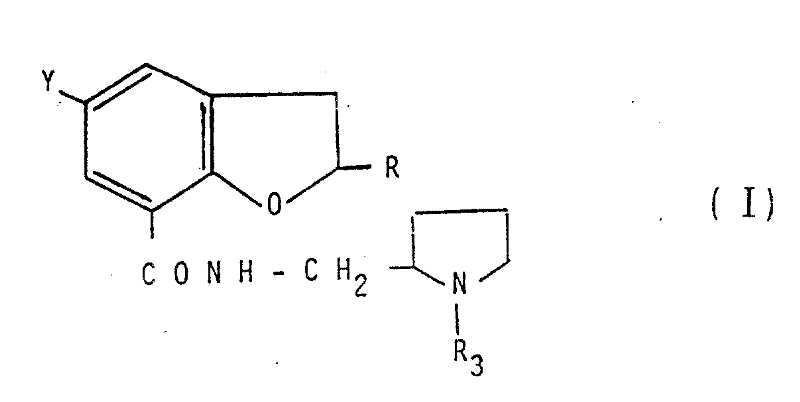
Scalable Synthesis of Dihydrobenzofuran Carboxamide Derivatives for Global Neuroleptic Manufacturing
The pharmaceutical landscape for central nervous system disorders continues to demand highly specialized intermediates that balance structural complexity with manufacturing feasibility. Patent CN1016420B introduces a sophisticated preparation method for new dihydrobenzofuran carboxamide derivatives, which serve as pivotal scaffolds in the development of potent neuroleptic agents. This technology outlines a versatile synthetic pathway that allows for precise modulation of the substituents at the 5-position of the benzofuran ring, directly influencing the pharmacological efficacy and safety profile of the final drug substance. By leveraging specific amidation and sulfonation techniques, manufacturers can achieve distinct chemical entities that exhibit superior antagonism against apomorphine-induced behaviors in preclinical models. The strategic design of these molecules addresses the longstanding challenge of optimizing therapeutic windows while maintaining robust synthetic accessibility for global supply chains. Understanding the nuances of this patented methodology is essential for R&D teams aiming to secure a competitive edge in the production of next-generation antipsychotic medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for benzofuran-based neuroleptics often suffer from harsh reaction conditions that compromise the integrity of sensitive functional groups and lead to complex impurity profiles. Conventional methods frequently rely on non-selective alkylation or acylation steps that require extensive downstream purification, resulting in significant yield losses and increased operational costs. Furthermore, older methodologies may struggle to introduce diverse sulfonyl or sulfamoyl moieties efficiently, limiting the chemical space available for structure-activity relationship studies. The use of unstable intermediates in legacy processes can also pose safety hazards during scale-up, particularly when handling reactive acid chlorides or strong sulfonating agents without adequate temperature control. These inefficiencies create bottlenecks in the supply chain, causing delays in clinical material availability and hindering the rapid iteration required for modern drug discovery programs. Consequently, there is a critical industry need for more refined protocols that enhance both safety and chemical precision.
The Novel Approach
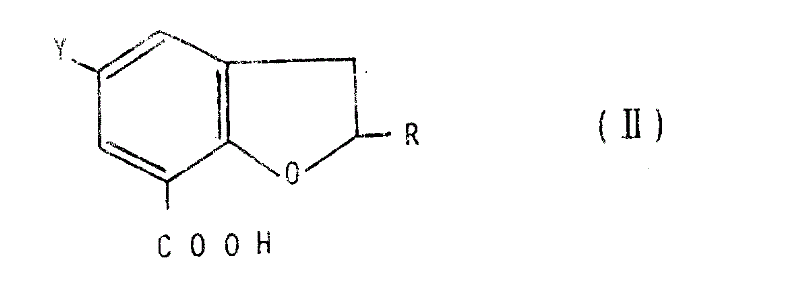
The methodology disclosed in CN1016420B presents a breakthrough by utilizing a modular approach to construct the dihydrobenzofuran core with high regioselectivity and yield. This novel strategy employs controlled chlorosulfonation followed by careful amination to install the requisite sulfonamide functionality without degrading the sensitive carboxylic acid handle. By activating the acid moiety through mixed anhydrides or acid chlorides under mild conditions, the process ensures efficient coupling with sterically hindered pyrrolidine amines. The ability to isolate stable intermediates, such as the 5-chlorosulfonyl or 5-methylsulfamoyl derivatives, provides crucial checkpoints for quality control before the final amidation step. This stepwise progression minimizes the formation of side products and simplifies the purification workflow, ultimately leading to higher overall throughput. Such advancements represent a significant leap forward in the manufacturing of complex pharmaceutical intermediates, offering a reliable foundation for commercial production.
Mechanistic Insights into Amidation and Sulfonamide Functionalization
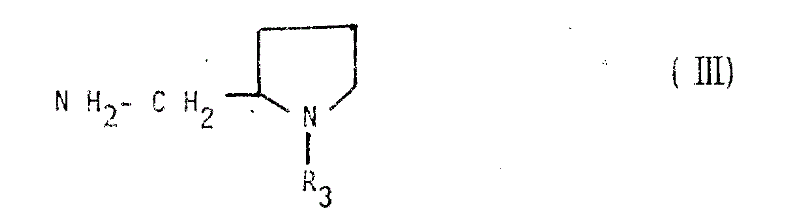
The core transformation in this synthesis involves the nucleophilic attack of a substituted pyrrolidine amine on an activated dihydrobenzofuran carboxylic acid derivative. Mechanistically, the activation of the carboxyl group using reagents like thionyl chloride or ethyl chloroformate generates a highly electrophilic species that readily reacts with the amine nitrogen. This reaction is typically conducted in aprotic solvents such as chloroform or dimethylformamide to solubilize the reactants and facilitate heat dissipation. The presence of a base, such as triethylamine, is critical to neutralize the acid byproducts formed during the coupling, thereby driving the equilibrium towards the desired amide product. Careful temperature maintenance between 0°C and 10°C during the addition phase prevents exothermic runaways and reduces the risk of racemization at chiral centers adjacent to the carbonyl group. This precise control over reaction kinetics is fundamental to achieving the high purity standards demanded by regulatory agencies for CNS-active compounds.
Impurity control is further enhanced by the specific sequence of introducing the sulfonyl group prior to the final amide bond formation. By establishing the substitution pattern on the aromatic ring early in the synthesis, chemists can leverage recrystallization techniques to remove isomeric impurities before they become embedded in the final molecular architecture. The patent details various workup procedures, including acid-base extractions and activated carbon treatments, which effectively remove colored impurities and residual metal catalysts. For instance, the conversion of thioethers to sulfones using hydrogen peroxide allows for the oxidation of sulfur without affecting the dihydrofuran ring stability. This orthogonal reactivity ensures that the final product meets stringent specifications for heavy metals and organic volatiles. Such meticulous attention to detail in the purification process underscores the robustness of this manufacturing route for high-value pharmaceutical ingredients.
How to Synthesize Dihydrobenzofuran Carboxamide Efficiently
Executing this synthesis requires a disciplined approach to reagent addition and thermal management to maximize yield and safety. The process begins with the preparation of the acid intermediate, followed by activation and subsequent coupling with the amine component under strictly anhydrous conditions. Detailed standardized operating procedures are essential to replicate the high yields reported in the patent examples across different production scales. Operators must be trained to recognize visual endpoints, such as color changes during acidification or crystallization patterns during cooling, to ensure batch-to-batch consistency. The following guide outlines the critical phases of the operation, emphasizing the importance of in-process controls.
- Preparation of the dihydrobenzofuran carboxylic acid scaffold via chlorosulfonation or reduction pathways depending on the desired substituent.
- Activation of the carboxylic acid group using thionyl chloride or mixed anhydride methods to facilitate nucleophilic attack.
- Coupling with substituted pyrrolidine amines under controlled low-temperature conditions followed by rigorous purification and salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this patented synthesis route offers substantial benefits regarding cost stability and raw material security. The reliance on commodity chemicals such as thionyl chloride, triethylamine, and common organic solvents mitigates the risk of supply disruptions associated with exotic or proprietary reagents. This accessibility ensures that production schedules can be maintained even during periods of market volatility, providing a reliable source of critical intermediates for downstream drug manufacturing. Additionally, the high yields achieved in key steps, such as the sulfonamide formation and final amidation, translate directly into reduced waste generation and lower disposal costs. By optimizing atom economy and minimizing the number of isolation steps, manufacturers can significantly lower the overall cost of goods sold without compromising on quality standards. These economic efficiencies make the technology highly attractive for long-term supply agreements and strategic partnerships.
- Cost Reduction in Manufacturing: The streamlined workflow eliminates the need for expensive transition metal catalysts often required in cross-coupling reactions, thereby reducing both material costs and the burden of heavy metal testing. The ability to recycle solvents like chloroform and ethyl acetate further enhances the economic viability of the process on a multi-ton scale. Moreover, the high purity of the crude product reduces the consumption of chromatography media and recrystallization solvents, leading to significant operational savings. These factors collectively contribute to a more competitive pricing structure for the final active pharmaceutical ingredient, benefiting the entire value chain.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for flexible manufacturing across multiple facilities, reducing the risk of single-source dependency. The intermediates generated are stable and can be stored for extended periods, enabling producers to build strategic inventory buffers against demand fluctuations. This stability is crucial for ensuring continuous supply to pharmaceutical clients who operate on tight just-in-time delivery schedules. Furthermore, the well-documented nature of the chemistry facilitates technology transfer between sites, ensuring that quality remains consistent regardless of the production location.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor equipment and avoiding extreme pressures or temperatures that would require specialized infrastructure. Waste streams are primarily aqueous and organic solvents that can be treated using conventional effluent treatment plants, ensuring compliance with environmental regulations. The avoidance of persistent organic pollutants and the use of recyclable materials align with modern green chemistry principles, enhancing the corporate sustainability profile of the manufacturer. This alignment is increasingly important for procurement teams evaluating suppliers based on environmental, social, and governance criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these dihydrobenzofuran derivatives. Answers are derived from the specific experimental data and claims found within the patent documentation to ensure accuracy. These insights are intended to assist decision-makers in evaluating the feasibility of integrating this technology into their existing portfolios. Comprehensive understanding of these aspects is vital for successful project implementation.
Q: What are the critical purity specifications for these neuroleptic intermediates?
A: The patent emphasizes rigorous recrystallization steps using solvents like ethanol and isopropanol to ensure high melting point consistency and minimal impurity profiles suitable for CNS active drugs.
Q: How does the sulfonamide modification impact the pharmacological profile?
A: Variations in the sulfonyl group (e.g., methylsulfamoyl vs. cyclopropylmethylsulfonyl) significantly alter the LD50 and catalepsy-inducing properties, allowing for fine-tuning of therapeutic indices.
Q: Is the synthesis route adaptable for large-scale commercial production?
A: Yes, the process utilizes common industrial reagents like thionyl chloride and triethylamine in standard solvents such as chloroform and ethyl acetate, facilitating straightforward scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydrobenzofuran Carboxamide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at adapting complex routes like the one described in CN1016420B to meet stringent purity specifications required for neuroleptic intermediates. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against identity and potency standards. Our commitment to quality ensures that the dihydrobenzofuran scaffolds we deliver are perfectly suited for subsequent formulation and clinical testing phases. Clients trust us to navigate the intricacies of sulfonamide chemistry with precision and reliability.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how we can optimize your supply chain. Partnering with us means gaining access to a wealth of chemical expertise and a dedication to delivering excellence in every kilogram shipped. Let us help you accelerate your drug development timeline with our superior manufacturing capabilities.
