Scalable Enantioselective Synthesis of (S)-4-Amino-hepta-5,6-dienoic Acid for Pharmaceutical Applications
Introduction to Advanced GABA-T Inhibitor Synthesis
The development of potent neurological therapeutics often hinges on the availability of high-purity chiral intermediates, specifically those capable of acting as irreversible enzyme inhibitors. Patent CN1093853C discloses a groundbreaking enantioselective process for the preparation of (S)-4-amino-hepta-5,6-dienoic acid and its pharmaceutically acceptable salts, which serve as critical precursors for GABA-T (gamma-aminobutyric acid transaminase) inhibitors. This technology represents a significant leap forward in process chemistry, moving away from inefficient racemic syntheses toward a streamlined, stereo-controlled pathway. For pharmaceutical manufacturers, mastering this synthesis is not merely a technical exercise but a strategic imperative to secure supply chains for next-generation neurological treatments. The patent outlines a robust methodology that begins with the resolution of a specific amine and proceeds through a series of carefully orchestrated transformations involving succinimide and lactam intermediates. By leveraging this intellectual property, chemical producers can achieve superior control over the impurity profile and optical purity of the final active pharmaceutical ingredient (API), addressing the stringent regulatory requirements of global health authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing allenic amino acids often suffer from significant drawbacks regarding stereochemical control and overall yield. Conventional routes frequently rely on the resolution of racemic mixtures at a late stage, which theoretically caps the maximum yield at 50% unless dynamic kinetic resolution is employed, a process that often requires complex and costly catalysts. Furthermore, earlier methods might involve harsh reaction conditions that compromise the integrity of the sensitive allene functionality, leading to polymerization or isomerization side products that are difficult to remove. The reliance on transition metal catalysis in some asymmetric syntheses introduces the risk of heavy metal residues, necessitating expensive purification steps to meet ICH Q3D guidelines for elemental impurities. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for procurement teams aiming to launch cost-effective generic or novel therapies. Additionally, the lack of robust intermediate isolation points in older methodologies makes process monitoring and quality control challenging during scale-up.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a highly efficient sequence starting from a resolved amine of formula (1), ensuring that chirality is established early in the synthesis. This method transforms the resolved amine into a succinimide derivative, which is then selectively reduced to a 5'-hydroxybutyrolactam derivative, as illustrated by the structure in Formula (3). 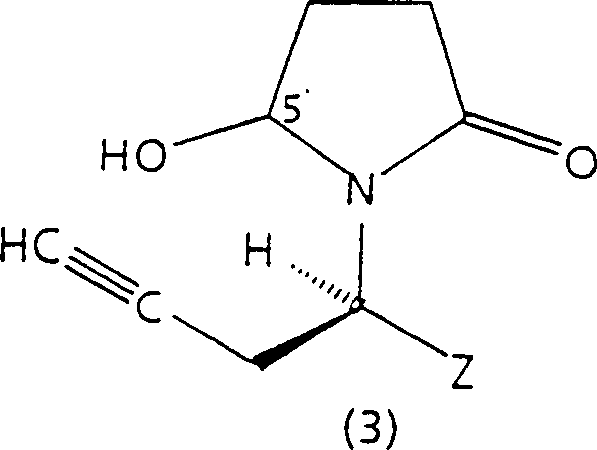 This specific intermediate is pivotal because it allows for a controlled acid-catalyzed rearrangement that constructs the crucial allene moiety with high stereospecificity. By avoiding late-stage resolution, the process maximizes atom economy and reduces waste generation. The use of common reagents such as succinic anhydride and borohydrides ensures that the supply chain remains resilient and cost-effective. This strategy effectively decouples the complexity of chiral induction from the construction of the carbon skeleton, allowing each step to be optimized independently for yield and purity. Consequently, this novel route offers a reliable pharmaceutical intermediate supplier pathway that significantly lowers the barrier to entry for manufacturing high-value neurological agents.
This specific intermediate is pivotal because it allows for a controlled acid-catalyzed rearrangement that constructs the crucial allene moiety with high stereospecificity. By avoiding late-stage resolution, the process maximizes atom economy and reduces waste generation. The use of common reagents such as succinic anhydride and borohydrides ensures that the supply chain remains resilient and cost-effective. This strategy effectively decouples the complexity of chiral induction from the construction of the carbon skeleton, allowing each step to be optimized independently for yield and purity. Consequently, this novel route offers a reliable pharmaceutical intermediate supplier pathway that significantly lowers the barrier to entry for manufacturing high-value neurological agents.
Mechanistic Insights into Acid-Catalyzed Lactam Rearrangement
The core chemical innovation of this process lies in the conversion of the 5'-hydroxybutyrolactam derivative into the (S)-5-propadienyl butyrolactam. This transformation is driven by a sophisticated mechanism involving dehydration and subsequent rearrangement. As depicted in the reaction scheme, the process is theorized to proceed through specific cationic intermediates. 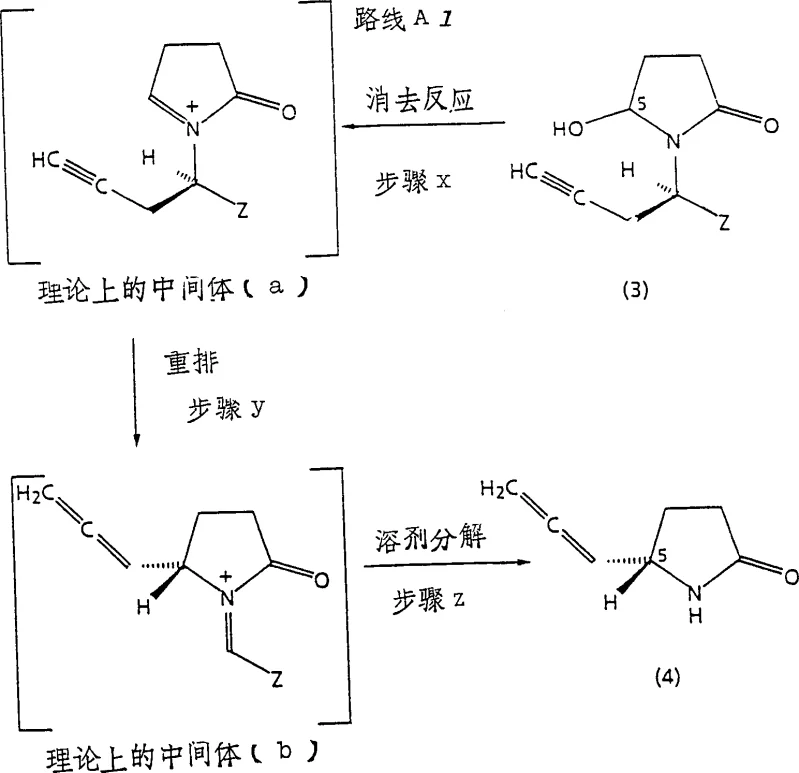 The reaction initiates with the protonation of the hydroxyl group by a suitable dehydroxylating acid, such as trifluoroacetic acid, facilitating the loss of water to generate a transient iminium ion species. This is followed by an elimination reaction that forms an exocyclic double bond, leading to the theoretical intermediate (a). Subsequent rearrangement steps, potentially involving allylic shifts, drive the formation of the allene system found in intermediate (b). The precise control of solvent polarity and acid strength is critical here; solvents like dichloromethane or tetrahydrofuran are employed to stabilize these charged intermediates without promoting unwanted side reactions. Understanding this mechanistic pathway is essential for R&D directors aiming to troubleshoot potential impurities, such as regioisomers or over-reduced species, ensuring that the final product meets the rigorous specifications required for clinical applications.
The reaction initiates with the protonation of the hydroxyl group by a suitable dehydroxylating acid, such as trifluoroacetic acid, facilitating the loss of water to generate a transient iminium ion species. This is followed by an elimination reaction that forms an exocyclic double bond, leading to the theoretical intermediate (a). Subsequent rearrangement steps, potentially involving allylic shifts, drive the formation of the allene system found in intermediate (b). The precise control of solvent polarity and acid strength is critical here; solvents like dichloromethane or tetrahydrofuran are employed to stabilize these charged intermediates without promoting unwanted side reactions. Understanding this mechanistic pathway is essential for R&D directors aiming to troubleshoot potential impurities, such as regioisomers or over-reduced species, ensuring that the final product meets the rigorous specifications required for clinical applications.
Furthermore, the stereochemical outcome of this rearrangement is governed by the configuration of the starting material and the conformational constraints of the lactam ring. The patent data suggests that the reaction preserves the optical integrity established during the initial amine resolution, converting the chiral information at the nitrogen-substituted carbon into the axial chirality or specific geometry of the allene side chain. Impurity control is managed by optimizing the stoichiometry of the solvolysis reagent, typically water or lower alkanols, which quenches the reactive intermediates to form the stable lactam product. This level of mechanistic understanding allows process chemists to define critical process parameters (CPPs) that guarantee consistent quality. By minimizing the formation of diastereomeric impurities at the 5' position, the downstream purification burden is drastically reduced, enhancing the overall efficiency of the manufacturing campaign.
How to Synthesize (S)-4-Amino-hepta-5,6-dienoic Acid Efficiently
Executing this synthesis requires strict adherence to the reaction conditions outlined in the patent to ensure high enantiomeric excess and yield. The process begins with the resolution of the racemic amine using chiral acids, followed by acylation with succinic anhydride to form the imide. The subsequent reduction step using lithium triethylborohydride must be conducted at low temperatures, typically around -78°C, to control the diastereoselectivity of the hydroxyl group formation. The critical rearrangement step involves treating the hydroxy-lactam with trifluoroacetic acid, followed by solvolysis. Detailed standardized operating procedures for each of these stages, including specific workup and purification protocols like ion exchange chromatography, are essential for reproducibility. For a comprehensive breakdown of the exact reagent quantities, temperature profiles, and isolation techniques, please refer to the technical guide below.
- Resolve the racemic amine using a chiral acid forming agent to obtain the optically pure amine of formula (1).
- React the resolved amine with succinic anhydride to form the succinimide derivative, followed by stereoselective reduction to the 5'-hydroxybutyrolactam.
- Treat the hydroxy-lactam with a dehydroxylating acid and solvolysis reagent to induce rearrangement into the (S)-5-propadienyl butyrolactam, followed by ring opening.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The elimination of precious metal catalysts, which are often subject to volatile market pricing and supply constraints, results in substantial cost savings and reduces the complexity of raw material sourcing. By relying on commodity chemicals like succinic anhydride and standard reducing agents, procurement managers can secure long-term contracts with multiple suppliers, mitigating the risk of single-source dependency. Furthermore, the robustness of the reaction conditions means that the process can be transferred between manufacturing sites with minimal re-validation, enhancing supply continuity. The ability to isolate stable intermediates, such as the succinimide and hydroxy-lactam derivatives, provides flexibility in production scheduling, allowing companies to stockpile key precursors and respond rapidly to fluctuations in market demand for the final API.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive chiral ligands or transition metal catalysts often associated with asymmetric synthesis. The reliance on classical resolution followed by high-yielding chemical transformations reduces the overall cost of goods sold (COGS). Additionally, the simplified purification steps, which avoid complex chromatographic separations typical of racemic mixtures, lower solvent consumption and waste disposal costs. This economic efficiency makes the production of GABA-T inhibitors more viable for both branded and generic drug markets, allowing for competitive pricing strategies without compromising margin.
- Enhanced Supply Chain Reliability: The starting materials and reagents utilized in this pathway are widely available from global chemical distributors, ensuring a stable and reliable supply chain. Unlike specialized catalysts that may have long lead times, commodities like succinic anhydride and trifluoroacetic acid can be sourced with short notice, reducing the risk of production delays. The modular nature of the synthesis, with distinct isolation points for intermediates, allows for decentralized manufacturing strategies where different steps can be performed at different locations if necessary, further strengthening supply chain security against geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: This methodology is inherently scalable, utilizing standard reactor types and operating conditions that are easily replicated from the laboratory to the pilot plant and finally to commercial scale. The avoidance of hazardous reagents and the use of recyclable solvents align with green chemistry principles, simplifying environmental compliance and permitting processes. The high atom economy of the rearrangement step minimizes waste generation, reducing the environmental footprint of the manufacturing process. This sustainability profile is increasingly important for meeting corporate social responsibility goals and satisfying the rigorous environmental audits conducted by major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on process feasibility and quality control. Understanding these aspects is crucial for technical teams evaluating the adoption of this route for their specific product portfolios. The answers reflect the practical realities of scaling this chemistry while maintaining the high standards required for pharmaceutical production.
Q: How is stereochemical purity maintained during the lactam rearrangement step?
A: The process utilizes a specific acid-catalyzed rearrangement of the 5'-hydroxybutyrolactam derivative. By controlling the reaction conditions with protic acids like trifluoroacetic acid and specific solvolysis reagents, the reaction proceeds through defined theoretical intermediates that preserve the chirality established in the initial resolution step, yielding the desired (S)-enantiomer with high fidelity.
Q: What are the advantages of this method over traditional catalytic asymmetric synthesis?
A: This method avoids the use of expensive and potentially toxic transition metal catalysts often required for asymmetric hydrogenation or coupling. Instead, it relies on classical chiral resolution followed by robust chemical transformations using readily available reagents like succinic anhydride and borohydrides, significantly simplifying the supply chain and reducing heavy metal contamination risks in the final API.
Q: Can this process be scaled for commercial production of GABA-T inhibitors?
A: Yes, the synthetic route described in patent CN1093853C utilizes standard unit operations such as crystallization, extraction, and conventional heating/reflux. The reagents are commodity chemicals, and the intermediates are stable enough for isolation, making the process highly amenable to scale-up from pilot kilogram quantities to multi-ton commercial manufacturing without requiring specialized high-pressure or cryogenic equipment beyond standard laboratory capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-Amino-hepta-5,6-dienoic Acid Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the enantiomeric excess of complex intermediates like (S)-4-amino-hepta-5,6-dienoic acid. We understand the critical nature of neurological APIs and apply our deep expertise in process optimization to deliver materials that consistently meet regulatory standards. Our commitment to quality and reliability makes us the preferred partner for global pharmaceutical companies seeking a secure source of high-value chiral building blocks.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this efficient manufacturing method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial viability. Let us collaborate to accelerate the development of life-saving therapies through superior chemical manufacturing.
