Scalable Asymmetric Synthesis of Chiral Beta-Amino Acid Intermediates for Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral building blocks, particularly beta-amino acids, which serve as critical scaffolds in the development of protease inhibitors and peptidomimetics. Patent CN1119321C presents a significant advancement in this domain by disclosing an efficient asymmetric synthesis route for ethyl 3S-amino-4-pentynoate and its derivatives. This specific intermediate is notably valuable as a precursor for platelet aggregation inhibitors, highlighting its strategic importance in cardiovascular drug discovery. The disclosed technology moves beyond traditional resolution methods, offering a direct asymmetric pathway that leverages chiral auxiliaries and organometallic coupling to achieve high stereochemical fidelity. For R&D teams evaluating synthetic routes, this patent provides a compelling alternative to older lithiation-based strategies, promising improved safety profiles and operational simplicity.
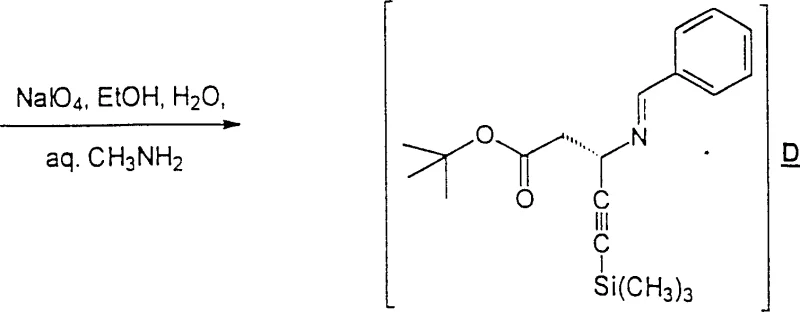
From a commercial manufacturing perspective, the ability to produce high-purity chiral intermediates without relying on preparative chromatography is a game-changer for cost structures. The process described in CN1119321C utilizes a Reformatsky-type reaction mediated by zinc, which is inherently safer and more scalable than cryogenic lithiation processes found in prior art such as US Patent 5,536,869. By integrating chiral induction early in the synthesis via L-phenylglycinol, the method ensures that the stereochemistry is locked in during the carbon-carbon bond-forming step. This approach not only streamlines the workflow but also aligns with modern green chemistry principles by reducing solvent waste associated with extensive purification steps. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is essential for translating lab-scale success into commercial reality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino acids like ethyl 3S-amino-4-pentynoate has been plagued by significant operational hurdles that impact both cost and scalability. Prior art methods, such as those detailed in US Patent 5,344,957, often rely on the use of n-butyllithium to generate acetylide anions, necessitating strict cryogenic conditions typically below -20°C to prevent side reactions and ensure safety. These low-temperature requirements impose a heavy energy burden on manufacturing facilities and limit the size of batches that can be safely processed. Furthermore, many conventional routes produce racemic mixtures that require subsequent resolution steps, effectively capping the theoretical yield at 50% unless dynamic kinetic resolution is employed. The reliance on chromatographic purification for enantiomeric enrichment is another major bottleneck, as column chromatography is notoriously difficult to scale and generates substantial volumes of hazardous solvent waste, driving up disposal costs and environmental compliance burdens.
The Novel Approach
The methodology outlined in CN1119321C circumvents these challenges by employing a chiral pool strategy combined with a zinc-mediated Reformatsky reaction. Instead of generating reactive acetylides with lithium bases, the process utilizes tert-butyl bromozincacetate, which reacts with a chiral imine derived from L-phenylglycinol and a propynal derivative. This shift eliminates the need for pyrophoric reagents and extreme cooling, allowing the reaction to proceed at more manageable temperatures ranging from -10°C to ambient conditions. Crucially, the chiral information from the phenylglycinol auxiliary directs the facial selectivity of the nucleophilic attack, resulting in high diastereomeric excess (de) directly from the reaction mixture. The downstream processing involves oxidative cleavage of the auxiliary using sodium periodate, a mild and selective oxidant, followed by hydrolysis. This sequence allows for the recovery of the chiral auxiliary and the isolation of the product through crystallization of stable salts, such as the p-toluenesulfonate, thereby completely bypassing the need for chromatography.
Mechanistic Insights into Zinc-Mediated Reformatsky Coupling
The core of this synthetic innovation lies in the stereoselective addition of the organozinc species to the chiral imine. The reaction begins with the condensation of 3-(trimethylsilyl)-2-propynal with L-phenylglycinol in toluene, forming an imine intermediate that serves as the electrophile. The presence of the hydroxyl group on the phenylglycinol moiety is critical, as it can coordinate with the zinc center of the Reformatsky reagent, creating a rigid cyclic transition state. This coordination locks the conformation of the imine, forcing the incoming nucleophile to attack from the less hindered face, thus establishing the desired 3S configuration with high fidelity. The use of polar aprotic solvents like N-methylpyrrolidone (NMP) or dimethyl sulfoxide (DMSO) further enhances the reactivity of the organozinc species by stabilizing the zinc cation and increasing the nucleophilicity of the carbon-zinc bond. This mechanistic understanding is vital for process chemists aiming to optimize reaction parameters such as temperature and addition rates to maximize yield and stereocontrol.
Following the coupling step, the removal of the chiral auxiliary is achieved through oxidative cleavage, a step that distinguishes this process from acid-hydrolysis-only methods. Treatment with sodium periodate (NaIO4) in the presence of methylamine selectively cleaves the carbon-carbon bond between the amino acid backbone and the phenylglycinol side chain, regenerating the imine functionality while releasing the chiral alcohol. This oxidative step is remarkably clean and proceeds under mild aqueous conditions, minimizing the risk of racemization which can occur under harsh acidic or basic hydrolysis conditions. The resulting imine is then hydrolyzed using p-toluenesulfonic acid to release the free amine, which is subsequently converted into a stable salt for isolation. This multi-step cascade demonstrates a sophisticated control over reactivity, ensuring that the sensitive alkyne functionality remains intact throughout the synthesis while achieving the necessary structural transformations to reveal the target beta-amino acid scaffold.
How to Synthesize Ethyl 3S-Amino-4-Pentynoate Efficiently
Implementing this synthesis requires careful attention to the preparation of the Reformatsky reagent and the control of exotherms during the coupling phase. The patent details a specific protocol where zinc metal is activated with dibromoethane in THF before reacting with tert-butyl bromoacetate to generate the organozinc species in situ. This reagent is then transferred into a solution of the chiral imine maintained at low temperatures to ensure high diastereoselectivity. The workup involves quenching with ammonium chloride and extracting the aminoalcohol intermediate, which is then subjected to the oxidative cleavage conditions. The final steps involve transesterification to convert the tert-butyl ester to the ethyl ester and desilylation to reveal the terminal alkyne. For a detailed breakdown of the specific molar ratios, temperature profiles, and isolation techniques required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Condense 3-(trimethylsilyl)-2-propynal with L-phenylglycinol in toluene to form the chiral imine intermediate.
- React the imine with tert-butyl bromozincacetate (Reformatsky reagent) in NMP/THF to establish the carbon-carbon bond with high diastereoselectivity.
- Perform oxidative cleavage using sodium periodate followed by acid hydrolysis and transesterification to yield the final ethyl ester hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1119321C offers tangible benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the supply chain for raw materials; the process utilizes commodity chemicals like zinc dust, bromoacetates, and commercially available chiral amino alcohols, rather than specialized, high-cost organolithium reagents that often have long lead times and stringent storage requirements. By shifting to a zinc-mediated process, manufacturers can reduce their dependency on volatile supply markets for pyrophoric materials, thereby enhancing supply chain resilience. Furthermore, the elimination of chromatographic purification steps significantly reduces the consumption of high-purity solvents and silica gel, leading to substantial cost reduction in pharmaceutical intermediates manufacturing. This efficiency gain allows for a more competitive pricing structure for the final API intermediate, making it an attractive option for generic drug manufacturers looking to optimize their bill of materials.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive purification technologies and hazardous reagents. Traditional lithiation routes require specialized equipment capable of handling cryogenic temperatures and inert atmospheres, which represents a significant capital expenditure. In contrast, the Reformatsky approach described here operates at near-ambient temperatures after the initial cooling phase, allowing the use of standard glass-lined or stainless steel reactors without expensive cooling jackets designed for extreme lows. Additionally, the ability to isolate intermediates as crystalline salts, such as the p-toluenesulfonate, enables purification via recrystallization rather than chromatography. This switch from batch chromatography to continuous or semi-continuous crystallization drastically lowers operating expenses related to solvent recovery and waste disposal, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any commercial manufacturing operation, and this synthetic route bolsters reliability by utilizing robust and widely available starting materials. The chiral auxiliary, L-phenylglycinol, is a bulk chemical available from multiple global suppliers, mitigating the risk of single-source bottlenecks. Moreover, the process tolerance is relatively high; the patent indicates that the reaction can proceed effectively even with slight variations in reagent quality, provided the activation of zinc is managed correctly. This robustness reduces the rate of batch failures and reprocessing, ensuring a steady flow of material to downstream synthesis stages. For supply chain heads, this translates to more predictable lead times and the ability to scale production volumes rapidly in response to market demand without the need for complex technology transfers or specialized contractor manufacturing organizations.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for scale-up. The use of zinc instead of lithium reduces the fire hazard profile of the facility, lowering insurance premiums and safety compliance costs. From an environmental standpoint, the avoidance of chlorinated solvents often associated with lithiation workups and the reduction of silica waste from chromatography aligns with increasingly stringent global environmental regulations. The process generates aqueous waste streams containing zinc salts which are easier to treat and dispose of compared to heavy metal catalyst residues or complex organic mixtures. This environmental compatibility facilitates smoother regulatory approvals and supports corporate sustainability goals, making the technology not just chemically superior but also strategically aligned with modern ESG (Environmental, Social, and Governance) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects.
Q: How does this process ensure high optical purity without chromatography?
A: The process utilizes L-phenylglycinol as a chiral auxiliary which directs the stereochemistry during the Reformatsky addition. The resulting diastereomers can be separated or enriched through crystallization of intermediate salts (such as the p-toluenesulfonate salt), eliminating the need for expensive preparative chromatography.
Q: What are the safety advantages over traditional lithiation methods?
A: Unlike prior art methods requiring n-butyllithium at cryogenic temperatures, this invention employs a zinc-mediated Reformatsky reaction. This avoids pyrophoric reagents and extreme low-temperature conditions, significantly improving operational safety and reducing energy consumption for cooling.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the patent explicitly describes the process as amenable to scale-up. It uses readily available starting materials, avoids complex separation techniques like chromatography, and relies on robust unit operations such as crystallization and filtration, which are ideal for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 3S-Amino-4-Pentynoate Supplier
The technological advancements detailed in CN1119321C represent a significant leap forward in the efficient production of chiral beta-amino acid intermediates, yet translating this intellectual property into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling moisture-sensitive organometallic reactions safely, alongside rigorous QC labs that ensure every batch meets stringent purity specifications required by global regulatory bodies. We understand that consistency is key in pharmaceutical supply, and our team is dedicated to optimizing every step of this Reformatsky-based synthesis to deliver high-quality intermediates that accelerate your drug development timelines.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing protocols, we can help you realize the full economic potential of this patent-protected route. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to secure your supply chain with high-purity pharmaceutical intermediates that drive innovation and efficiency in your therapeutic programs.
