Breakthrough Chemical Synthesis of Anticancer GM-95 for Commercial Scale-Up
The pharmaceutical industry has long sought a reliable chemical pathway for the production of Substance GM-95, a potent macrocyclic compound exhibiting significant anticancer activity. Historically, the supply of this critical molecule was entirely dependent on isolation from microbial cultures, as described in international publication WO00/24747, which introduced inherent limitations regarding yield consistency and scalability. Patent CN1479742A represents a paradigm shift by disclosing the first viable total chemical synthesis method for GM-95, effectively transitioning the manufacturing paradigm from unpredictable biological fermentation to controlled organic synthesis. This breakthrough enables the production of high-purity API intermediates with defined stereochemistry and impurity profiles, addressing the urgent needs of oncology drug developers for stable raw material sources. By establishing a robust synthetic route involving sequential macrocyclization and heterocyclic ring formation, this technology lays the foundation for a secure global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN1479742A, the sole source of Substance GM-95 was extraction from fermentation broths, a method fraught with logistical and technical inefficiencies that hindered commercial viability. Biological production is notoriously sensitive to environmental variables, leading to fluctuating titers and complex downstream processing requirements to separate the target macrocycle from a myriad of cellular metabolites. Furthermore, the reliance on specific bacterial strains introduces risks of genetic drift or contamination, which can abruptly halt production and compromise the continuity of supply for clinical trials. The purification of natural products often requires extensive chromatography and solvent usage, resulting in high operational costs and significant environmental waste generation. For procurement managers, this dependency translates into volatile pricing and long lead times, making it difficult to forecast budgets or guarantee material availability for large-scale pharmaceutical manufacturing.
The Novel Approach
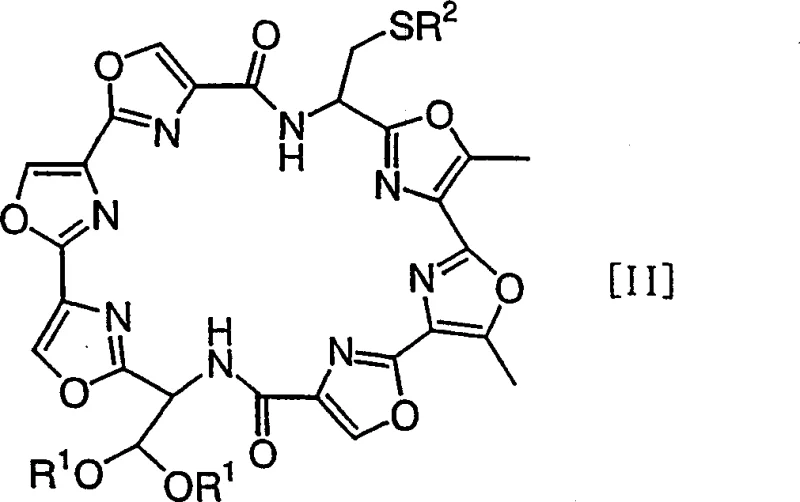
The novel chemical synthesis approach outlined in the patent overcomes these biological constraints by constructing the GM-95 skeleton through precise organic transformations starting from well-defined trioxazole building blocks. Instead of harvesting from nature, the process assembles the seven oxazole rings and one thiazoline ring through a convergent strategy involving amide coupling and intramolecular cyclization. This method allows for the strategic use of protecting groups, such as acetals and trityl groups, which enable orthogonal deprotection sequences to control the order of ring closure. By shifting to a synthetic route, manufacturers gain full control over reaction parameters including temperature, stoichiometry, and reaction time, leading to reproducible outcomes. This transition not only mitigates the supply risks associated with fermentation but also opens the door for structural analogs and derivatives, providing R&D teams with greater flexibility in drug development programs.
Mechanistic Insights into Macrocyclization and Heterocycle Formation
The core of this synthetic innovation lies in the sophisticated management of macrocyclization and the subsequent formation of the thiazoline moiety, which are critical for maintaining the bioactive conformation of the molecule. The process begins with the coupling of linear precursors, followed by a high-dilution macrocyclization step to form the initial oxazole-containing ring system, typically utilizing reagents like diphenylphosphoryl azide (DPPA) to drive amide bond formation while suppressing intermolecular polymerization. Following this, the acetal protecting groups are removed under acidic conditions to reveal formyl groups, which then undergo intramolecular condensation with adjacent amide nitrogens to close additional oxazole rings. The final and perhaps most delicate step involves the deprotection of the thiol group and its cyclization with an amide to form the thiazoline ring, often mediated by strong Lewis acids like titanium tetrachloride. Understanding these mechanistic nuances is essential for R&D directors aiming to optimize reaction yields and minimize the formation of diastereomers or regioisomers during scale-up.
Impurity control is rigorously managed through the selection of specific protecting groups that offer differential stability under varying reaction conditions. For instance, the use of trityl groups for thiol protection allows for selective removal in the presence of other functional groups, preventing premature cyclization or side reactions that could generate difficult-to-remove impurities. The patent specifies conditions such as the use of triphenylphosphine and iodine for oxazole formation, which are chosen to minimize racemization at chiral centers adjacent to the reaction sites. By controlling the stereochemistry of the starting materials, specifically the asymmetric carbon atoms in the thiol derivatives, the synthesis ensures that the final GM-95 product retains the correct optical configuration required for anticancer activity. This level of mechanistic control ensures that the impurity profile is predictable and manageable, meeting the stringent purity specifications required for regulatory submission.
How to Synthesize Substance GM-95 Efficiently
The synthesis of Substance GM-95 requires a meticulous sequence of coupling and cyclization reactions that demand precise control over reaction conditions to achieve high purity and yield. The process generally initiates with the preparation of linear amide precursors through dehydration condensation, followed by macrocyclization under high dilution to favor intramolecular ring closure over oligomerization. Subsequent steps involve the sequential deprotection of acetal and thiol groups to trigger the formation of the remaining oxazole and thiazoline rings respectively. Detailed operational parameters, including specific solvent choices like dimethylformamide or dichloromethane and exact stoichiometric ratios of coupling agents, are critical for success. For a comprehensive guide on executing these steps with laboratory-grade precision, please refer to the standardized protocol below.
- Couple trioxazole derivatives with thiol derivatives using dehydration condensation agents like EDC/HOBt to form the linear amide precursor.
- Perform intramolecular cyclization of the linear precursor under high dilution conditions using DPPA to form the macrocyclic oxazole ring system.
- Execute final deprotection and thiazoline ring closure using Titanium Tetrachloride (TiCl4) to yield the final GM-95 structure.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from fermentation-based isolation to total chemical synthesis offers profound commercial advantages that directly address the pain points of procurement and supply chain leadership in the pharmaceutical sector. By decoupling production from biological systems, manufacturers can eliminate the volatility associated with fermentation yields and the extensive downstream processing required to purify natural products. This shift enables a more predictable manufacturing schedule, allowing for better inventory management and reduced safety stock requirements. Furthermore, the use of standard organic synthesis equipment means that production can be easily scaled across multiple facilities without the need for specialized bioreactors, enhancing supply chain resilience against regional disruptions. These factors collectively contribute to a more stable and cost-effective supply of this critical oncology intermediate.
- Cost Reduction in Manufacturing: The synthetic route eliminates the high capital and operational expenditures associated with maintaining fermentation facilities and performing complex extractions from biological media. By utilizing commodity chemicals and standard reagents, the process significantly reduces the cost of goods sold (COGS) compared to the labor-intensive isolation of natural products. Additionally, the ability to recycle solvents and optimize reaction stoichiometry in a chemical plant setting leads to substantial waste reduction and lower disposal costs. This economic efficiency allows for more competitive pricing models for the final API, benefiting the entire value chain from intermediate supplier to drug manufacturer.
- Enhanced Supply Chain Reliability: Chemical synthesis provides a robust alternative to the fragile supply chains of natural product extraction, which are susceptible to biological contamination and strain instability. With a fully synthetic route, production timelines become deterministic rather than probabilistic, ensuring that delivery commitments can be met with high confidence. This reliability is crucial for pharmaceutical companies managing tight clinical trial schedules or commercial launch plans where material shortages can have devastating financial impacts. The ability to source raw materials from established chemical supply chains further mitigates the risk of single-source dependency.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing reaction conditions that are compatible with standard multi-purpose chemical reactors ranging from pilot to commercial scale. Unlike fermentation, which generates large volumes of biological waste, the synthetic process produces waste streams that are easier to characterize and treat, facilitating compliance with increasingly stringent environmental regulations. The modular nature of the synthesis allows for capacity expansion through the addition of parallel reactor trains rather than the construction of massive new fermentation suites, offering a flexible path for meeting growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of Substance GM-95, based on the detailed disclosures within patent CN1479742A. These insights are intended to clarify the feasibility of the synthetic route and its implications for long-term sourcing strategies. Understanding these aspects is vital for stakeholders evaluating the integration of this intermediate into their development pipelines.
Q: What is the primary advantage of the chemical synthesis of GM-95 over fermentation?
A: Chemical synthesis offers superior supply chain reliability by eliminating the variability associated with biological fermentation, such as strain degradation and seasonal crop fluctuations, ensuring consistent batch-to-batch quality.
Q: How does the new process improve impurity control?
A: The synthetic route utilizes specific protecting group strategies (acetal and trityl) that allow for orthogonal deprotection, minimizing side reactions and facilitating easier purification compared to complex natural product extraction.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process relies on standard organic synthesis unit operations like condensation and acid-catalyzed cyclization, which are readily scalable from kilogram to multi-ton production without specialized bioreactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GM-95 Supplier
The successful chemical synthesis of Substance GM-95 marks a significant milestone in the availability of high-quality oncology intermediates, and NINGBO INNO PHARMCHEM is uniquely positioned to capitalize on this technological advancement. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of GM-95 meets the exacting standards required for pharmaceutical applications. We understand the critical nature of anticancer drug development and are committed to providing a seamless supply experience.
We invite you to engage with our technical procurement team to discuss how our synthetic capabilities can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to our chemically synthesized GM-95. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your specific formulation requirements. Let us collaborate to accelerate the delivery of life-saving therapies to patients worldwide.
