Advanced Synthesis of Enantiomerically Pure Cycloalkanol Indole Carboxylic Acids for Commercial Scale-Up
Advanced Synthesis of Enantiomerically Pure Cycloalkanol Indole Carboxylic Acids for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust methodologies for generating chiral intermediates that serve as the backbone for potent therapeutic agents. Patent CN1184109A discloses a groundbreaking approach for the preparation of enantiomerically pure cycloalkanol indole-carboxylic acids, azaindole carboxylic acids, and pyrimido[1,2-a]-indole-carboxylic acids, which are critical precursors for synthesizing antiatherogenic active compounds. This technology addresses the longstanding inefficiencies associated with resolving racemic mixtures, offering a pathway that combines high stereochemical control with operational simplicity. By leveraging diastereoselective alkylation of chiral esters derived from tolueneacetic acid, manufacturers can bypass the economic and technical bottlenecks of traditional chromatographic separation. For procurement leaders and R&D directors alike, understanding this synthetic route is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at a commercial scale.
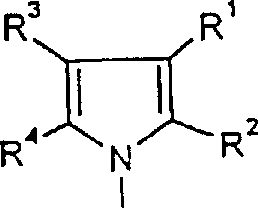
The structural versatility of these compounds, defined by various substituents on the indole and cycloalkyl rings, allows for the fine-tuning of biological activity while maintaining a consistent and scalable manufacturing process. The ability to produce these complex molecules with specific stereochemistry is not merely a scientific achievement but a commercial imperative, ensuring that downstream drug development programs are not hindered by supply constraints or inconsistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of enantiomerically pure cycloalkanol indole-carboxylic acids has relied heavily on the separation of racemic mixtures through techniques such as chromatographic resolution or fractional crystallization of diastereomers. These conventional methods suffer from inherent inefficiencies, primarily because they typically yield a maximum of 50% of the desired enantiomer, leaving the remaining half as an unwanted byproduct that often cannot be recycled into the process. Furthermore, chromatographic separation imposes stringent equipment requirements, necessitating specialized columns and vast quantities of solvents, which drastically increases both the capital expenditure and the operational costs associated with manufacturing. The economic impact of losing half the theoretical yield is particularly damaging in large-scale industrial production, where raw material costs and waste disposal fees can erode profit margins significantly. Additionally, the chiral resolving agents used in these traditional processes are frequently expensive and themselves require complex synthetic routes to prepare, further compounding the cost burden and supply chain complexity for manufacturers seeking cost reduction in API manufacturing.
The Novel Approach
In stark contrast to these legacy techniques, the method described in patent CN1184109A introduces a highly efficient strategy that begins with the esterification of tolueneacetic acid using a chiral alcohol, followed by a diastereoselective substitution at the alpha carbon atom. This innovative route allows for the direct construction of the chiral center with high specificity, effectively circumventing the 50% yield loss associated with racemic resolution. The process involves halogenating the toluene group of the chiral ester and subsequently reacting it with a suitable cycloalkanoindole, azaindole, or pyrimido[1,2-a]-indole core. 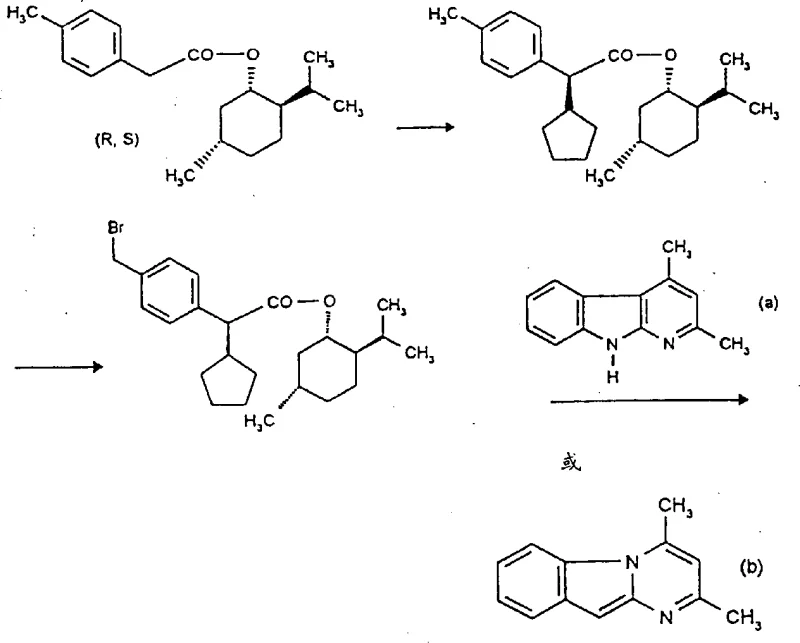
This sequence ensures that the stereochemical integrity is maintained throughout the synthesis, resulting in enantiomerically pure carboxylic acids with exceptional purity levels. The novelty lies not just in the chemical transformations but in the strategic use of chiral auxiliaries that can be removed or transformed later, enabling a linear and high-yielding synthesis that is far more amenable to industrial scale-up than the cumbersome separation techniques of the past.
Mechanistic Insights into Diastereoselective Alkylation and Coupling
The core of this technological advancement rests on the mechanistic precision of the cis-selective alkylation step, where a chiral ester of tolueneacetic acid reacts with a cycloalkyl halide in the presence of a strong base. The steric environment created by the chiral alcohol moiety, such as menthol or phenylethylamine derivatives, directs the incoming alkyl group to a specific face of the enolate intermediate, thereby establishing the desired stereochemistry with high diastereomeric excess. This step is critical because it sets the chiral foundation for the entire molecule, and the patent highlights that by carefully selecting the solvent and base, unwanted diastereomers can even be epimerized back to the desired form, further optimizing the overall yield. The reaction conditions are mild yet effective, typically conducted in polar aprotic solvents like DMF at controlled temperatures, ensuring that the delicate chiral information is not scrambled during the bond-forming event.
Following the establishment of the chiral center, the synthetic route proceeds through a radical halogenation of the aromatic methyl group, converting it into a reactive benzyl halide without affecting the adjacent stereocenter. This chemoselectivity is paramount, as harsh conditions could lead to racemization at the alpha-carbon, ruining the enantiomeric purity achieved in the previous step. The resulting benzyl halide then undergoes a nucleophilic substitution with the nitrogen-containing heterocyclic core, such as a carboline or azaindole derivative. 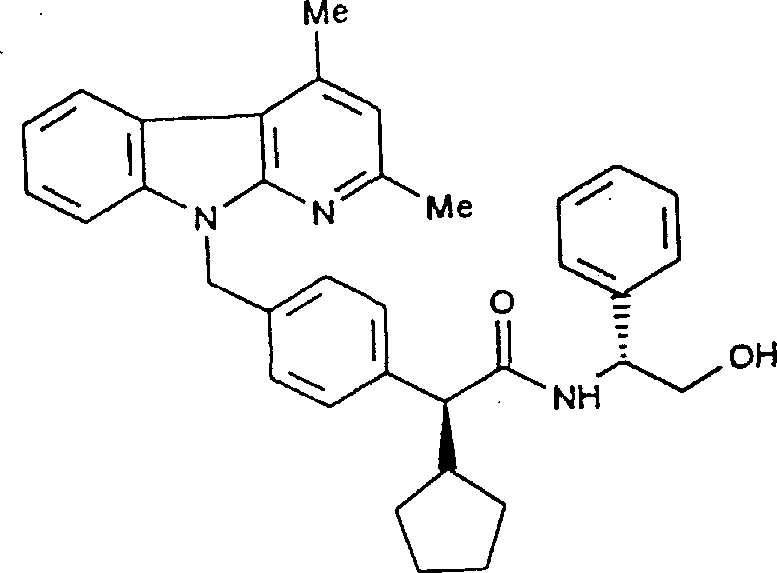
Mechanistically, this coupling proceeds smoothly under basic conditions, and subsequent hydrolysis or activation of the ester group yields the final carboxylic acid or its reactive acid chloride derivative. The entire sequence is designed to minimize side reactions and maximize the retention of optical purity, making it a robust platform for generating high-value pharmaceutical intermediates.
How to Synthesize Enantiomerically Pure Cycloalkanol Indole Carboxylic Acids Efficiently
Implementing this synthesis requires a disciplined approach to reaction conditions and purification strategies to fully realize the benefits of the patented method. The process begins with the preparation of the chiral ester, followed by the crucial alkylation step where temperature control and base selection are vital for achieving high diastereoselectivity. Subsequent halogenation and coupling steps must be monitored closely to prevent any degradation of the chiral center, and the final purification often involves simple crystallization rather than complex chromatography. For detailed operational parameters, including specific molar ratios, solvent choices, and temperature profiles that ensure optimal results, please refer to the standardized synthesis guide below.
- Esterify tolueneacetic acid with a chiral alcohol (e.g., menthol) to form a chiral ester, followed by cis-selective alkylation with a cycloalkyl halide to introduce the chiral center with high diastereomeric excess.
- Perform radical halogenation on the methyl group of the aromatic ring using N-bromosuccinimide (NBS) or similar agents to generate the reactive benzyl bromide intermediate without racemization.
- Couple the halogenated chiral ester with the appropriate cycloalkanoindole or azaindole core under basic conditions, followed by hydrolysis or activation to yield the final enantiomerically pure carboxylic acid or its reactive derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers transformative advantages that directly impact the bottom line and operational resilience. By shifting away from resolution-based processes that inherently discard half the material, manufacturers can achieve a drastic improvement in mass balance and raw material utilization. This efficiency translates into substantial cost savings, as fewer starting materials are required to produce the same amount of active intermediate, and the waste stream is significantly reduced. Furthermore, the reliance on standard unit operations like crystallization and filtration, rather than specialized chromatographic columns, simplifies the equipment landscape and reduces the risk of production bottlenecks. This streamlined approach enhances supply chain reliability by shortening production cycles and minimizing the dependency on scarce or expensive chiral resolving agents that often plague traditional routes.
- Cost Reduction in Manufacturing: The elimination of the 50% yield loss inherent in racemic resolution fundamentally alters the cost structure of producing these intermediates. By synthesizing the desired enantiomer directly through diastereoselective alkylation, the process maximizes the conversion of raw materials into valuable product, thereby lowering the cost per kilogram significantly. Additionally, the avoidance of expensive chromatographic media and the reduction in solvent consumption for separation purposes contribute to a leaner and more cost-effective manufacturing process. The ability to recycle mother liquors through epimerization further optimizes resource usage, ensuring that every gram of input contributes to the final output, which is a critical factor for maintaining competitiveness in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more predictable and stable supply of critical intermediates. Since the process does not rely on the availability of specific chiral resolving agents that may have limited suppliers, the risk of supply disruption is markedly reduced. The use of commodity chemicals and standard reagents means that sourcing is straightforward and less susceptible to market volatility. Moreover, the scalability of the process, demonstrated by its successful execution in multi-kilogram batches within the patent examples, assures partners that large-volume orders can be fulfilled consistently without the need for extensive process re-engineering or capacity expansion.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a cleaner profile compared to traditional resolution techniques. The reduction in solvent waste and the elimination of complex separation steps align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The process generates less hazardous waste, simplifying disposal and lowering associated costs. Furthermore, the simplicity of the workup procedures, often involving straightforward crystallization and filtration, makes the technology highly scalable from pilot plant to commercial production, allowing for rapid response to market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these enantiomerically pure intermediates. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production pipelines or for assessing the quality of potential suppliers.
Q: How does this method improve yield compared to traditional racemic separation?
A: Traditional methods often involve separating racemic mixtures via chromatography or fractional crystallization, which theoretically limits yield to 50% for the desired enantiomer and requires expensive equipment. The patented diastereoselective alkylation method allows for the direct formation of the desired stereoisomer with high purity, significantly reducing material loss and eliminating the need for complex separation of the unwanted enantiomer.
Q: What represents the key advantage regarding equipment requirements?
A: The process utilizes standard organic synthesis techniques such as esterification, alkylation, and halogenation in common solvents like DMF or toluene. Unlike chromatographic separation which demands specialized columns and large solvent volumes, this route relies on crystallization and standard reactor vessels, making it highly suitable for large-scale industrial production with lower capital expenditure.
Q: Is the chiral integrity maintained during the halogenation and coupling steps?
A: Yes, the patent explicitly demonstrates that the halogenation of the benzylic position and the subsequent nucleophilic substitution with the indole core proceed without racemization at the alpha-carbon of the carboxylic acid function. This ensures that the high enantiomeric excess achieved in the initial alkylation step is preserved throughout the synthesis of the final active pharmaceutical ingredient intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enantiomerically Pure Indole Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN1184109A can be translated into reliable, large-scale supply. We are committed to meeting stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of cycloalkanol indole carboxylic acid meets the exacting standards required for pharmaceutical applications. Our expertise in handling complex stereochemical syntheses allows us to deliver materials that support your R&D and commercial goals with unwavering consistency.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall material costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
