Advanced Asymmetric Hydrogenation for High-Purity L-5-MTHF Calcium Production
Advanced Asymmetric Hydrogenation for High-Purity L-5-MTHF Calcium Production
The global demand for bioactive folate forms has surged as the pharmaceutical and nutraceutical industries recognize the superior efficacy of L-5-methyltetrahydrofolic acid (L-5-MTHF) over synthetic folic acid. A pivotal advancement in this sector is detailed in patent CN113603691A, which discloses a robust preparation process for L-5-methyltetrahydrofolic acid calcium. This technology addresses long-standing challenges in stereoselectivity and yield by employing an asymmetric catalytic hydrogenation strategy. Unlike conventional reduction methods that generate racemic mixtures requiring complex resolution, this novel route utilizes a specialized Rhodium-ligand catalyst system to directly synthesize the bioactive (6S)-isomer with exceptional diastereomeric excess. For R&D directors and procurement specialists, this patent represents a significant leap forward, offering a pathway to high-purity intermediates that aligns with stringent regulatory standards for active pharmaceutical ingredients and premium vitamin supplements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of tetrahydrofolic acid derivatives has relied heavily on non-selective chemical reduction techniques, such as using sodium borohydride or catalytic hydrogenation with noble metals like palladium on carbon without chiral control. These traditional approaches suffer from a fundamental flaw: they reduce the prochiral imine moiety of folic acid to produce a nearly 1:1 mixture of (6S) and (6R) diastereomers. Since the (6R)-isomer possesses negligible biological activity and may even accumulate in the central nervous system causing toxicity, extensive and costly chiral resolution steps are mandatory. Furthermore, chemical reductants like sodium borohydride often require large excesses, leading to significant chemical waste, alkalinity issues that promote side reactions, and ultimately lower overall yields and purity profiles that complicate downstream purification.
The Novel Approach
The process outlined in the patent introduces a paradigm shift by integrating asymmetric catalysis directly into the reduction step. By utilizing a Rhodium precatalyst coordinated with specific chiral ligands, the reaction achieves high conversion rates of folic acid (>92%) while simultaneously establishing the critical (6S) stereochemistry with a diastereomeric excess exceeding 86% in the crude product. This high initial selectivity drastically simplifies the subsequent purification workflow. The process flow is streamlined into four distinct stages, as illustrated below, moving from asymmetric hydrogenation to methylation, reduction, and final salt formation. This integrated approach not only minimizes the consumption of expensive chiral resolving agents but also enhances the overall material throughput, making it a highly attractive option for cost reduction in vitamin manufacturing.
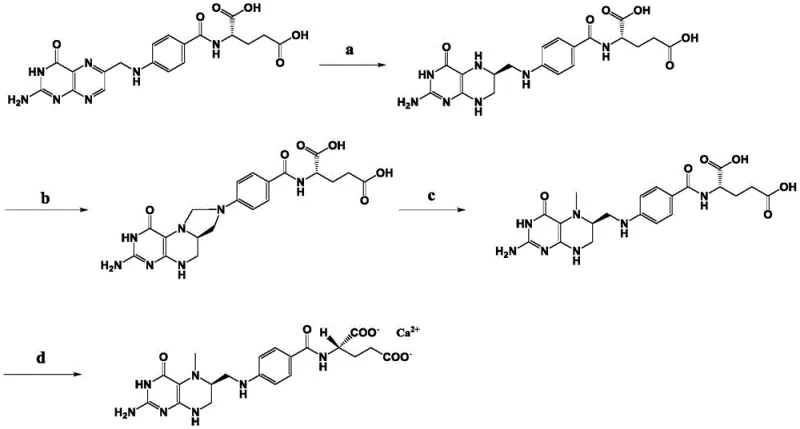
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
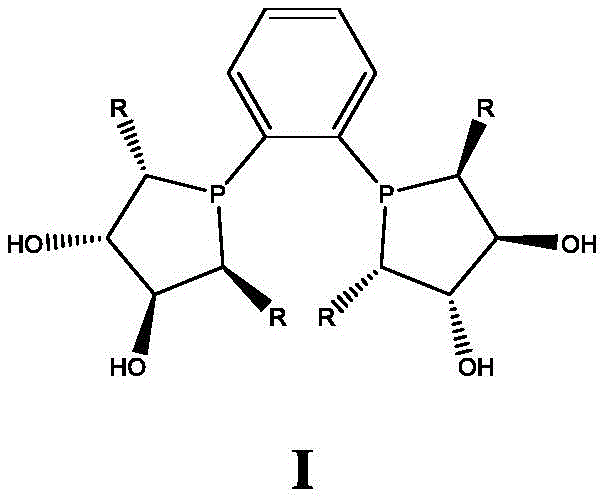
The cornerstone of this technological breakthrough is the catalyst system, which comprises a Rhodium precatalyst and a novel class of chiral diphosphine ligands defined by Formula I. The structural rigidity and specific electronic properties of these ligands create a highly defined chiral environment around the metal center. When the folic acid substrate coordinates to this complex, the ligand sterics direct the approach of molecular hydrogen, ensuring that hydride transfer occurs preferentially to one face of the prochiral double bond. This precise spatial control is what drives the high diastereoselectivity observed in the reaction. The ligand structure, featuring a biphenyl backbone linked to phosphorylated cyclopentane rings with specific hydroxyl and alkyl substituents, is critical for maintaining catalyst stability and activity under the reaction conditions of 30 to 50 bar hydrogen pressure and temperatures ranging from 70 to 80°C.
Beyond the primary reduction, the process incorporates a sophisticated purification mechanism via salt formation. After the initial hydrogenation, the crude (6S)-tetrahydrofolic acid is treated with benzenesulfonic acid or p-toluenesulfonic acid. This step exploits the solubility differences between the diastereomeric salts, allowing the desired (6S)-isomer to crystallize out while leaving impurities and the minor (6R)-isomer in the mother liquor. Through this recrystallization, the diastereomeric excess is boosted to over 97%, ensuring that the final L-5-methyltetrahydrofolic acid calcium meets the rigorous purity specifications required for human consumption. This dual strategy of kinetic control during catalysis and thermodynamic control during crystallization provides a robust defense against impurity formation.
How to Synthesize L-5-Methyltetrahydrofolic Acid Calcium Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly during the asymmetric hydrogenation phase where catalyst activation and substrate solubility are paramount. The process begins with the in-situ formation of the active catalyst species in an alcohol solvent, followed by its introduction to a buffered aqueous solution of folic acid. Maintaining the pH within a narrow window (7.1-7.5) is crucial for substrate stability and catalyst performance. Following the hydrogenation, the workup involves precise pH adjustments to isolate the intermediate, which is then subjected to methylation using formic acid and subsequent reduction. The detailed standardized operating procedures for each stage, including specific molar ratios, temperature controls, and isolation techniques, are essential for reproducing the high yields and purity reported in the patent examples.
- Perform asymmetric catalytic hydrogenation of folic acid using a Rhodium precatalyst and chiral ligand to obtain (6S)-tetrahydrofolic acid with high diastereomeric excess.
- React the purified (6S)-tetrahydrofolic acid with formic acid and trifluoroacetic acid to form (6S)-5,10-methylenetetrahydrofolic acid.
- Reduce the methylene intermediate using sodium borohydride to generate L-5-methyltetrahydrofolic acid.
- React the final acid with food-grade calcium chloride to precipitate and purify the L-5-methyltetrahydrofolic acid calcium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric hydrogenation technology translates into tangible strategic benefits beyond mere technical superiority. The elimination of inefficient chiral resolution steps significantly reduces the consumption of raw materials and solvents, directly impacting the cost of goods sold. Furthermore, the reliance on catalytic hydrogenation, a unit operation that is well-established and easily scalable in the fine chemical industry, mitigates the risks associated with introducing entirely new processing equipment. This compatibility with existing infrastructure ensures a smoother technology transfer and faster time-to-market for commercial production, addressing the critical need for reducing lead time for high-purity vitamin intermediates in a volatile global market.
- Cost Reduction in Manufacturing: The primary economic driver of this process is the drastic reduction in waste generation and reagent consumption. By avoiding the use of stoichiometric amounts of expensive chiral resolving agents and minimizing the excess of chemical reductants like sodium borohydride, the overall material cost is significantly lowered. Additionally, the high yield of the final calcium salt, reported to be over 80% based on folic acid, means that less starting material is required to produce the same amount of finished product, thereby optimizing the utilization of capital and resources throughout the manufacturing value chain.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system contributes to greater batch-to-batch consistency, a critical factor for maintaining uninterrupted supply to downstream pharmaceutical customers. The use of stable intermediates and the ability to purify via crystallization rather than complex chromatography enhances the reliability of the production schedule. This stability ensures that suppliers can meet large-volume orders with predictable quality, reducing the risk of supply disruptions that often plague processes reliant on sensitive biological enzymes or unstable chemical reagents.
- Scalability and Environmental Compliance: From an environmental perspective, the process offers substantial advantages by reducing the E-factor (mass of waste per mass of product). The catalytic nature of the key step means that the heavy metal load is minimized compared to stoichiometric reductions, simplifying wastewater treatment and compliance with increasingly strict environmental regulations. The scalability of high-pressure hydrogenation is well-understood in the industry, allowing for seamless transition from pilot plant to multi-ton commercial scale-up of complex folate derivatives without the need for bespoke reactor designs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of L-5-methyltetrahydrofolic acid calcium using this advanced methodology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's potential impact on your supply chain and product development pipelines.
Q: How does the new asymmetric hydrogenation method improve upon traditional chemical reduction?
A: Traditional methods using sodium borohydride often result in nearly equal mixtures of (6S) and (6R) isomers, requiring expensive and inefficient chiral resolution. The patented asymmetric hydrogenation method directly produces the bioactive (6S)-isomer with a diastereomeric excess exceeding 86% in the crude product, which can be further enriched to over 97% through simple salt formation and crystallization, significantly reducing waste and processing steps.
Q: What represents the key technological breakthrough in this synthesis route?
A: The core innovation lies in the use of a specific Rhodium-based catalyst system paired with novel chiral phosphine ligands (Formula I). This catalyst system enables highly stereoselective hydrogenation of the prochiral imine moiety in folic acid under relatively mild conditions (30-50 bar H2, 70-80°C), achieving conversion rates above 92% while maintaining exceptional stereocontrol that was previously difficult to attain on an industrial scale.
Q: Why is the calcium salt form preferred for L-5-methyltetrahydrofolic acid?
A: L-5-methyltetrahydrofolic acid is inherently sensitive to air and moisture and exhibits low solubility in water, making the free acid form unstable for storage and transport. Converting it into the calcium salt significantly enhances its stability against oxidation and degradation, improves its handling properties, and ensures a longer shelf life, which is critical for both pharmaceutical formulations and nutraceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-5-MTHF Calcium Supplier
The technical potential of this asymmetric hydrogenation route is immense, offering a clear path to producing high-quality folate intermediates that meet the evolving needs of the global health market. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into efficient industrial realities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against international pharmacopoeia standards, guaranteeing that our clients receive materials that are safe, effective, and consistent.
We invite you to collaborate with us to leverage this cutting-edge technology for your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can drive value and efficiency for your organization.
