Advanced Synthetic Route for 4-(Sulfonylphenyl)piperidines: Scalable Manufacturing for CNS Therapeutics
Advanced Synthetic Route for 4-(Sulfonylphenyl)piperidines: Scalable Manufacturing for CNS Therapeutics
The pharmaceutical landscape for central nervous system (CNS) disorders continues to demand highly pure and structurally complex intermediates capable of modulating dopamine neurotransmission. A pivotal advancement in this domain is detailed in Chinese Patent CN101068782A, which discloses a robust and efficient process for the preparation of 4-(sulfonylphenyl)-piperidines of formula VI. These compounds serve as critical scaffolds for therapeutic agents targeting Alzheimer's disease, Parkinson's disease, and schizophrenia. The innovation lies in a strategic sequence of catalytic oxidation followed by dehydration and catalytic reduction, offering a significant departure from traditional multi-step syntheses that often suffer from low yields and impurity profiles. By optimizing the oxidation state of the sulfur moiety early in the sequence and utilizing mild catalytic hydrogenation for the final saturation, this methodology addresses key pain points in the manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-(sulfonylphenyl)piperidines has been fraught with challenges related to chemoselectivity and step economy. Conventional routes often require the installation of the sulfonyl group on a pre-formed piperidine ring or involve harsh oxidation conditions that can degrade the sensitive nitrogen-containing heterocycle. Traditional stoichiometric oxidants, such as potassium permanganate or excess peracids, generate substantial amounts of inorganic waste and metal residues, complicating downstream purification and increasing the environmental burden. Furthermore, achieving the correct oxidation state (sulfone versus sulfoxide) without over-oxidation or side reactions on the aromatic ring typically demands rigorous temperature control and extensive workup procedures. These inefficiencies translate into higher production costs, longer lead times, and potential supply chain vulnerabilities for manufacturers relying on outdated synthetic protocols.
The Novel Approach
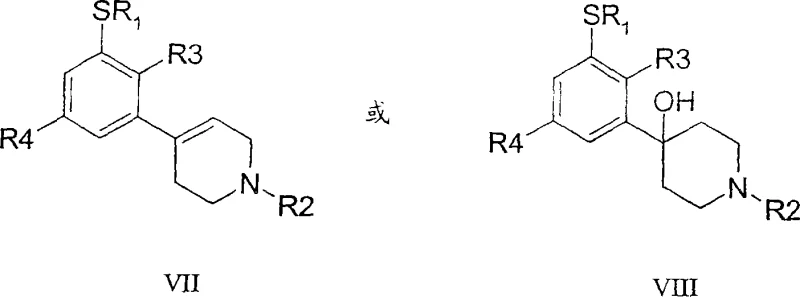
The methodology presented in the patent introduces a streamlined pathway that decouples the oxidation of the sulfur atom from the saturation of the piperidine ring. By starting with a sulfide precursor (Formula VII or VIII) and employing a catalytic oxidation system, the process achieves high conversion to the corresponding sulfone (Formula IX or X) under mild acidic conditions. This is followed by a dehydration step to generate a tetrahydropyridine intermediate, which is then subjected to catalytic hydrogenation. This modular approach allows for precise control over each transformation, minimizing the formation of by-products. The use of catalytic oxidants like sodium tungstate with hydrogen peroxide not only enhances atom economy but also simplifies the isolation of the intermediate. Consequently, this novel approach offers a reliable [Pharmaceutical Intermediates] supplier solution that balances technical feasibility with economic efficiency.
Mechanistic Insights into Catalytic Oxidation and Reduction
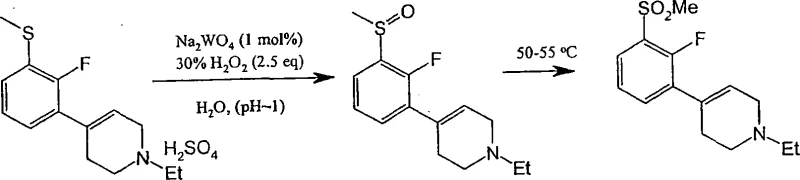
The core of this synthetic strategy relies on the selective oxidation of the thioether linkage to a sulfone using a catalytic cycle. In the preferred embodiment, a tungsten-based catalyst, such as sodium tungstate, activates hydrogen peroxide to form a peroxotungstate species. This active oxidant transfers oxygen atoms to the sulfur center of the substrate. The reaction is conducted at a pH below 3, often adjusted with sulfuric acid, which is critical for maintaining the activity of the catalyst and preventing the decomposition of hydrogen peroxide. The temperature is carefully maintained between 50°C and 55°C to ensure rapid kinetics while avoiding thermal degradation of the organic framework. This specific window allows for the complete conversion of the sulfide to the sulfone without affecting other functional groups like the fluorine substituent on the aromatic ring. The mechanistic precision here is vital for ensuring the high-purity [Pharmaceutical Intermediates] required for subsequent biological testing and clinical applications.
Following oxidation, the process may involve an acid-catalyzed dehydration if the starting material is an alcohol (Formula VIII), yielding a tetrahydropyridine derivative (Formula IX). The final step involves the catalytic reduction of the olefinic bond in the tetrahydropyridine ring. This is typically achieved using palladium on carbon (Pd/C) under a hydrogen atmosphere. The mechanism involves the adsorption of hydrogen onto the metal surface, followed by syn-addition across the double bond. The choice of solvent, such as ethanol or isopropanol, and the presence of additives like calcium acetate can influence the rate and selectivity of this reduction. By controlling the pressure (e.g., 50 psi) and temperature, the process ensures complete saturation of the ring while preserving the integrity of the sulfone group, which is susceptible to reduction under more forcing conditions. This careful orchestration of redox chemistry exemplifies the depth of process optimization achieved in this patent.
How to Synthesize 1-Ethyl-4-[2-fluoro-3-(methylsulfonyl)phenyl]piperidine Efficiently
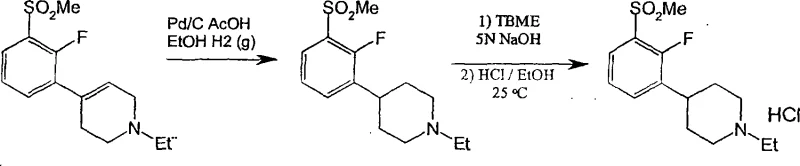
The synthesis of specific analogues, such as 1-ethyl-4-[2-fluoro-3-(methylsulfonyl)phenyl]piperidine, serves as a prime example of the platform's versatility. The procedure begins with the preparation of the sulfide precursor, often via lithiation of a bromo-fluoro-benzene derivative followed by reaction with dimethyl disulfide. This intermediate is then coupled with an N-substituted piperidone to form the alcohol, which is subsequently dehydrated to the tetrahydropyridine. The critical oxidation step utilizes aqueous hydrogen peroxide and a catalytic amount of sodium tungstate dihydrate in an acidic medium. After the oxidation is complete, the mixture is basified and extracted to isolate the sulfonyl-tetrahydropyridine. The final hydrogenation is performed in an alcoholic solvent using Pd/C, followed by salt formation to yield the stable hydrochloride salt. For the detailed standardized synthesis steps, please refer to the guide below.
- Oxidize the sulfide precursor (Formula VII or VIII) using a catalytic oxidant such as sodium tungstate and hydrogen peroxide at controlled pH and temperature.
- Perform dehydration of the resulting alcohol intermediate (Formula X) using strong acids like sulfuric acid or polyphosphoric acid to form the tetrahydropyridine derivative.
- Execute catalytic reduction of the tetrahydropyridine double bond using palladium on carbon under hydrogen pressure to obtain the final saturated piperidine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The shift from stoichiometric oxidants to catalytic systems drastically reduces the volume of hazardous waste generated per kilogram of product, aligning with modern environmental compliance standards and reducing disposal costs. Furthermore, the reagents employed, such as hydrogen peroxide and palladium on carbon, are commodity chemicals with robust global supply chains, mitigating the risk of raw material shortages. The ability to perform these reactions in common solvents like toluene, ethanol, and water simplifies solvent recovery and recycling processes, contributing to overall cost reduction in [Pharmaceutical Intermediates] manufacturing.
- Cost Reduction in Manufacturing: The implementation of catalytic oxidation eliminates the need for expensive and hazardous stoichiometric oxidants, leading to substantial cost savings in raw material procurement. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, thereby reducing the load on purification units and increasing the overall yield of the process. This efficiency translates directly into a lower cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: By utilizing widely available catalysts and oxidants, the process reduces dependency on niche suppliers. The robustness of the reaction conditions, which tolerate slight variations in temperature and pH without significant loss of yield, ensures consistent production output. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, preventing delays in drug development timelines.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from gram to kilogram quantities without loss of efficiency. The use of aqueous workups and the potential for solvent recycling make this route environmentally sustainable. The reduced generation of heavy metal waste and organic by-products simplifies regulatory compliance and facilitates smoother audits by environmental agencies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What are the key advantages of the catalytic oxidation method described in CN101068782A?
A: The method utilizes catalytic amounts of tungsten or ruthenium oxidants with hydrogen peroxide or ozone, significantly reducing chemical waste compared to stoichiometric oxidants. It operates at mild temperatures (50-55°C) and controlled pH (<3), ensuring high selectivity for the sulfone group without degrading the sensitive piperidine ring.
Q: How does this process improve supply chain reliability for CNS drug intermediates?
A: By minimizing synthetic steps through a direct oxidation-reduction sequence, the process reduces the dependency on multiple specialized reagents. The use of commercially available catalysts like Pd/C and common oxidants like H2O2 enhances raw material availability and simplifies logistics for large-scale production.
Q: Is this synthesis route suitable for commercial scale-up?
A: Yes, the patent demonstrates scalability through examples using kilogram quantities of starting materials. The reaction conditions, such as hydrogenation at 50 psi and oxidation in aqueous-organic biphasic systems, are compatible with standard industrial reactor setups, facilitating a smooth transition from lab to plant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(Sulfonylphenyl)piperidines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation CNS therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated oxidation-reduction chemistry described in CN101068782A can be translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-(sulfonylphenyl)piperidines meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance ensures that our clients receive materials that facilitate smooth regulatory filings and clinical trials.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development programs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can accelerate your path to market.
