Advanced Semi-Synthetic Route for Lipophilic Camptothecin Derivatives Enhancing Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways for potent anticancer agents, particularly highly lipophilic camptothecin derivatives (HLCDs) such as Karenitecin™ (BNP1350). Patent CN1894257A introduces a transformative semi-synthetic methodology that addresses critical bottlenecks in the production of these silicon-containing compounds. By shifting away from unstable aldehyde precursors to stable silylated heterocyclic intermediates, this innovation enables a modified Minisci-type alkylation that significantly enhances both yield and operational simplicity. For R&D directors and process chemists, this represents a pivotal advancement in optimizing the impurity profile and scalability of complex oncology intermediates. The protocol detailed herein leverages commercially available starting materials and standard reaction conditions to achieve superior outcomes compared to legacy methods. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is essential for evaluating the commercial viability of next-generation API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of silicon-containing HLCDs relied heavily on reacting camptothecin with trimethylsilyl aldehyde in the presence of metal sulfates and strong oxidizers. While effective on a laboratory scale, these conventional methods suffered from inherent instability of the aldehyde reactant, leading to inconsistent reaction kinetics and the generation of difficult-to-remove impurities. Previous patented processes typically resulted in crude theoretical yields ranging from 50% to 60%, which further diminished to merely 25% to 35% after rigorous column chromatography purification. This reliance on chromatographic separation not only inflated manufacturing costs but also introduced significant challenges in solvent recovery and waste management, making large-scale production economically unfeasible. Furthermore, the instability of the reactants often necessitated precise control over addition rates and temperatures, increasing the risk of batch failure during commercial scale-up. These limitations underscored the urgent need for a more robust precursor that could withstand the harsh oxidative conditions of the Minisci reaction without degrading.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these issues by utilizing a trialkylsilyl-alkyl-cyclic compound, specifically 2-(2-trimethylsilylethyl)-1,3-dioxolane, as the key alkylating agent. This intermediate is synthesized via a straightforward Grignard reaction between 2-bromoethyl-1,3-dioxolane and chlorotrimethylsilane, creating a stable species that does not suffer from the volatility or degradation issues of its aldehyde counterpart. 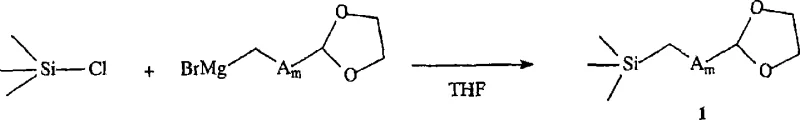 This stability allows for a cleaner reaction profile, minimizing the formation of side products derived from co-solvent interactions. Consequently, the new method demonstrates a crude theoretical yield of approximately 80%, with isolated yields reaching 45% to 50% after simple double crystallization. This shift from chromatography to crystallization is a game-changer for cost reduction in API manufacturing, as it drastically simplifies downstream processing and improves overall throughput. The ability to use standard solvents like tetrahydrofuran and tert-butanol further enhances the practicality of this route for industrial applications.
This stability allows for a cleaner reaction profile, minimizing the formation of side products derived from co-solvent interactions. Consequently, the new method demonstrates a crude theoretical yield of approximately 80%, with isolated yields reaching 45% to 50% after simple double crystallization. This shift from chromatography to crystallization is a game-changer for cost reduction in API manufacturing, as it drastically simplifies downstream processing and improves overall throughput. The ability to use standard solvents like tetrahydrofuran and tert-butanol further enhances the practicality of this route for industrial applications.
Mechanistic Insights into Modified Minisci-Type Alkylation
The core of this synthesis lies in the modified Minisci-type hemolytic alkylation, a radical-based substitution reaction that targets the electron-deficient positions of the camptothecin scaffold. In this specific transformation, the reaction is initiated by the generation of radicals from the silylated heterocyclic intermediate using a ferrous sulfate and hydrogen peroxide system (Fenton-like chemistry). The iron(II) catalyst facilitates the homolytic cleavage of the peroxide bond, generating hydroxyl radicals that abstract a hydrogen atom from the alkyl chain of the silylated intermediate. This creates a carbon-centered radical which subsequently attacks the protonated N-1 position of the camptothecin ring system, preferentially substituting at the 7-position due to electronic and steric factors. 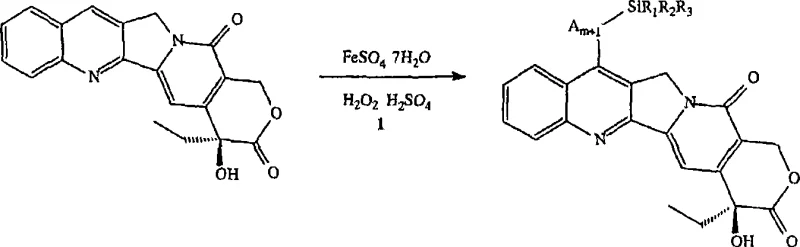 The use of strong acid, such as 30% sulfuric acid, is critical to protonate the quinoline nitrogen, thereby activating the ring towards nucleophilic radical attack. This mechanistic pathway ensures high regioselectivity, minimizing the formation of isomeric by-products that often complicate purification in traditional electrophilic aromatic substitutions. Understanding this radical mechanism is vital for R&D teams aiming to replicate or optimize the process, as slight deviations in oxidant addition or pH can alter the radical flux and impact the final impurity profile.
The use of strong acid, such as 30% sulfuric acid, is critical to protonate the quinoline nitrogen, thereby activating the ring towards nucleophilic radical attack. This mechanistic pathway ensures high regioselectivity, minimizing the formation of isomeric by-products that often complicate purification in traditional electrophilic aromatic substitutions. Understanding this radical mechanism is vital for R&D teams aiming to replicate or optimize the process, as slight deviations in oxidant addition or pH can alter the radical flux and impact the final impurity profile.
Controlling the impurity profile is paramount for pharmaceutical intermediates intended for clinical use. The novel process inherently reduces impurity formation by avoiding the in situ generation of unstable aldehyde species that can polymerize or react non-specifically with the solvent matrix. By employing a pre-formed, stable dioxolane-protected intermediate, the reaction mixture remains cleaner throughout the oxidation phase. Post-reaction workup involves quenching excess peroxide with sodium sulfite and extracting the product into organic phases like dichloromethane, leaving polar inorganic salts and acidic by-products in the aqueous layer. The final purification via recrystallization from ethanol and DMF effectively removes trace organic impurities and residual metals, ensuring the final product meets stringent purity specifications of over 99%. This robust control over the chemical environment translates directly to reduced analytical burden and faster release times for quality control laboratories.
How to Synthesize 7-(2-trimethylsilylethyl)camptothecin Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing high-purity BNP1350 suitable for further drug development. The process begins with the preparation of the silylated building block, followed by the critical coupling step with the camptothecin core. Operators must maintain strict anhydrous conditions during the Grignard formation to prevent premature quenching of the organometallic species. Subsequent steps involve careful temperature control during the exothermic oxidation phase to prevent thermal runaway and ensure consistent radical generation. The detailed standardized synthetic steps see the guide below for specific molar ratios and timing.
- Preparation of Silylated Intermediate: React 2-bromoethyl-1,3-dioxolane with magnesium and iodine in anhydrous THF to form a Grignard reagent, then react with chlorotrimethylsilane to produce 2-(2-trimethylsilylethyl)-1,3-dioxolane.
- Minisci Alkylation Reaction: Dissolve camptothecin and ferrous sulfate heptahydrate in 30% sulfuric acid, add the silylated intermediate in tert-butanol, and slowly introduce 30% hydrogen peroxide while maintaining temperature between 15°C and 20°C.
- Purification and Isolation: Quench excess oxidant with sodium sulfite, extract with dichloromethane, dry over sodium sulfate, and purify the crude product through double recrystallization using ethanol and anhydrous DMF to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers substantial strategic benefits beyond mere chemical elegance. The elimination of column chromatography in favor of crystallization represents a massive reduction in solvent consumption and processing time, directly impacting the cost of goods sold. By utilizing stable, commercially available reagents like 2-bromoethyl-1,3-dioxolane and chlorotrimethylsilane, the supply chain becomes more resilient against raw material shortages that often plague specialized aldehyde suppliers. The improved yield profile means that less starting camptothecin is required to produce the same amount of final API, optimizing the utilization of this valuable natural product resource. Furthermore, the simplified workup procedure reduces the load on waste treatment facilities, aligning with increasingly strict environmental compliance standards in chemical manufacturing.
- Cost Reduction in Manufacturing: The most significant economic driver is the drastic simplification of the purification process. Replacing labor-intensive and solvent-heavy column chromatography with scalable crystallization steps significantly lowers operational expenditures. The increase in isolated yield from roughly 30% to nearly 50% effectively doubles the output per batch without requiring additional reactor volume, leading to substantial cost savings in fixed asset utilization. Additionally, the use of common industrial solvents like THF, MTBE, and cyclohexane avoids the need for exotic or highly regulated solvents, further streamlining procurement logistics and reducing material costs.
- Enhanced Supply Chain Reliability: The reliance on stable intermediates mitigates the risk of batch-to-batch variability caused by reactant degradation. Unlike TMS-aldehyde, which requires careful handling and storage to prevent polymerization, the silylated dioxolane intermediate is robust and can be stockpiled or sourced from multiple vendors without quality concerns. This stability ensures consistent production schedules and reduces the likelihood of delays caused by reagent failure. Moreover, the process tolerates standard commercial grades of camptothecin, allowing manufacturers to source raw materials from a broader range of suppliers in China, India, and Europe, thereby diversifying supply risk and negotiating better pricing terms.
- Scalability and Environmental Compliance: The one-pot nature of the intermediate synthesis and the straightforward aqueous workup of the final coupling reaction make this process highly amenable to scale-up from kilograms to metric tons. The reduction in silica gel usage eliminates a major solid waste stream, while the ability to recover and recycle solvents like dichloromethane and DMF improves the overall environmental footprint. This alignment with green chemistry principles not only reduces disposal costs but also facilitates regulatory approval in markets with stringent environmental guidelines. The robustness of the reaction conditions ensures that the process performs consistently across different reactor sizes, minimizing the tech transfer risks associated with moving from pilot plant to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived directly from the experimental data and comparative analysis provided in the patent documentation. These insights are intended to clarify the operational advantages and technical feasibility for potential partners and licensees evaluating this synthetic route.
Q: What is the primary advantage of this new synthesis method over previous patents?
A: The primary advantage is the significant improvement in yield and purification efficiency. While previous methods yielded only 25% to 35% after column chromatography, this novel process achieves approximately 80% crude theoretical yield and 45% to 50% isolated yield after double crystallization, eliminating the need for complex chromatographic separation.
Q: Why is the silylated heterocyclic compound preferred over TMS-aldehyde?
A: The silylated heterocyclic compound, specifically 2-(2-trimethylsilylethyl)-1,3-dioxolane, offers superior stability compared to trimethylsilyl aldehyde. This stability prevents the formation of unwanted by-products derived from co-solvents and ensures a more consistent reaction profile during the Minisci-type alkylation.
Q: What purity levels can be achieved with this process?
A: Through the described double recrystallization process using ethanol and anhydrous N,N-dimethylformamide, the final product (BNP1350) can achieve a purity of 99.12%, meeting stringent specifications required for active pharmaceutical ingredient intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Karenitecin Supplier
The technological breakthroughs detailed in Patent CN1894257A highlight the immense potential for optimizing the production of high-value oncology intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like modified Minisci alkylations are executed with precision and safety. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including the 99%+ purity benchmarks required for clinical grade materials. We understand the critical importance of supply continuity and cost efficiency in the pharmaceutical sector, and our engineering teams are dedicated to translating laboratory innovations into robust industrial processes.
We invite global partners to collaborate with us to leverage this advanced synthesis technology for their pipeline projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can accelerate your drug development timelines while optimizing your budget. Let us be your strategic partner in bringing life-saving camptothecin derivatives to the market efficiently and reliably.
