Revolutionizing Beta-Lactam Synthesis: Efficient Production of Triazolylmethylpenicillane Intermediates
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex beta-lactam structures, which serve as the backbone for numerous life-saving antibiotics and enzyme inhibitors. Patent CN1022835C introduces a groundbreaking methodology for the preparation of 2α-methyl-2β-(1,2,3-triazol-1-yl)methylpenicillane-3α-carboxylic acid derivatives. This technology represents a significant leap forward in medicinal chemistry, specifically addressing the challenges associated with introducing triazole moieties into the penicillane nucleus. By leveraging a direct nucleophilic substitution on penicillanic acid sulfoxide derivatives, this process bypasses the cumbersome and hazardous steps characteristic of legacy synthetic routes. For R&D directors and procurement specialists alike, understanding this innovation is crucial for optimizing the supply chain of high-purity pharmaceutical intermediates. The ability to access these critical scaffolds through a simplified, one-step reaction not only enhances process safety but also offers substantial potential for cost reduction in API manufacturing, making it a highly attractive candidate for commercial adoption by reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazole-substituted penicillanes has been plagued by inefficiency and significant safety hazards. As illustrated in the reaction scheme below, traditional methodologies typically involve a convoluted multi-step sequence designated as Steps A through E. This archaic route necessitates the conversion of penicillanic acid sulfoxides into azetidinone disulfides, followed by transformation into unstable 2β-halomethylpenicillin derivatives. The subsequent steps involve the use of highly explosive azide compounds and acetylene derivatives to install the triazole ring. These reagents pose severe risks in a manufacturing environment, requiring stringent safety protocols and limiting the scale of operation. Furthermore, the selectivity of introducing the triazole ring at the specific 2β-position is notoriously difficult to control, often leading to complex mixtures of isomers. Consequently, the overall yield of the desired product in these conventional processes is extremely poor, frequently reported in the range of merely 9% to 27%. Such low efficiency results in excessive waste generation and inflated production costs, rendering these methods economically unviable for large-scale commercialization.
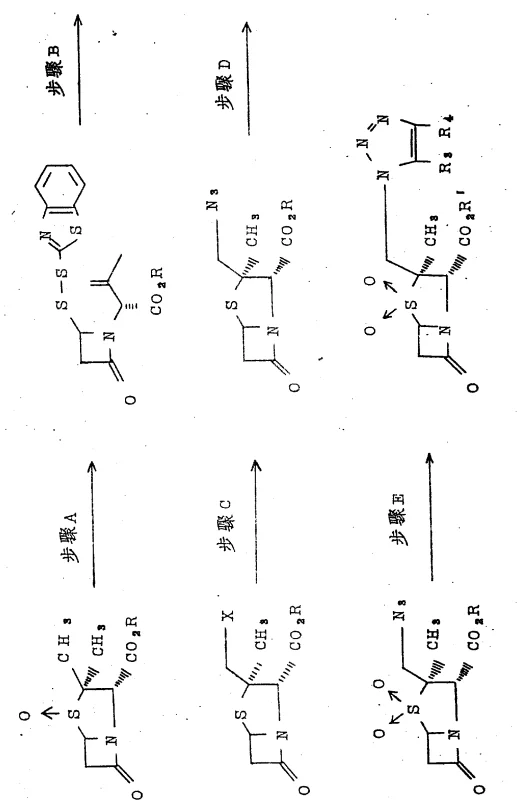
The Novel Approach
In stark contrast to the perilous and inefficient legacy methods, the novel approach disclosed in the patent data streamlines the entire synthesis into a single, robust chemical transformation. This method capitalizes on the unique reactivity of penicillanic acid sulfoxide derivatives when reacted directly with triazole derivatives. By eliminating the need for halogenated intermediates and explosive azides, the process inherently mitigates the most significant safety risks associated with beta-lactam modification. The reaction proceeds smoothly in common organic solvents such as acetonitrile or methyl ethyl ketone under moderate thermal conditions. This simplification allows for a dramatic improvement in process throughput and operational safety. Moreover, the direct substitution mechanism exhibits superior regioselectivity, ensuring that the triazole group is installed precisely at the 2β-position while maintaining the critical 2α-methyl configuration. This high level of stereocontrol minimizes the formation of impurities, thereby reducing the burden on downstream purification processes. For supply chain managers, this translates to a more reliable and predictable production schedule, essential for maintaining the continuity of high-purity pharmaceutical intermediates required for global drug markets.
Mechanistic Insights into Sulfoxide-Mediated Nucleophilic Substitution
The core of this technological breakthrough lies in the activation of the penicillane ring system via the sulfoxide functionality. In the presence of heat and appropriate solvent conditions, the sulfoxide group at the 1-position acts as a potent leaving group activator, facilitating the nucleophilic attack by the nitrogen atom of the triazole derivative. This mechanism avoids the formation of free carbocation intermediates that often lead to racemization or ring-opening degradation products common in acid-catalyzed substitutions. Instead, the reaction likely proceeds through a concerted or tight-ion-pair mechanism that preserves the stereochemical integrity of the beta-lactam ring. The steric bulk of the 2α-methyl group further directs the incoming nucleophile to the 2β-face, ensuring the correct stereochemistry required for biological activity. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate or optimize the process, as it highlights the importance of maintaining anhydrous conditions and precise temperature control to maximize the efficiency of the sulfoxide displacement. This deep chemical understanding allows for better troubleshooting and scale-up strategies, ensuring that the theoretical benefits of the patent are realized in practical manufacturing scenarios.
Furthermore, the impurity profile of this novel route is significantly cleaner compared to traditional methods. By avoiding the use of halogenating agents and azide salts, the process eliminates entire classes of genotoxic impurities and heavy metal contaminants that are difficult to purge from the final active pharmaceutical ingredient. The primary byproducts are typically reduced sulfur species which are easier to separate via standard aqueous workups or chromatography. This inherent purity advantage is critical for meeting the stringent regulatory standards imposed by health authorities worldwide. For quality control laboratories, this means fewer out-of-specification results and a more robust validation process. The ability to consistently produce material with high chemical and optical purity directly impacts the bioavailability and efficacy of the final drug product. Therefore, adopting this sulfoxide-mediated pathway is not merely a cost-saving measure but a strategic quality enhancement that aligns with the principles of Quality by Design (QbD) in modern pharmaceutical development.
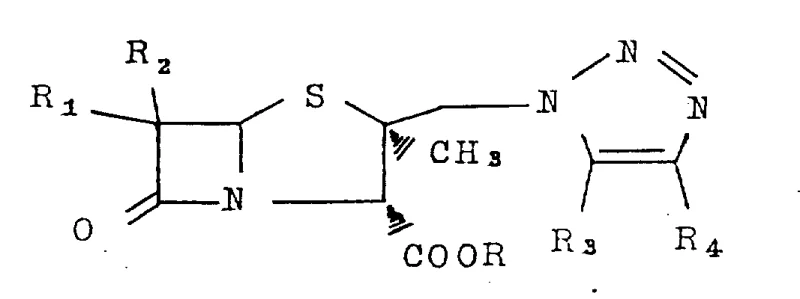
How to Synthesize 2α-Methyl-2β-(1,2,3-triazol-1-yl)methylpenicillane Efficiently
Implementing this synthesis requires careful attention to reaction parameters to ensure optimal yield and safety. The process involves charging a pressure-rated vessel with the penicillanic acid sulfoxide precursor and the silylated triazole nucleophile in a polar aprotic solvent. The mixture is then heated to promote the substitution reaction. While the patent describes various substituents for the R groups, the core protocol remains consistent across different derivatives. Detailed standard operating procedures regarding stoichiometry, specific temperature ramps, and workup techniques are essential for reproducibility. For a comprehensive guide on the exact experimental conditions and purification methods validated in the patent examples, please refer to the standardized synthesis steps outlined below.
- Mix penicillanic acid sulfoxide derivative (Formula III) with a triazole derivative (Formula IV) in a solvent such as acetonitrile or methyl ethyl ketone.
- Heat the reaction mixture in a sealed tube or autoclave at temperatures between 110°C and 120°C for approximately 3 to 5 hours.
- Isolate the product via standard workup procedures including solvent removal and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers transformative benefits for procurement and supply chain operations. The elimination of hazardous reagents such as organic azides and acetylenes removes the need for specialized explosion-proof facilities and costly safety mitigation systems. This fundamental shift in process safety profile allows for manufacturing in a broader range of facilities, thereby increasing supply flexibility and reducing overhead costs associated with hazard management. Additionally, the reduction in synthetic steps from a multi-stage sequence to a single pot reaction drastically cuts down on solvent consumption, labor hours, and equipment occupancy time. These operational efficiencies compound to deliver significant cost reductions without compromising on the quality of the final intermediate. For procurement managers negotiating contracts, this technology provides a strong leverage point for securing more competitive pricing on critical beta-lactam building blocks.
- Cost Reduction in Manufacturing: The streamlined one-step nature of this process inherently lowers the cost of goods sold (COGS). By removing the need for isolating unstable halogenated intermediates and performing multiple purification cycles, the consumption of raw materials and utilities is significantly minimized. The avoidance of expensive and dangerous reagents further contributes to a leaner cost structure. This economic efficiency allows manufacturers to offer more competitive pricing models to their clients, fostering long-term partnerships based on value and reliability rather than just transactional volume.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of stable, commercially available starting materials. Unlike the bespoke and often scarce reagents required for the conventional azide route, the sulfoxide and triazole precursors used in this method are robust and easier to source in bulk quantities. This stability in raw material supply reduces the risk of production stoppages due to vendor shortages. Furthermore, the simplified process flow reduces the likelihood of batch failures, ensuring a consistent and dependable output of high-purity intermediates that keeps downstream API production lines running smoothly.
- Scalability and Environmental Compliance: The reaction conditions described, involving heating in sealed vessels at moderate pressures, are highly scalable using standard industrial autoclaves. This scalability ensures that the process can meet the demands of commercial production volumes ranging from pilot batches to multi-ton annual requirements. Additionally, the greener profile of the process, characterized by the absence of heavy metals and explosive waste streams, simplifies environmental compliance and waste disposal. This alignment with green chemistry principles not only reduces regulatory burdens but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature. They are designed to clarify the operational feasibility and strategic advantages of adopting this method for the production of penicillane derivatives. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing manufacturing portfolios.
Q: What are the primary safety advantages of this new synthesis method?
A: Unlike conventional methods that require explosive azide and acetylene compounds, this novel process utilizes stable sulfoxide and triazole precursors, significantly reducing operational hazards and eliminating the need for specialized explosion-proof infrastructure.
Q: How does the yield compare to traditional multi-step routes?
A: Traditional routes often suffer from low overall yields ranging between 9% and 27% due to multiple purification steps. This direct substitution method achieves isolated yields typically exceeding 40-50% in a single reaction step, drastically improving material efficiency.
Q: Can this process be scaled for commercial API production?
A: Yes, the reaction conditions involving heating in solvents like acetonitrile at moderate pressures (1-5 atm) are highly amenable to scale-up using standard industrial autoclaves, ensuring consistent supply continuity for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2α-Methyl-2β-(1,2,3-triazol-1-yl)methylpenicillane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the potential of the sulfoxide-mediated substitution method described in CN1022835C and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from laboratory concept to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest global standards for safety and efficacy.
We invite you to collaborate with us to unlock the full commercial potential of this advanced synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your project needs, obtain specific COA data for our available intermediates, and receive detailed route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable and innovative chemical solutions.
