Advanced Acid-Assisted Crystallization for High-Purity Temozolomide Manufacturing and Commercial Scale-Up
Advanced Acid-Assisted Crystallization for High-Purity Temozolomide Manufacturing and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust purification methodologies for potent oncology agents, particularly for alkylating agents like Temozolomide, where trace impurities can lead to severe adverse effects. Patent CN102285991A introduces a transformative refinement strategy that addresses the longstanding stability and purity challenges associated with this critical active pharmaceutical ingredient (API). By leveraging a specific acid-assisted recrystallization technique within an acetone-water binary solvent system, this technology enables the production of Temozolomide with exceptional purity profiles exceeding 99.5 percent. The core innovation lies in the precise control of solution pH during the thermal dissolution phase, which mitigates the inherent thermal instability of the imidazotetrazine core. This technical breakthrough not only ensures compliance with stringent international pharmacopoeia standards, such as those set by the ICH and European Union, but also offers a scalable pathway for reliable API intermediate supplier networks aiming to optimize their manufacturing footprints.
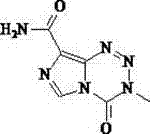
For R&D directors evaluating process feasibility, the structural integrity of the molecule during processing is paramount. As illustrated in the chemical structure above, the fused imidazotetrazine ring system is highly sensitive to hydrolytic degradation, especially under thermal stress in neutral or basic conditions. The patented method ingeniously circumvents this by introducing a controlled acidic environment (using acids like acetic, hydrochloric, or sulfuric acid) during the dissolution step at 50 to 60 degrees Celsius. This protonation effect stabilizes the tetrazine ring against nucleophilic attack by water molecules, thereby preserving the molecular scaffold while allowing soluble impurities to remain in the mother liquor upon cooling. The result is a product that consistently demonstrates single impurity levels below 0.1 percent, a critical specification for injectable formulations where patient safety is the ultimate priority.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Temozolomide has been plagued by significant technical hurdles that compromise both yield and product quality. Traditional protocols often rely on high-boiling polar aprotic solvents such as dimethyl sulfoxide (DMSO) or complex multi-step recrystallizations using ethanol and methylene chloride. These conventional approaches suffer from inherent inefficiencies; for instance, DMSO is notoriously difficult to remove completely due to its high boiling point and strong solvation properties, often leading to residual solvent issues that require extensive downstream processing. Furthermore, prior art methods frequently result in products with undesirable physical characteristics, such as a rose-pink coloration, indicating the presence of conjugated oxidative impurities that are difficult to purge. The reliance on column chromatography in some legacy processes further exacerbates the problem, introducing scalability bottlenecks and substantial solvent waste, making these methods economically unviable for large-scale commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel acid-assisted crystallization method described in the patent data offers a streamlined, robust alternative that fundamentally alters the purification landscape. By utilizing a binary solvent system of acetone and water, the process capitalizes on the differential solubility of Temozolomide and its impurities across a temperature gradient. The addition of a catalytic amount of acid acts as a stabilizing agent, preventing the thermal degradation that typically occurs during the hot dissolution phase in neutral media. This approach allows for a "hot filtration" step that effectively removes insoluble mechanical impurities and colored byproducts, often aided by activated carbon treatment without compromising the yield. The subsequent cooling crystallization yields pristine white crystals directly, eliminating the need for repetitive recrystallization cycles. This simplicity translates directly into operational efficiency, reducing the overall processing time and minimizing the exposure of the sensitive API to potentially degrading conditions.
Mechanistic Insights into Acid-Stabilized Recrystallization
To fully appreciate the technical merit of this purification route, one must understand the delicate balance of solubility and stability governed by the solvent system and pH. Temozolomide undergoes rapid non-enzymatic conversion to its active metabolite MTIC at physiological pH, but in the manufacturing context, premature degradation in the reactor is detrimental. In neutral or alkaline aqueous environments, the tetrazine ring is susceptible to hydrolysis, opening the ring and destroying the pharmacological activity. The introduction of acid (preferably acetic acid at 0.5 percent to 1.0 percent by weight of the solvent mixture) shifts the equilibrium, protonating basic sites on the molecule or impurities and suppressing the nucleophilicity of water. This kinetic stabilization allows the crude material to be dissolved at elevated temperatures (50 to 60 degrees Celsius) without significant decomposition, a feat that is impossible in pure water or neutral organic solvents.
Furthermore, the choice of acetone as the primary organic modifier is mechanistically sound due to its ability to disrupt the crystal lattice of impurities while maintaining Temozolomide in solution at higher temperatures. Upon cooling, the solubility of Temozolomide drops sharply, driving the formation of a pure crystal lattice that excludes structurally dissimilar impurities through the principle of selective crystallization. The acid also plays a role in keeping basic impurities in their salt forms, which are more soluble in the polar acetone-water mixture and thus remain in the mother liquor rather than co-crystallizing with the product. This dual mechanism of thermal stabilization and selective solubility ensures that the final product meets the rigorous requirement of having no single impurity greater than 0.1 percent, a standard that is often difficult to achieve with neutral solvent systems.
How to Synthesize Temozolomide Efficiently
The implementation of this purification protocol requires precise adherence to the specified parameters to maximize yield and purity. The process begins with the preparation of the solvent system, where acetone and water are mixed in a weight ratio ranging from 5 to 40 times the weight of the crude product, depending on the initial impurity load. An acid, such as acetic acid, is then introduced to the mixture before heating. The detailed standardized synthesis steps for this high-efficiency purification route are outlined in the guide below.
- Dissolve crude Temozolomide in a heated (50-60°C) mixture of acetone and water (5-40x weight ratio) containing 0.01-10% acid.
- Perform hot filtration immediately to remove insoluble particulates and optional activated carbon treatment for decolorization.
- Cool the filtrate to induce crystallization, followed by filtration and vacuum drying to obtain white crystals with >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the shift to this acid-assisted acetone/water methodology represents a significant strategic advantage in terms of cost structure and operational reliability. The elimination of high-boiling solvents like DMSO and the avoidance of column chromatography drastically simplify the downstream processing requirements. This simplification reduces the energy consumption associated with solvent recovery and distillation, as acetone is volatile and easily removed under vacuum. Consequently, the overall manufacturing cost is significantly reduced, not through arbitrary percentage claims, but through the tangible removal of expensive unit operations and the reduction of solvent inventory costs. For procurement managers, this translates to a more predictable cost base for cost reduction in pharmaceutical intermediates manufacturing, shielding the supply chain from the volatility of specialized solvent markets.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive, hard-to-remove solvents with commodity chemicals. Acetone and water are among the most widely available and cost-effective solvents in the chemical industry, ensuring stable pricing and easy sourcing. Additionally, the high yield (consistently above 80 percent) minimizes the loss of valuable API material, which is crucial given the high cost of goods for oncology drugs. By avoiding the need for multiple recrystallization steps or chromatographic purification, the facility saves on labor, equipment time, and waste disposal fees, creating a leaner and more cost-efficient production model.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on niche reagents or complex purification technologies that are prone to bottlenecks. This method utilizes standard reactor equipment (dissolution tanks, filters, dryers) found in almost any multipurpose pharmaceutical plant, reducing the risk of equipment-specific delays. The robustness of the process, evidenced by its tolerance to varying crude qualities (as seen in the patent examples), ensures that production schedules can be maintained even if the upstream synthesis of the crude material fluctuates slightly in quality. This resilience is vital for maintaining the steady flow of high-purity pharmaceutical intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: Scaling a purification process from the lab to the plant floor often reveals hidden complexities, but this method is inherently scalable due to its reliance on simple crystallization physics rather than complex separation mechanics. The use of acetone and water simplifies wastewater treatment compared to chlorinated solvents or DMSO, aligning with increasingly strict environmental regulations. The process generates less hazardous waste and reduces the carbon footprint associated with solvent incineration or recovery. This environmental compatibility facilitates faster regulatory approvals and ensures long-term operational sustainability for reducing lead time for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this process for adoption.
Q: Why is acid addition critical in the Temozolomide refining process?
A: Temozolomide exhibits poor thermal stability in neutral or alkaline aqueous solutions, leading to degradation during hot dissolution. The addition of acid (e.g., acetic acid) stabilizes the imidazotetrazine ring system, preventing hydrolysis and ensuring high recovery yields.
Q: How does this method compare to traditional DMSO-based purification?
A: Traditional methods using DMSO or ethanol often result in pink-colored products with difficult-to-remove impurities and lower yields. The acid-assisted acetone/water method produces white crystals with single impurity levels below 0.1% and simplifies solvent removal due to the volatility of acetone.
Q: What represents the primary cost advantage of this purification route?
A: The process eliminates the need for expensive column chromatography and reduces solvent consumption significantly. Furthermore, the use of commodity solvents like acetone and water lowers raw material costs and simplifies waste treatment compared to high-boiling polar aprotic solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Temozolomide Supplier
The technical potential of the acid-assisted crystallization method for Temozolomide is immense, offering a clear path to high-quality, cost-effective production that meets global regulatory standards. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this sophisticated chemistry to life. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the critical single impurity thresholds (<0.1 percent) demanded by modern oncology drug applications. We understand that consistency is key in the pharmaceutical supply chain, and our process validation protocols ensure that every batch meets the exacting standards required for API manufacturing.
We invite global partners to collaborate with us to leverage this advanced purification technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project timelines and quality expectations. Let us be your partner in delivering high-purity Temozolomide with the reliability and efficiency your business demands.
