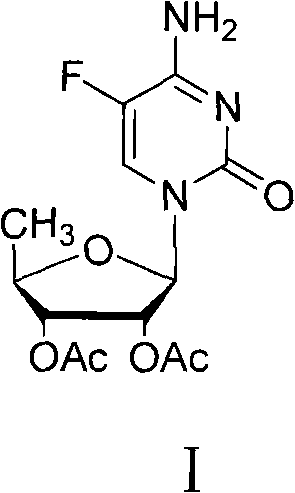
Advanced Synthesis of 5'-Deoxy-2',3'-diacetyl-5-fluorocytidine for Scalable API Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical oncology intermediates, and patent CN102190695B presents a transformative approach to manufacturing 5'-deoxy-2',3'-diacetyl-5-fluorocytidine. This compound serves as a pivotal precursor in the synthesis of Capecitabine, a widely prescribed oral chemotherapeutic agent. The disclosed innovation fundamentally alters the sequence of reagent addition during the glycosylation step, specifically optimizing the interaction between the silylated pyrimidine base and the Lewis acid catalyst. By prioritizing the mixing of anhydrous tin tetrachloride with the silylated base before introducing the sugar donor, the process circumvents the severe exothermic spikes and uncontrolled side reactions that have historically plagued this transformation. This strategic adjustment not only elevates the molar yield to over 95% but also ensures a final product purity exceeding 98%, thereby establishing a new benchmark for efficiency in nucleoside chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key nucleoside intermediate has been hindered by rigid operational constraints that compromise both safety and economic viability on an industrial scale. Traditional protocols, as documented in various international patents, dictate that the silylated base and the acetylated sugar must be mixed first, followed by the slow, dropwise addition of the Lewis acid catalyst at cryogenic temperatures ranging from -5°C to 5°C. This sequence creates a highly unstable reaction environment where the exotherm is difficult to manage, necessitating prolonged addition times that extend the overall production cycle significantly. Furthermore, the inability to maintain precise thermal control often triggers substantial N-isomerization, generating impurity levels between 7% and 15% which severely impacts the yield. The resulting crude product typically possesses low purity, mandating cumbersome downstream purification steps such as recrystallization that further erode process efficiency and increase solvent consumption.
The Novel Approach
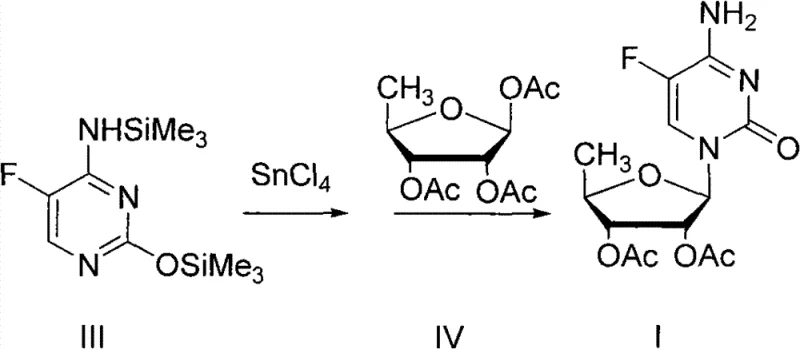
In a decisive break from established norms, the novel methodology described in the patent data reverses the addition order to achieve superior kinetic control and thermodynamic stability. Instead of adding the catalyst last, the process initiates by directly mixing the silylated base (Compound III) with anhydrous tin tetrachloride in an inert solvent, creating a stable activation complex prior to the introduction of the sugar moiety. This modification allows the reaction to proceed under much milder thermal conditions, ideally between -20°C and 40°C, effectively removing the need for energy-intensive cryogenic cooling systems. The outcome is a dramatic enhancement in process performance, with molar yields consistently surpassing 95% and N-isomerization impurities suppressed to below 2%. This streamlined workflow not only accelerates the reaction timeline but also delivers a high-purity intermediate that requires minimal post-reaction processing, representing a significant leap forward in manufacturing capability.
Mechanistic Insights into Lewis Acid Mediated Glycosylation
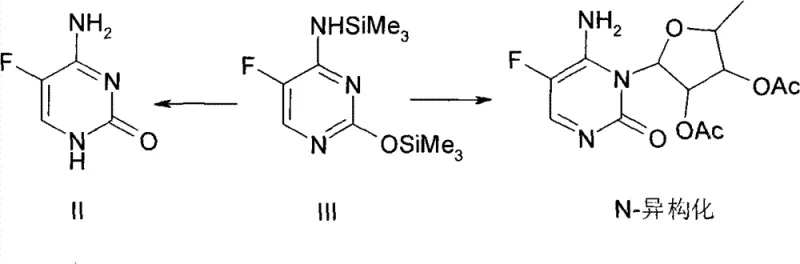
The success of this optimized protocol lies in the fundamental alteration of the reaction mechanism regarding the activation of the glycosyl donor and the stability of the nucleobase. In the conventional approach, the simultaneous presence of the sugar, base, and Lewis acid creates a chaotic environment where the catalyst may indiscriminately attack the silyl protecting groups on the base, leading to deprotection and the regeneration of the starting material (Compound II). However, by pre-mixing the Lewis acid with the silylated base, the system ensures that the catalyst is primarily engaged in activating the sugar donor (Compound IV) to form the reactive oxocarbenium ion species without compromising the integrity of the nucleobase. This selective activation pathway ensures that the coupling reaction proceeds with high regioselectivity, favoring the formation of the desired N1-glycosidic bond while minimizing the formation of N3-isomers. The stability of Compound III in the presence of the Lewis acid, even at elevated temperatures, is a critical factor that enables this broader operating window.
Furthermore, the suppression of N-isomerization is directly linked to the controlled generation of the electrophilic species and the immediate availability of the nucleophile in its activated state. When the Lewis acid is added last in traditional methods, local concentration gradients can lead to hotspots of high acidity that promote the migration of the glycosyl group or the scrambling of stereochemistry. The new method homogenizes the catalytic environment, ensuring that the reactive sugar intermediate encounters the base under uniform conditions that favor the thermodynamic product. This mechanistic clarity explains the drastic reduction in impurity profiles, dropping N-isomer content from double digits to less than 2%, which is crucial for meeting the stringent quality specifications required for pharmaceutical intermediates. The elimination of the deprotection pathway also means that nearly 100% of the starting base is converted, maximizing atom economy and reducing raw material waste.
How to Synthesize 5'-Deoxy-2',3'-diacetyl-5-fluorocytidine Efficiently
The implementation of this synthesis route offers a straightforward yet highly effective protocol for laboratory and pilot-scale operations aiming to produce high-quality nucleoside intermediates. The procedure begins with the dissolution of the silylated fluoropyrimidine in a suitable inert organic solvent, such as dichloromethane or chloroform, followed by the immediate addition of the Lewis acid catalyst to form a homogeneous mixture. Once this activation phase is complete, the acetylated ribose derivative is introduced to the reaction vessel, where the glycosylation proceeds rapidly without the need for rigorous temperature monitoring below 5°C. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during execution.
- Dissolve the silylated base (Compound III) in an inert organic solvent such as dichloromethane and mix directly with anhydrous tin tetrachloride.
- Add the sugar donor (Compound IV) to the pre-mixed Lewis acid solution without strict low-temperature control, maintaining a range of -20 to 40°C.
- Quench the reaction with water and sodium bicarbonate, followed by filtration and solvent removal to obtain the product with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized synthetic route translates into tangible operational improvements that directly impact the bottom line and supply reliability. The elimination of strict cryogenic requirements removes a significant bottleneck in production scheduling, allowing facilities to utilize standard reactor setups without specialized cooling infrastructure. This flexibility enhances the overall throughput of the manufacturing plant, enabling faster turnaround times for batch completion and reducing the dependency on complex utility systems that are prone to failure. Moreover, the substantial increase in yield means that less raw material is required to produce the same quantity of final product, effectively lowering the cost of goods sold and mitigating the risk associated with volatile raw material pricing markets.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing workflow. Since the reaction yields a product with purity exceeding 98% directly after workup, the costly and time-consuming steps of recrystallization or column chromatography are rendered unnecessary. This reduction in unit operations leads to significant savings in solvent consumption, energy usage for heating and cooling, and labor hours required for purification. Additionally, the higher conversion rate minimizes the loss of expensive starting materials, ensuring that every kilogram of input generates maximum output value, which is critical for maintaining competitive pricing in the global API intermediate market.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the robustness of this method ensures a more predictable and consistent supply of critical intermediates. The tolerance for a wider temperature range reduces the likelihood of batch failures due to minor thermal fluctuations, which are common in large-scale reactors where heat transfer is less efficient than in the lab. This reliability allows supply chain planners to commit to tighter delivery schedules with greater confidence, knowing that the production process is less susceptible to the variability that often plagues sensitive chemical transformations. Consequently, manufacturers can maintain lower safety stock levels while still meeting customer demand, optimizing working capital and storage capacity.
- Scalability and Environmental Compliance: The scalability of this process is inherently superior due to the mitigation of violent exotherms, which pose safety risks and equipment limitations in large vessels. By controlling the heat release through the modified addition order, the process becomes safer to operate at the multi-ton scale, facilitating easier technology transfer from pilot plants to commercial production lines. Furthermore, the reduction in solvent usage and the elimination of extensive purification steps contribute to a smaller environmental footprint, aligning with increasingly stringent global regulations on waste disposal and emissions. This compliance advantage reduces the regulatory burden on the manufacturing site and enhances the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this advanced synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational advantages and quality improvements associated with the new protocol for stakeholders evaluating its adoption. Understanding these details is essential for making informed decisions about process integration and supplier qualification.
Q: How does the new addition order improve product purity compared to conventional methods?
A: By mixing the Lewis acid with the silylated base first, the process prevents the premature deprotection of the base and significantly reduces N-isomerization impurities from 7-15% down to less than 2%.
Q: What are the temperature requirements for this optimized glycosylation reaction?
A: Unlike prior art requiring strict cryogenic conditions (-5 to 5°C), this method operates efficiently between -20°C and 40°C, allowing for room temperature processing which simplifies industrial scale-up.
Q: Is secondary purification required for the final intermediate?
A: No, the optimized protocol yields product purity exceeding 98% directly after workup, eliminating the need for energy-intensive recrystallization or column chromatography steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5'-Deoxy-2',3'-diacetyl-5-fluorocytidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development and commercialization of oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 5'-deoxy-2',3'-diacetyl-5-fluorocytidine adheres to the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall production costs.
