Advanced Semi-Synthetic Route for High-Purity Taxane Intermediates and Docetaxel Production
Advanced Semi-Synthetic Route for High-Purity Taxane Intermediates and Docetaxel Production
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for the production of complex antineoplastic agents, particularly taxanes like docetaxel. Patent CN101048395A discloses a groundbreaking semi-synthetic method for preparing 10-deacetyl-N-debenzoyl-paclitaxel (I), a critical synthon used in the final steps of docetaxel manufacturing. This technology represents a significant leap forward in process chemistry by replacing hazardous reagents such as zinc and hydrofluoric acid with a milder, more selective protocol involving stable oxazolidinic acids and trichloroacetyl protecting groups. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this route is essential for securing a stable supply chain of high-purity active pharmaceutical ingredients.
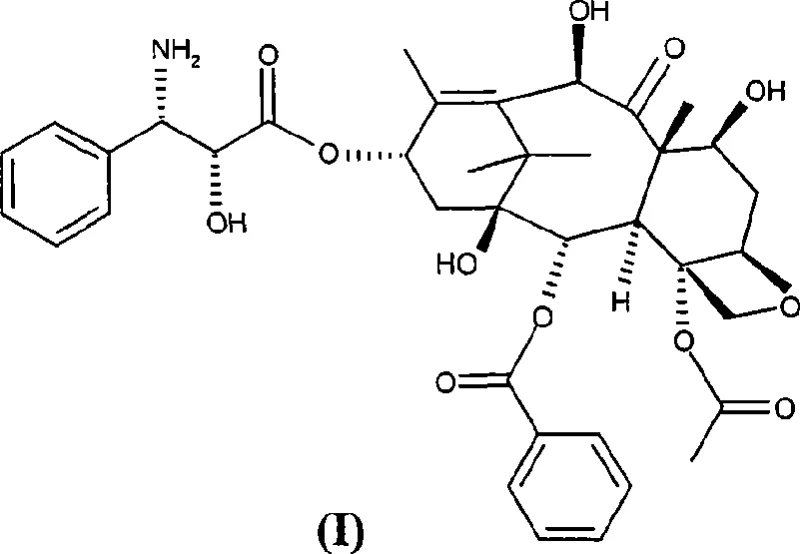
The core innovation lies in the strategic use of 2-(2,4-dimethoxyphenyl)-3-(2-nitrophenylsulfanyl)-4(S)-phenyl-5(R)-oxazolidinecarboxylic acid (V) coupled with a specifically protected baccatin III derivative. This approach not only streamlines the synthesis but also ensures that the final product achieves a purity exceeding 98% without the need for cumbersome chromatographic purification, a factor that drastically impacts cost reduction in API manufacturing. By leveraging this patented methodology, manufacturers can mitigate the risks associated with heavy metal contamination and aggressive acid handling, thereby aligning with modern green chemistry principles while maintaining rigorous quality standards required for oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 10-deacetyl-N-debenzoyl-paclitaxel relied on processes described in earlier literature, such as WO 94/07877, which utilized oxazolidines coupled with baccatin derivatives protected by silyl groups or other labile esters. A major drawback of these conventional methods was the necessity for harsh deprotection conditions to liberate the crucial amino and hydroxyl functionalities. Specifically, the removal of silyl protecting groups often required the use of hydrofluoric acid, a highly corrosive and dangerous reagent that poses severe safety risks and creates significant waste disposal challenges in a commercial plant setting. Furthermore, alternative routes employing zinc and acid for reductive cleavage introduced the risk of heavy metal residues, necessitating expensive and time-consuming purification steps to meet stringent regulatory limits for parenteral drugs. These legacy processes often suffered from lower overall yields due to side reactions and the instability of intermediates under the aggressive conditions required for deprotection, leading to inconsistent batch quality and supply chain volatility.
The Novel Approach
In stark contrast, the novel approach detailed in CN101048395A introduces a sophisticated protection-deprotection strategy that circumvents these historical bottlenecks by utilizing trichloroacetyl groups at the 7 and 10 positions of the baccatin core. This method employs a remarkably stable oxazolidinic acid (V) which facilitates a clean esterification reaction under mild conditions, typically using dicyclohexylcarbodiimide (DCC) and a catalytic amount of DMAP in dichloromethane at ambient temperature. The true brilliance of this route is revealed in the sequential deprotection steps, where the trichloroacetyl groups are selectively hydrolyzed using weak bases like ammonium hydroxide before the final acid treatment releases the amino and hydroxyl groups. This sequence prevents the undesirable migration of acyl groups to the newly freed amino function, a common side reaction that plagues other synthetic routes and generates difficult-to-remove impurities. Consequently, this new methodology offers a streamlined path to high-purity intermediates, significantly simplifying the downstream processing and enhancing the overall economic viability of docetaxel production.
Mechanistic Insights into Selective Esterification and Deprotection
The mechanistic elegance of this synthesis begins with the preparation of the key starting material, 10-deacetyl-di-7,10-trichloroacetylbaccatin III (VI), which is obtained by treating natural 10-deacetylbaccatin III with trichloroacetyl chloride. The electron-withdrawing nature of the trichloroacetyl groups renders the ester linkages at positions 7 and 10 sufficiently stable to withstand the subsequent coupling conditions, yet labile enough to be removed selectively later. When compound (VI) reacts with the oxazolidinic acid (V), the carboxylic acid moiety of (V) attacks the C-13 hydroxyl of the baccatin core, facilitated by the carbodiimide activator, to form the ester intermediate (VII). The stability of acid (V) is paramount here, as it prevents premature decomposition or racemization during the activation phase, ensuring that the stereochemical integrity of the taxane skeleton is preserved throughout the transformation.
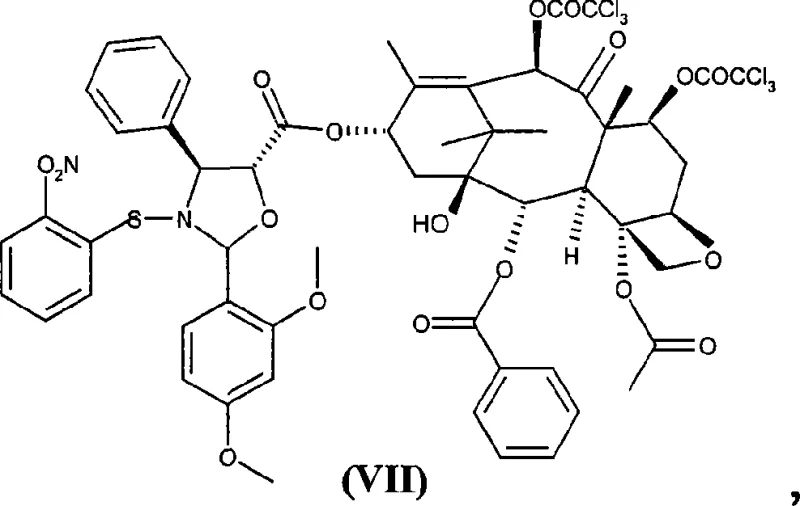
Following the formation of ester (VII), the process requires the careful removal of the 7 and 10 protecting groups to yield intermediate (VIII). It has been empirically observed that attempting to free the amino and hydroxyl groups from the oxazolidine ring prior to removing the trichloroacetyl groups leads to substantial acyl migration, where the trichloroacetyl moiety transfers from the baccatin oxygen to the newly exposed amine nitrogen. To avoid this, the protocol mandates a specific order of operations: first, treatment with ammonium hydroxide in tetrahydrofuran selectively cleaves the trichloroacetate esters without disturbing the oxazolidine ring, yielding compound (VIII). This intermediate is then subjected to mild acid hydrolysis, typically with aqueous hydrochloric acid in methanol, which cleanly opens the oxazolidine ring to release the free amino and hydroxyl functions, finally affording the target synthon (I). This precise control over reaction orthogonality is what enables the high purity levels reported in the patent.
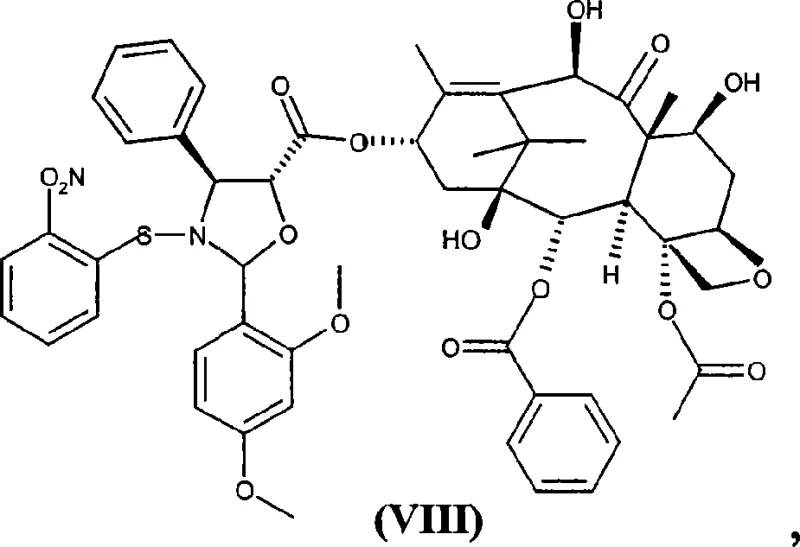
Impurity control is further enhanced by the physical properties of the intermediates. The patent specifies that the starting baccatin derivative (VI) can be purified via silica gel chromatography or crystallization to ensure that mono-trichloroacetyl derivatives are kept below 0.1%, a critical specification for preventing downstream impurity carryover. Similarly, the final synthon (I) can be isolated by precipitation from aliphatic hydrocarbons like n-hexane after extraction, bypassing the need for preparative HPLC or column chromatography on a large scale. This reliance on crystallization and precipitation rather than chromatography is a key indicator of a mature, scalable process, as it allows for the efficient processing of multi-kilogram batches with consistent quality, directly addressing the needs of commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 10-Deacetyl-N-Debenzoyl-Paclitaxel Efficiently
The synthesis of this critical taxane intermediate involves a logical sequence of protection, coupling, and deprotection steps designed to maximize yield and minimize hazard. The process begins with the rigorous purification of the baccatin starting material to eliminate regioisomeric impurities, followed by a mild esterification with the specialized oxazolidinic acid. The subsequent hydrolysis steps must be carefully monitored to ensure complete removal of protecting groups without degrading the sensitive taxane core. For detailed operational parameters, including solvent ratios, temperature profiles, and workup procedures, operators should refer to the standardized synthesis guidelines provided below.
- Perform esterification between 10-deacetyl-di-7,10-trichloroacetylbaccatin III and 2-(2,4-dimethoxyphenyl)-3-(2-nitrophenylsulfanyl)-4(S)-phenyl-5(R)-oxazolidinecarboxylic acid using DCC and DMAP.
- Hydrolyze the trichloroacetyl protecting groups at the 7 and 10 positions using ammonium hydroxide in tetrahydrofuran to prevent acyl migration.
- Treat the resulting intermediate with aqueous hydrochloric acid in methanol to liberate the amino and hydroxyl functional groups, followed by crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this semi-synthetic method offers tangible benefits that extend beyond mere technical feasibility, directly impacting the bottom line through operational efficiency and risk mitigation. By eliminating the need for hazardous reagents like hydrofluoric acid and zinc dust, the process significantly reduces the costs associated with specialized waste treatment and safety compliance, which are often hidden but substantial expenses in fine chemical manufacturing. Furthermore, the ability to isolate high-purity products through crystallization rather than chromatography translates to faster batch cycles and lower solvent consumption, effectively driving down the cost of goods sold (COGS) for the final API. This efficiency makes the supply of 10-deacetyl-N-debenzoyl-paclitaxel more resilient to market fluctuations and raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as hydrofluoric acid and zinc removes the need for specialized corrosion-resistant equipment and complex heavy metal scavenging processes. Additionally, the high selectivity of the trichloroacetyl protection strategy minimizes the formation of side products, leading to higher overall yields and reduced raw material waste. This streamlined workflow allows for substantial cost savings in both capital expenditure for plant infrastructure and operational expenditure for waste disposal and purification media.
- Enhanced Supply Chain Reliability: The starting materials for this route, including 10-deacetylbaccatin III and the oxazolidinic acid precursors, are readily available from established suppliers, reducing the risk of bottlenecks associated with exotic or custom-synthesized reagents. The robustness of the reaction conditions, which tolerate ambient temperatures and common solvents like dichloromethane and methanol, ensures that production can be maintained consistently across different manufacturing sites without requiring highly specialized technical expertise, thereby securing a continuous supply of critical oncology intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations such as filtration, crystallization, and liquid-liquid extraction that are easily transferred from pilot plant to commercial production scales ranging from 100 kgs to 100 MT annual capacity. By avoiding the generation of heavy metal waste and minimizing the use of aggressive acids, the process aligns with increasingly strict environmental regulations, reducing the regulatory burden and facilitating smoother audits and inspections by global health authorities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology resolves traditional pain points in taxane chemistry. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: Why is the trichloroacetyl protection strategy superior to silyl protection in this synthesis?
A: The trichloroacetyl groups provide exceptional stability during the esterification step and can be selectively removed under mild basic conditions (ammonium hydroxide) without affecting the sensitive oxazolidine ring or the baccatin core, avoiding the need for hazardous hydrofluoric acid.
Q: How does this process achieve high purity without extensive chromatography?
A: The process utilizes highly selective crystallization steps for both the intermediate 10-deacetyl-di-7,10-trichloroacetylbaccatin III and the final 10-deacetyl-N-debenzoyl-paclitaxel, allowing impurities like mono-trichloroacetyl derivatives to be reduced to below 0.1%.
Q: What are the safety advantages of this method compared to conventional zinc reduction?
A: This novel route eliminates the use of zinc dust and strong mineral acids for reduction, as well as hydrofluoric acid for desilylation, significantly reducing hazardous waste generation and improving operational safety for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-Deacetyl-N-Debenzoyl-Paclitaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against cancer, and we are committed to delivering 10-deacetyl-N-debenzoyl-paclitaxel with uncompromising quality and reliability. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of material that meets stringent purity specifications. Our state-of-the-art rigorous QC labs employ advanced analytical techniques to verify that every batch conforms to the highest standards, with impurity profiles tightly controlled to support the registration of finished drug products in major markets worldwide.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced semi-synthetic technology for their docetaxel programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis to understand how switching to this optimized route can improve your project economics. We encourage you to contact us today to obtain specific COA data for our current inventory and to discuss route feasibility assessments tailored to your specific volume requirements and timeline constraints.
