Advanced Copper-Catalyzed Deoxyfluorination for Scalable Benzoyl Fluoride Production
Introduction to Next-Generation Acyl Fluoride Synthesis
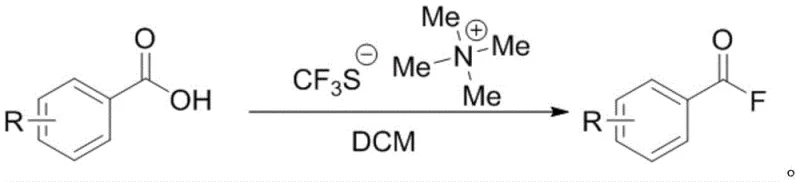
The synthesis of benzoyl fluoride compounds represents a critical capability in modern organic synthesis, particularly for the development of high-value pharmaceutical intermediates and agrochemical precursors. As detailed in the recent patent CN114773185B, a groundbreaking methodology has been established that utilizes sodium trifluoromethanesulfinate as a green fluorinating agent under copper catalysis. This innovation addresses the long-standing industry challenge of accessing acyl fluorides safely and economically, moving away from hazardous reagents towards a more sustainable chemical paradigm. For R&D directors and procurement specialists, this technology offers a robust pathway to generate versatile building blocks that can undergo subsequent amidation or esterification with exceptional efficiency. The strategic implementation of this copper-catalyzed deoxyfluorination process not only enhances the safety profile of chemical manufacturing but also aligns with global trends towards greener synthesis routes, making it a highly attractive option for reliable benzoyl fluoride supplier partnerships seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct preparation of benzoyl fluoride compounds has been plagued by significant operational and safety hurdles that hinder widespread industrial adoption. Traditional methods often rely on highly toxic and moisture-sensitive reagents such as DAST (Diethylaminosulfur trifluoride) or Deoxo-Fluor, which pose severe risks during handling and require stringent anhydrous conditions that drive up infrastructure costs. Furthermore, alternative approaches utilizing aminodifluorosulfinium tetrafluoroborate salts, while crystalline, suffer from inherent instability that limits their substrate scope and shelf-life, creating bottlenecks in continuous manufacturing processes. Other literature methods involving phosphine/fluoride systems often necessitate multi-step procedures or harsh conditions that degrade sensitive functional groups on the aromatic ring, thereby restricting the versatility of the synthesis. These conventional limitations result in higher waste generation, increased regulatory compliance burdens, and unpredictable production timelines, which are critical pain points for supply chain heads managing complex API intermediate portfolios.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach disclosed in patent CN114773185B leverages the in-situ generation of reactive fluorinating species from stable, inexpensive sodium trifluoromethanesulfinate. This method employs a synergistic catalytic system comprising triphenylphosphine as a reducing agent and a copper complex to activate the sulfinate salt, enabling a smooth deoxyfluorination of carboxylic acids under relatively mild thermal conditions. The process eliminates the need for storing dangerous fluorinating gases or unstable liquids, as the key reactive intermediate, thiocarbonyl fluoride, is generated transiently within the reaction mixture. This strategic shift allows for a broader substrate tolerance, accommodating various substituents such as halogens and alkoxy groups without compromising yield or purity. By simplifying the reagent profile to bench-stable solids and common solvents like dichloromethane, this novel approach drastically reduces the barrier to entry for cost reduction in pharmaceutical intermediates manufacturing, offering a streamlined route that is both economically and environmentally superior.
Mechanistic Insights into Copper-Catalyzed Deoxyfluorination
The mechanistic elegance of this transformation lies in the cooperative interaction between the copper catalyst and the phosphine reductant to unlock the fluorinating potential of the sulfinate salt. Initially, the copper catalyst facilitates the reduction of sodium trifluoromethanesulfinate to generate a trifluoromethylthio anion intermediate, which subsequently decomposes to release highly reactive thiocarbonyl fluoride and fluoride anions. This thiocarbonyl fluoride species acts as a potent electrophile, attacking the hydroxyl group of the benzoic acid substrate to form a mixed anhydride-like intermediate containing a good leaving group. The abundance of fluoride anions in the system then drives a nucleophilic substitution at the carbonyl carbon, displacing the leaving group and forging the strong carbon-fluorine bond characteristic of the acyl fluoride product. Understanding this catalytic cycle is crucial for R&D teams aiming to troubleshoot potential side reactions or optimize reaction kinetics, as the balance between the reduction rate and the decomposition of the sulfinate dictates the overall efficiency of the fluorination event.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of halogenated byproducts often seen with harsher fluorinating agents. The mild nature of the fluoride source and the specific activation via the copper center ensure that the reaction proceeds with high chemoselectivity, preserving other sensitive moieties on the aromatic ring that might otherwise be compromised. The use of triphenylphosphine oxide as a byproduct is also beneficial, as it is easily separable from the organic phase during the aqueous workup, contributing to the high purity of the final isolated material. For quality assurance professionals, this predictable impurity profile simplifies the validation process and ensures that the resulting high-purity OLED material or pharmaceutical intermediate meets stringent regulatory specifications without requiring extensive downstream purification steps that erode profit margins.
How to Synthesize Benzoyl Fluoride Efficiently
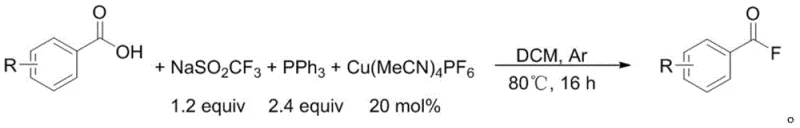
Executing this synthesis requires careful attention to the stoichiometry of the reagents and the maintenance of an inert atmosphere to prevent catalyst deactivation. The standard protocol involves charging a sealed reaction vessel with the sodium trifluoromethanesulfinate, triphenylphosphine, and the copper catalyst, followed by thorough degassing to remove oxygen which could oxidize the phosphine or the copper center. Once the benzoic acid substrate and anhydrous dichloromethane are added, the mixture is heated to promote the catalytic cycle, typically requiring a reaction time of 16 to 18 hours to reach full conversion. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to ensure reproducibility and safety during scale-up operations.
- Charge a sealed Schlenk tube with sodium trifluoromethanesulfinate, triphenylphosphine, and tetrakis(acetonitrile)copper(I) hexafluorophosphate under inert atmosphere.
- Add the benzoic acid substrate and anhydrous dichloromethane solvent, then heat the mixture to 80°C for 16 to 18 hours.
- Upon completion, cool to room temperature, extract with dichloromethane and water, dry the organic phase, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed methodology translates into tangible strategic benefits that extend beyond simple reaction yields. The primary advantage lies in the substantial cost savings achieved by replacing exotic, high-cost fluorinating reagents with commodity chemicals like sodium trifluoromethanesulfinate and triphenylphosphine, which are available in bulk quantities at stable prices. This shift in raw material sourcing mitigates the risk of supply disruptions caused by the limited manufacturing capacity of specialized fluorinating agents, thereby enhancing supply chain reliability and ensuring consistent production schedules for critical intermediates. Furthermore, the elimination of cryogenic conditions and the use of standard solvents simplify the engineering requirements for reactor setups, allowing for easier commercial scale-up of complex pharmaceutical intermediates without the need for specialized low-temperature infrastructure.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive, single-use fluorinating reagents with catalytic amounts of copper and stoichiometric amounts of inexpensive solids. By avoiding the procurement of hazardous reagents that require special shipping and storage protocols, manufacturers can significantly lower their operational expenditures related to safety compliance and waste disposal. The simplified workup procedure, which relies on standard extraction and filtration rather than complex distillations, further reduces energy consumption and labor costs, contributing to a leaner and more competitive manufacturing model that maximizes return on investment for large-volume production runs.
- Enhanced Supply Chain Reliability: Utilizing bench-stable solid reagents fundamentally improves the resilience of the supply chain against logistical shocks and storage degradation issues. Unlike liquid fluorinating agents that may decompose over time or require cold chain logistics, the solid reagents used in this method maintain their integrity under ambient conditions, allowing for flexible inventory management and reduced warehousing costs. This stability ensures that production campaigns can be initiated on demand without the lead time associated with sourcing fresh batches of unstable reagents, providing a critical buffer against market volatility and ensuring uninterrupted delivery of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than traditional methods, aligning with increasingly strict global regulations on chemical manufacturing emissions and waste. The generation of less hazardous waste streams and the avoidance of toxic byproducts simplify the permitting process for new production lines and reduce the liability associated with environmental compliance. From a scalability standpoint, the robustness of the copper catalytic system allows for seamless translation from gram-scale laboratory optimization to multi-kilogram pilot plant operations, facilitating rapid technology transfer and accelerating the time-to-market for new drug candidates relying on these acyl fluoride building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, providing clarity for stakeholders evaluating its integration into their existing workflows. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring that the information provided is accurate and actionable for decision-makers. Understanding these nuances is essential for assessing the feasibility of adopting this technology for specific project requirements and for engaging in informed discussions with technical service providers.
Q: What are the primary advantages of using sodium trifluoromethanesulfinate over traditional fluorinating reagents?
A: Unlike traditional reagents such as DAST or Deoxo-Fluor which are toxic and thermally unstable, sodium trifluoromethanesulfinate is a stable, bench-top solid that significantly reduces safety hazards and storage costs while maintaining high reactivity under copper catalysis.
Q: Does this copper-catalyzed method tolerate diverse functional groups on the aromatic ring?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully converting substrates with electron-withdrawing groups like chloro and bromo, as well as electron-donating groups like methoxy and tert-butyl, into their corresponding acyl fluorides with moderate to good yields.
Q: Is the purification process complex for large-scale manufacturing?
A: The workup is straightforward involving standard aqueous extraction and drying, followed by column chromatography for high purity, which avoids the need for complex distillation setups often required for volatile or sensitive acyl fluoride derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoyl Fluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed deoxyfluorination technology and are fully equipped to leverage it for your custom synthesis needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benzoyl fluoride or related intermediate meets the exacting standards required by the global pharmaceutical and agrochemical industries, providing you with a partner who prioritizes quality and consistency above all else.
We invite you to engage with our technical procurement team to discuss how this innovative method can be tailored to your specific molecular targets and volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your project, along with access to specific COA data and route feasibility assessments that demonstrate our commitment to transparency and technical excellence. Let us help you optimize your supply chain and accelerate your development timelines with our advanced manufacturing capabilities and deep expertise in fine chemical synthesis.
