ensures that the resulting intermediate possesses both high chemical purity and exceptional optical purity. This advancement represents a significant leap forward for manufacturers aiming to produce reliable pharmaceutical intermediate supplier grades that meet the stringent regulatory requirements of global health authorities. The ability to strictly control the R-configuration at the parent chiral carbon atoms is not merely a technical achievement but a fundamental requirement for the clinical efficacy and safety profile of the final drug product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key intermediates like 6,7-dimethoxy-1-(R)-3,4,5-trimethoxybenzyl-1,2,3,4-tetrahydroisoquinoline has been plagued by significant purification challenges. Prior art methods, such as those reported in literature like Tetrahedron: Asymmetry (2013), often rely on standard hydrogenation followed by tedious workup procedures that fail to adequately separate stereoisomers. These conventional routes typically yield viscous target substances with suboptimal optical purity, often capping at around 95% ee, which implies a dangerous 2.5% contamination of the unwanted S-configuration compound. Furthermore, the chemical purity in these older processes is frequently poor, hovering around 70%, necessitating extensive and costly downstream purification steps that erode overall process efficiency.
The Novel Approach
The methodology disclosed in CN114716375A fundamentally restructures the purification workflow by introducing a chiral resolution strategy directly into the synthesis sequence. Instead of attempting to purify the free base through difficult distillation or chromatography, the process converts the crude reaction mixture into a diastereomeric salt using specific chiral organic acids such as D-tartaric acid or D-malic acid. This salt formation step acts as a powerful filter, selectively crystallizing the desired R-enantiomer while leaving impurities and the S-enantiomer in the mother liquor. Consequently, this approach achieves chemical purities exceeding 99.5% and optical purities greater than 99.9%, effectively eliminating the safety risks associated with isomeric impurities while streamlining the manufacturing workflow for cost reduction in API manufacturing.
Mechanistic Insights into Asymmetric Hydrogenation and Chiral Resolution
The core of this synthetic breakthrough lies in the synergistic combination of transition metal catalysis and classical resolution chemistry. The initial reduction step utilizes a sophisticated Ruthenium-based catalyst, specifically (S,S)-N-(p-toluenesulfonyl)-1,2-diphenylethanediamine (p-isopropylbenzene) ruthenium (II) chloride, to effect the asymmetric hydrogenation of the dihydroisoquinoline precursor. This catalyst facilitates the transfer of hydrogen from a formic acid/triethylamine azeotrope to the imine bond with high stereoselectivity, establishing the initial chiral center with a strong preference for the R-configuration.
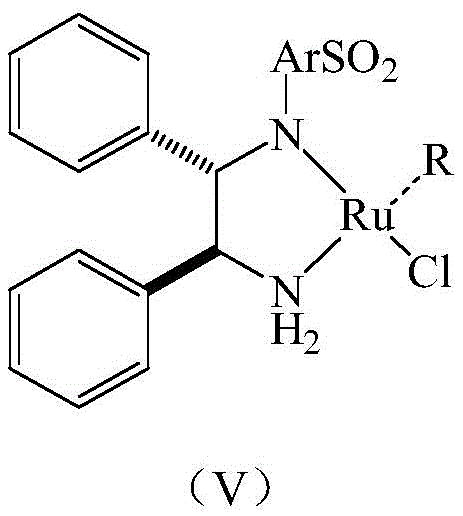
However, the true innovation is the subsequent treatment with chiral organic acids. By adding acids like D-camphoric acid or diacetyl-D-tartaric acid to the crude mixture, the process leverages the differences in solubility between diastereomeric salts. The desired R-isomer forms a less soluble salt that precipitates out of solution upon cooling, while the undesired S-isomer and other byproducts remain dissolved. This mechanism allows for a dramatic upgrade in purity without the need for expensive chiral HPLC columns, making it an ideal strategy for the commercial scale-up of complex pharmaceutical intermediates. The result is a highly pure intermediate that serves as a robust foundation for the subsequent N-alkylation and condensation steps required to build the final bis-benzylisoquinoline structure.
How to Synthesize Tetrahydrobenzyl Isoquinoline Efficiently
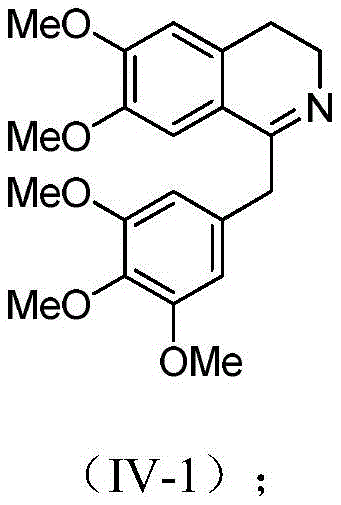
The operational protocol for this synthesis is designed for reproducibility and scalability, moving seamlessly from laboratory optimization to pilot plant execution. The process begins with the preparation of a catalyst stock solution, followed by the addition of the dihydroisoquinoline substrate in a suitable solvent such as dichloromethane or acetonitrile. After the hydrogenation is complete, the reaction is quenched, and the organic phase is concentrated. The critical resolution step involves dissolving the residue in an alcohol solvent, adding the chiral acid, and inducing crystallization through controlled cooling. For a detailed breakdown of the specific reaction conditions and stoichiometry, please refer to the visual guide below which outlines the transformation of compound IV-1 to I-1.
- Perform asymmetric hydrogenation of the dihydroisoquinoline precursor using a specific Ruthenium catalyst and formic acid/triethylamine mixture.
- Add a chiral organic acid (e.g., D-tartaric acid) to the crude mixture to form a diastereomeric salt.
- Crystallize, filter, and liberate the free base using alkali to obtain the target compound with >99.9% optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits beyond mere technical superiority. The primary advantage lies in the drastic simplification of the purification train. By achieving high purity through crystallization rather than chromatography, manufacturers can significantly reduce solvent consumption, waste generation, and processing time. This efficiency translates directly into a more stable supply chain, as the reliance on specialized purification media is eliminated, reducing the risk of bottlenecks that often plague the production of high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps results in significant operational savings. Traditional methods often require expensive silica gel or chiral columns which have limited lifespans and high replacement costs. By shifting to a crystallization-based purification driven by chiral acid salt formation, the process utilizes commodity chemicals and standard filtration equipment. This shift lowers the cost of goods sold (COGS) and improves the overall margin profile for the intermediate, allowing for more competitive pricing in the global market without compromising on the stringent quality standards required for neuromuscular blockers.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted drug supply. The high yields reported, often exceeding 90% even after the rigorous purification steps, mean that less raw material is wasted. Furthermore, the use of readily available chiral acids like D-tartaric acid ensures that the supply of resolving agents is secure and not subject to the volatility often seen with exotic proprietary reagents. This reliability reduces lead time for high-purity pharmaceutical intermediates and provides a buffer against market fluctuations.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process is inherently greener. The reduction in solvent usage and the avoidance of heavy metal contamination (since the catalyst is used in low loading and effectively removed) simplify waste treatment protocols. The scalability of crystallization processes is well-understood in the chemical industry, allowing for seamless transition from kilogram to ton-scale production. This ease of scale-up ensures that manufacturers can rapidly respond to increased market demand for Mivacurium and Cisatracurium, securing their position as a reliable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy techniques in terms of purity and yield.
Q: How does this method improve optical purity compared to prior art?
A: Unlike conventional methods yielding only 95% ee, this process utilizes chiral acid salt formation to upgrade optical purity to over 99.9%, critical for patient safety.
Q: What catalyst is used in the hydrogenation step?
A: The process employs (S,S)-N-(p-toluenesulfonyl)-1,2-diphenylethanediamine (p-isopropylbenzene) ruthenium (II) chloride for efficient asymmetric reduction.
Q: Is this process suitable for industrial scale-up?
A: Yes, the method avoids complex chromatography, relying on crystallization which is highly scalable and cost-effective for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrobenzyl Isoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of neuromuscular blocker intermediates requires not just chemical expertise but a deep commitment to quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. We understand that for compounds like the tetrahydrobenzyl isoquinoline derivatives, maintaining stringent purity specifications is non-negotiable. Our rigorous QC labs are equipped to verify optical purity levels exceeding 99.9%, guaranteeing that every batch meets the safety profiles necessary for clinical applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to this crystallization-based purification method. We encourage you to contact us for specific COA data and route feasibility assessments to ensure that your project timelines and quality goals are met with precision and reliability.
