Advanced Catalytic Bromination for High-Purity Antiviral Pyrazine Intermediates and Commercial Scale-Up
Introduction to Next-Generation Antiviral Intermediate Manufacturing
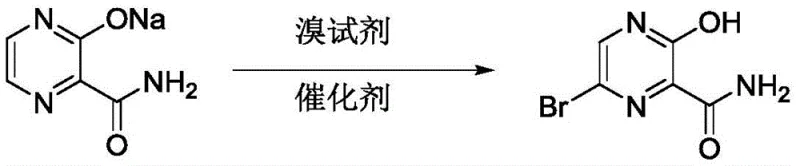
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for critical antiviral intermediates, particularly those serving as precursors for novel influenza treatments. Patent CN113234030A introduces a transformative preparation method for 6-bromo-3-hydroxy-2-pyrazinecarboxamide, a pivotal building block in the synthesis of potent pyrazine-based therapeutics. This innovation addresses long-standing inefficiencies in heterocyclic bromination by leveraging a refined catalytic system that utilizes sodium 2-carbamoylpyrazine-3-hydroxyl as the starting material. By integrating a specific phase transfer catalyst with a controlled dropwise addition of bromine water, the disclosed technology achieves a remarkable acceleration in reaction kinetics while maintaining exceptional product integrity. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the supply chain for high-value antiviral APIs, ensuring both cost-efficiency and consistent quality in a competitive market landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-bromo-3-hydroxy-2-pyrazinecarboxamide has been plagued by operational complexities and suboptimal efficiency metrics that hinder large-scale adoption. Traditional protocols often rely on the continuous supplementation of tetrabutylammonium bromide throughout the reaction course, a practice that not only extends the overall processing time but also complicates the stoichiometric control of the bromination event. Furthermore, existing methodologies frequently necessitate the use of specialized and costly equipment, such as microchannel reactors, to achieve acceptable yields, which creates a significant barrier to entry for standard manufacturing facilities. The reliance on such capital-intensive infrastructure limits flexibility and increases the fixed costs associated with production, making it difficult to respond agilely to fluctuating market demands for antiviral intermediates. Additionally, the prolonged reaction times inherent in these older processes increase the risk of side reactions and impurity formation, thereby burdening downstream purification units and reducing the overall atom economy of the synthesis.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113234030A offers a streamlined and economically superior alternative that simplifies the production workflow while drastically enhancing output. By employing a single, optimized charge of tetrabutylammonium bromide coupled with the precise dropwise addition of bromine water, the new process eliminates the need for continuous reagent feeding, thereby shortening the total reaction cycle by more than three hours. This approach allows for the use of standard stirred-tank reactors rather than expensive micro-reactors, significantly lowering the capital expenditure required for implementation. The strategic adjustment of reaction parameters, including temperature control between 30°C and 35°C, ensures that the exothermic nature of the bromination is managed effectively without compromising the structural integrity of the sensitive pyrazine ring. Consequently, this novel route delivers a substantial improvement in yield, reported to be approximately 20% higher than traditional methods, while simultaneously generating a cleaner crude product profile that facilitates easier isolation and purification.
Mechanistic Insights into Catalytic Electrophilic Bromination
The core of this technological advancement lies in the sophisticated interplay between the phase transfer catalyst and the brominating agent within a polar organic solvent matrix. The reaction proceeds via an electrophilic aromatic substitution mechanism where the electron-rich pyrazine ring undergoes selective bromination at the 6-position. The presence of tetrabutylammonium bromide acts as a crucial phase transfer catalyst, facilitating the transport of bromide ions into the organic phase where they interact with the molecular bromine to generate the active electrophilic species. This catalytic cycle enhances the local concentration of the reactive brominating agent near the substrate, thereby accelerating the rate-determining step of the substitution. The use of a mixed solvent system comprising acetonitrile and methanol is not merely for solubility; it stabilizes the transition state and helps dissipate the heat generated during the exothermic addition, preventing thermal degradation of the product. This precise control over the reaction microenvironment is what allows for the high regioselectivity observed, minimizing the formation of poly-brominated byproducts or isomers that typically plague non-catalyzed halogenation reactions.
Furthermore, the mechanism inherently supports superior impurity control, which is a critical parameter for pharmaceutical grade intermediates. The dropwise addition mode of the bromine water ensures that the concentration of free bromine remains low throughout the reaction, preventing over-bromination and oxidative degradation of the amide functionality. By maintaining the reaction temperature within a narrow window of 30°C to 35°C, the kinetic energy of the system is kept sufficient for the desired substitution but insufficient to trigger competing decomposition pathways. The subsequent crystallization step, induced by the addition of cooling water to the reaction mixture, leverages the differential solubility of the product versus potential impurities in the aqueous-organic mixture. This 'crash out' crystallization effectively purges soluble impurities and residual catalyst from the crystal lattice, yielding a light yellow solid of high purity without the need for extensive chromatographic purification, thus aligning perfectly with the principles of green chemistry and cost-effective manufacturing.
How to Synthesize 6-Bromo-3-Hydroxy-2-Pyrazinecarboxamide Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding solvent ratios and addition rates to maximize the benefits of the catalytic system. The process begins with the dissolution of the sodium salt precursor and the catalyst in a specific mass ratio of acetonitrile to methanol, creating a homogeneous reaction medium that supports rapid mass transfer. Operators must exercise precise control over the滴加 (dropwise addition) of the bromine reagent to manage the exotherm and maintain the stoichiometric balance required for high conversion. The detailed standardized operating procedures, including specific stirring speeds and crystallization timelines, are essential for reproducing the high yields reported in the patent data. For a comprehensive breakdown of the exact experimental steps and safety protocols required for execution, please refer to the technical guide below.
- Dissolve sodium 2-carbamoylpyrazine-3-hydroxyl and tetrabutylammonium bromide catalyst in a mixed solvent system of acetonitrile and methanol.
- Maintain the reaction temperature between 30°C and 35°C while slowly dropwise adding bromine water to the stirred mixture.
- Upon completion, cool the reaction mixture to 0-10°C, induce crystallization with cooling water, and filter to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis route offers profound benefits that extend far beyond simple chemical transformation, directly impacting the bottom line and supply chain resilience. The elimination of continuous catalyst supplementation and the reduction in reaction time translate directly into increased throughput capacity for existing manufacturing assets, allowing suppliers to meet surging demand for antiviral intermediates without significant capital investment in new reactor trains. The simplified work-up procedure, which relies on straightforward filtration and drying rather than complex extraction or chromatography, significantly reduces the consumption of auxiliary materials and solvents, leading to a leaner and more cost-effective production model. Moreover, the robustness of the process against minor variations in operating conditions enhances supply continuity, reducing the risk of batch failures that can disrupt downstream API synthesis schedules. This reliability is paramount for procurement managers tasked with securing long-term contracts for critical healthcare ingredients in a volatile global market.
- Cost Reduction in Manufacturing: The economic implications of this process are driven by the drastic simplification of the operational workflow and the enhanced efficiency of raw material utilization. By removing the requirement for expensive microchannel reactors and replacing them with standard vessel technology, the barrier to entry for production is lowered, fostering a more competitive supplier landscape. The significant reduction in reaction time means that utility costs, such as heating, cooling, and agitation, are incurred for a shorter duration per kilogram of product, directly lowering the variable cost of goods sold. Additionally, the improved yield ensures that less starting material is wasted, maximizing the return on investment for every ton of precursor purchased and effectively reducing the cost per unit of the final active intermediate.
- Enhanced Supply Chain Reliability: In the context of global pharmaceutical supply chains, the simplicity and robustness of this synthesis method serve as a powerful risk mitigation tool. The reliance on readily available and commodity-grade reagents like bromine water and common organic solvents minimizes the risk of supply bottlenecks associated with exotic or highly regulated specialty chemicals. The ability to produce high-purity material using standard equipment means that multiple qualified manufacturers can potentially adopt this route, diversifying the supply base and reducing dependency on single-source vendors. This redundancy is crucial for ensuring uninterrupted availability of key antiviral building blocks, especially during public health emergencies when demand spikes unexpectedly and supply chain agility is tested to its limits.
- Scalability and Environmental Compliance: The environmental footprint of chemical manufacturing is increasingly scrutinized, and this process offers a cleaner alternative that aligns with modern sustainability goals. The use of a closed-loop bromination system with controlled addition minimizes the release of hazardous bromine vapors, enhancing workplace safety and reducing the load on scrubber systems. The aqueous work-up and crystallization strategy generates less hazardous organic waste compared to traditional extraction-heavy methods, simplifying wastewater treatment and disposal compliance. Furthermore, the demonstrated scalability from laboratory to industrial scales without the need for specialized flow chemistry equipment ensures that production can be ramped up rapidly to meet commercial volumes while maintaining strict adherence to environmental regulations and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific bromination technology. These insights are derived directly from the comparative data and process descriptions found within the patent literature, providing a factual basis for evaluating its suitability for your specific application. Understanding these nuances is essential for technical teams assessing the feasibility of technology transfer and for commercial teams negotiating supply agreements based on process efficiency. We encourage stakeholders to review these points carefully to fully appreciate the value proposition of this advanced synthetic route.
Q: What are the critical advantages of using bromine water over traditional brominating agents in this synthesis?
A: The use of bromine water in conjunction with a phase transfer catalyst significantly reduces reaction time by over 3 hours compared to conventional methods, while simultaneously improving raw material conversion rates and overall yield by approximately 20%.
Q: How does the solvent system impact the purity of the final pyrazine derivative?
A: The optimized binary solvent system comprising acetonitrile and methanol ensures optimal solubility of the sodium salt precursor while facilitating efficient heat dissipation during the exothermic bromination, resulting in superior impurity profiles.
Q: Is this process suitable for large-scale industrial manufacturing of antiviral intermediates?
A: Yes, the process eliminates the need for expensive microchannel reactors and utilizes standard stirred tank reactors with simple ice-water crystallization, making it highly adaptable for multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Bromo-3-Hydroxy-2-Pyrazinecarboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this catalytic bromination process are fully realized in a GMP-compliant environment. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle reactive chemistries safely and efficiently, allowing us to offer a secure and reliable supply of this critical antiviral precursor to our global partners.
We invite you to collaborate with us to optimize your supply chain and reduce your overall cost of goods through the adoption of this superior manufacturing route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and technical support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
