Scalable Synthesis of 7-Iodopyrrolo[2,1-F][1,2,4]Triazine-4-Amine for Antiviral Drug Production
Scalable Synthesis of 7-Iodopyrrolo[2,1-F][1,2,4]Triazine-4-Amine for Antiviral Drug Production
The rapid global demand for effective antiviral therapeutics has placed immense pressure on the supply chains of critical pharmaceutical intermediates, particularly those required for nucleoside analogs like Remdesivir. Patent CN113880846A introduces a transformative preparation method for 7-iodopyrrolo[2,1-F][1,2,4]triazine-4-amine, a pivotal building block in the synthesis of these life-saving medications. This technical disclosure addresses long-standing manufacturing bottlenecks associated with traditional heterocyclic synthesis, specifically targeting the issues of high viscosity, excessive reagent costs, and complex purification workflows that have historically plagued the production of pyrrolo-triazine derivatives. By shifting from solid-base mediated reactions to a gas-liquid interaction system utilizing ammonia, the invention offers a streamlined pathway that enhances both operational safety and economic efficiency. For R&D directors and procurement strategists, understanding the nuances of this patented route is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at a commercial scale. The following analysis dissects the chemical innovations and supply chain advantages embedded within this technology, demonstrating its potential to redefine cost structures in antiviral drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-aminopyrrolo[2,1-f][1,2,4]triazine, the precursor to the target iodide, has been hindered by inefficient reaction dynamics and prohibitive raw material costs. Prior art, such as that disclosed in Patent CN110092787A, relies heavily on the use of pre-synthesized formamidine acetate and substantial quantities of solid inorganic bases like potassium phosphate. This approach creates a heterogeneous reaction mixture with extremely high viscosity, often requiring solvent volumes up to sixteen times the weight of the substrate to maintain any semblance of fluidity. Even with such excessive dilution, mass transfer remains poor, leading to incomplete conversions and molar yields that struggle to exceed 75%. Furthermore, the reliance on expensive, pre-packaged formamidine acetate significantly inflates the bill of materials, while the disposal of large amounts of solid salt waste generates substantial environmental liabilities. These factors combine to create a fragile supply chain where production scalability is limited by mechanical stirring capabilities and waste treatment capacities, making it difficult to meet the surging demands of the global pharmaceutical market without incurring massive operational expenses.
The Novel Approach
In stark contrast, the methodology outlined in CN113880846A revolutionizes the synthetic landscape by implementing an in-situ generation strategy that eliminates the need for costly external reagents and problematic solid bases. The novel process initiates by reacting triethyl orthoformate with glacial acetic acid under a pressurized ammonia atmosphere, effectively synthesizing formamidine acetate directly within the reaction vessel. This clever manipulation of reaction conditions not only reduces raw material costs but also generates ethanol as a byproduct, which subsequently serves as the solvent for the cyclization step, thereby removing the need for additional solvent charging. The resulting reaction system is homogeneous and significantly less viscous, allowing for superior heat transfer and mixing efficiency even at high concentrations. Following the formation of the triazine core, the process proceeds to a highly selective iodination step using iodine and N-iodosuccinimide (NIS) in DMF.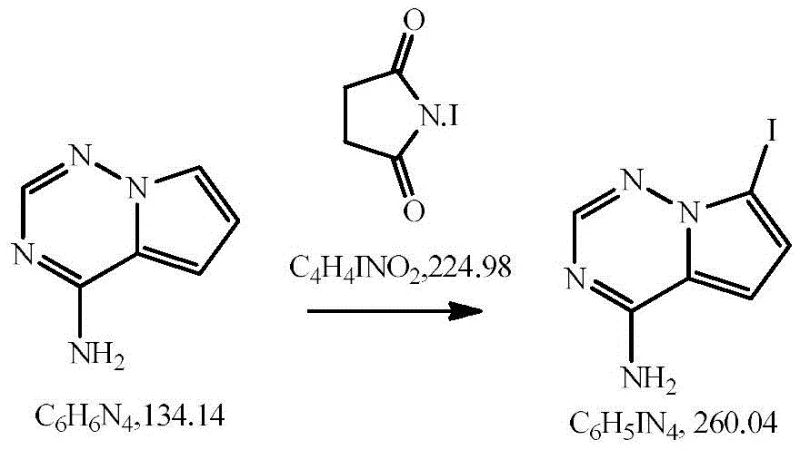
Mechanistic Insights into In-Situ Formamidine Acetate Generation and Cyclization
The core chemical innovation of this patent lies in the mechanistic shift from a solid-liquid interface to a gas-liquid reaction system, which fundamentally alters the kinetics and thermodynamics of the heterocycle formation. By introducing ammonia gas into a mixture of triethyl orthoformate and acetic acid, the reaction generates formamidine acetate and ethanol simultaneously. The ammonia acts as both a reactant and a base, neutralizing the acidic byproducts and maintaining the necessary alkaline environment for the subsequent nucleophilic attack by 1-aminopyrrole-2-carbonitrile hydrochloride. This in-situ generation ensures that the concentration of the reactive formamidine species is optimized continuously, preventing the decomposition or side reactions often seen when using stored, bulk quantities of the salt. Moreover, the ethanol produced during this initial stage serves as an ideal polar protic solvent for the cyclization, facilitating the dissolution of the pyrrole nitrile salt and promoting the ring-closure reaction without the need for external solvent addition. This self-sustaining solvent system not only simplifies the workup procedure but also drastically reduces the volume of waste liquor, aligning the process with green chemistry principles and reducing the environmental footprint of the manufacturing facility.
Following the cyclization, the crude 4-aminopyrrolo[2,1-f][1,2,4]triazine is isolated and purified to ensure the high quality required for downstream iodination. The patent specifies a recrystallization process using alcohols such as methanol or ethanol, which effectively removes unreacted starting materials and minor impurities, yielding a refined product with liquid phase purity exceeding 99.75%.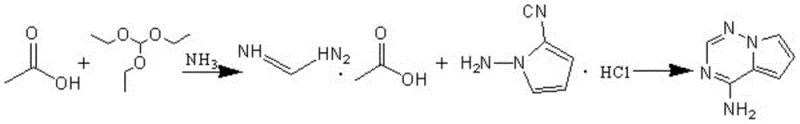
This high level of purity is critical because any residual impurities in the amine intermediate can propagate through the subsequent iodination step, complicating the purification of the final 7-iodo product and potentially affecting the efficacy of the final API. The robust nature of this purification step, combined with the high conversion rates of the cyclization, ensures a consistent and reliable supply of the intermediate, which is essential for maintaining the rigorous quality standards demanded by regulatory bodies for antiviral drug production. The ability to achieve such purity without complex chromatographic separations underscores the industrial viability of this route, offering a clear advantage over legacy methods that often require multiple purification cycles to reach acceptable quality thresholds.
How to Synthesize 7-Iodopyrrolo[2,1-F][1,2,4]Triazine-4-Amine Efficiently
The practical implementation of this synthesis route involves a carefully controlled sequence of temperature and pressure adjustments to maximize yield and safety. The process begins with the preparation of the triazine core, where precise control of ammonia pressure and reaction temperature is vital to ensure complete conversion of the orthoformate. Once the intermediate is secured, the iodination step requires strict temperature management, typically maintaining the reaction below -5°C initially to control the exotherm and ensure regioselectivity at the 7-position. Detailed standard operating procedures for scaling this chemistry from laboratory to pilot plant are critical for success, and the specific parameters for reagent addition rates and quenching protocols are outlined in the technical documentation below.
- Prepare 4-aminopyrrolo[2,1-f][1,2,4]triazine by reacting triethyl orthoformate and glacial acetic acid with ammonia gas, followed by cyclization with 1-aminopyrrole-2-carbonitrile hydrochloride.
- Purify the crude triazine intermediate via crystallization using methanol or ethanol to achieve high liquid phase purity exceeding 99.7%.
- Perform regioselective iodination of the purified amine using iodine and N-iodosuccinimide (NIS) in DMF at controlled low temperatures to yield the final 7-iodo product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic benefits that extend far beyond simple chemical yield improvements. The elimination of expensive, pre-formed formamidine acetate and solid inorganic bases translates directly into a significantly reduced cost of goods sold (COGS), as the primary nitrogen source is commodity-grade ammonia gas. This shift not only lowers raw material expenditure but also simplifies inventory management by reducing the number of specialized SKUs required for production. Furthermore, the drastic reduction in reaction viscosity means that existing reactor infrastructure can be utilized more efficiently, potentially increasing batch sizes without the need for capital-intensive equipment upgrades. The simplified workup and reduced solvent usage also lead to substantial cost savings in waste treatment and solvent recovery, enhancing the overall sustainability profile of the manufacturing operation. These factors collectively contribute to a more resilient and cost-effective supply chain, capable of withstanding market fluctuations and meeting the high-volume demands of the global pharmaceutical industry.
- Cost Reduction in Manufacturing: The substitution of costly solid reagents with gaseous ammonia and the in-situ generation of solvents eliminate significant line items from the production budget. By avoiding the purchase of formamidine acetate and large quantities of solid bases, manufacturers can achieve a leaner cost structure that improves margin potential in a competitive market. Additionally, the reduced need for external solvents lowers procurement costs and minimizes the energy expenditure associated with solvent distillation and recovery, further driving down operational expenses.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like ammonia, acetic acid, and triethyl orthoformate mitigates the risk of supply disruptions often associated with specialized fine chemical intermediates. These raw materials are widely available from multiple global suppliers, ensuring continuity of supply even during periods of market volatility. The robustness of the process also means that production schedules are less likely to be impacted by equipment failures related to high-viscosity mixing, leading to more predictable lead times and improved on-time delivery performance for customers.
- Scalability and Environmental Compliance: The low-viscosity nature of the reaction mixture facilitates seamless scale-up from pilot to commercial production, as heat and mass transfer limitations are minimized. This scalability is crucial for meeting the rapidly growing demand for antiviral therapeutics without compromising on quality or safety. Moreover, the reduction in solid waste and solvent discharge aligns with increasingly stringent environmental regulations, reducing the regulatory burden and potential liability associated with hazardous waste disposal, thereby future-proofing the manufacturing asset against evolving compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses highlight the specific advantages in terms of purity, yield, and operational simplicity that distinguish this method from conventional approaches.
Q: How does the new method reduce reaction system viscosity compared to traditional routes?
A: Traditional methods rely on solid bases like potassium phosphate and pre-formed formamidine acetate, creating a thick slurry that hinders stirring. The patented process utilizes ammonia gas to generate the necessary alkaline conditions and formamidine acetate in situ, resulting in a homogeneous liquid phase that significantly improves mixing efficiency and heat transfer.
Q: What purity levels can be achieved for the 7-iodopyrrolo triazine intermediate?
A: The optimized synthetic route ensures exceptional quality control, with the final 7-iodopyrrolo[2,1-F][1,2,4]triazine-4-amine achieving a liquid phase purity of over 99.2% without requiring additional purification steps, meeting the stringent specifications for downstream antiviral API synthesis.
Q: Does this process eliminate the need for external solvents during the cyclization step?
A: Yes, the process is designed to be self-sufficient regarding solvation. The ethanol generated as a byproduct during the initial formation of formamidine acetate serves as the reaction medium for the subsequent cyclization, thereby reducing raw material consumption and minimizing waste liquid discharge.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Iodopyrrolo[2,1-F][1,2,4]Triazine-4-Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of next-generation antiviral therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 7-iodopyrrolo[2,1-F][1,2,4]triazine-4-amine meets the exacting standards required for API synthesis. Our state-of-the-art facilities are equipped to handle the specific gas-liquid reaction conditions described in the patent, allowing us to leverage the cost and efficiency benefits of this innovative route for our clients.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your supply chain goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of sourcing this intermediate from our optimized production lines. We encourage potential partners to contact us for specific COA data and route feasibility assessments, ensuring that our solutions are perfectly aligned with your project timelines and quality expectations. Let us be your partner in advancing the availability of life-saving medications through superior chemical manufacturing.
