Advanced Manufacturing Of Tricyclic Aminoalcohol Derivatives Via Asymmetric Reduction Technology
The pharmaceutical industry continuously seeks robust synthetic pathways for complex bioactive molecules, particularly those targeting metabolic disorders such as diabetes and obesity. Patent CN1360574A discloses a groundbreaking methodology for the preparation of tricyclic aminoalcohol derivatives, which serve as critical intermediates in the development of beta-3 adrenergic agonists. 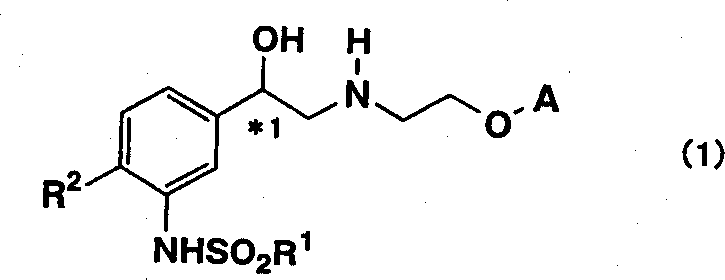 . This intellectual property represents a significant leap forward in process chemistry by addressing the longstanding inefficiencies associated with stereoselective synthesis. The disclosed technology replaces cumbersome traditional methods with a streamlined catalytic approach that ensures high optical purity while minimizing operational complexity. For global procurement teams and R&D directors, understanding the nuances of this patent is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates. The innovation lies not just in the final molecule but in the strategic design of the synthetic route that prioritizes industrial feasibility and cost-effectiveness from the very first step.
. This intellectual property represents a significant leap forward in process chemistry by addressing the longstanding inefficiencies associated with stereoselective synthesis. The disclosed technology replaces cumbersome traditional methods with a streamlined catalytic approach that ensures high optical purity while minimizing operational complexity. For global procurement teams and R&D directors, understanding the nuances of this patent is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates. The innovation lies not just in the final molecule but in the strategic design of the synthetic route that prioritizes industrial feasibility and cost-effectiveness from the very first step.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in earlier patent publications, relied heavily on the use of stoichiometric chiral auxiliaries to induce asymmetry during the reduction of ketone precursors. 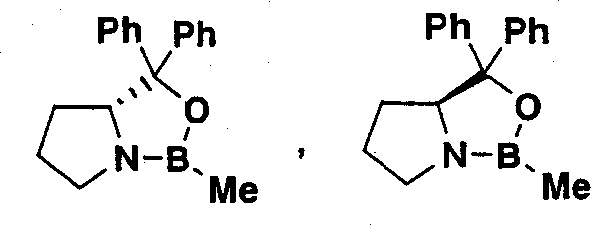 . These conventional approaches suffered from severe drawbacks including the high cost and complex preparation of the chiral reagents themselves. Furthermore, the use of borane-based reducing agents necessitated strictly anhydrous reaction conditions and rigorous temperature control to prevent safety hazards and side reactions. The post-reaction workup was equally problematic, often requiring multiple purification steps including column chromatography to remove the spent auxiliary and byproducts. This reliance on chromatographic purification is a major bottleneck in commercial manufacturing as it consumes vast quantities of solvents and silica gel, leading to substantial waste generation and extended production cycles. Consequently, the overall yield was often compromised, and the scalability of such processes was limited by safety concerns and economic inefficiency.
. These conventional approaches suffered from severe drawbacks including the high cost and complex preparation of the chiral reagents themselves. Furthermore, the use of borane-based reducing agents necessitated strictly anhydrous reaction conditions and rigorous temperature control to prevent safety hazards and side reactions. The post-reaction workup was equally problematic, often requiring multiple purification steps including column chromatography to remove the spent auxiliary and byproducts. This reliance on chromatographic purification is a major bottleneck in commercial manufacturing as it consumes vast quantities of solvents and silica gel, leading to substantial waste generation and extended production cycles. Consequently, the overall yield was often compromised, and the scalability of such processes was limited by safety concerns and economic inefficiency.
The Novel Approach
In stark contrast, the novel approach detailed in CN1360574A introduces a catalytic asymmetric reduction strategy that fundamentally transforms the production landscape. 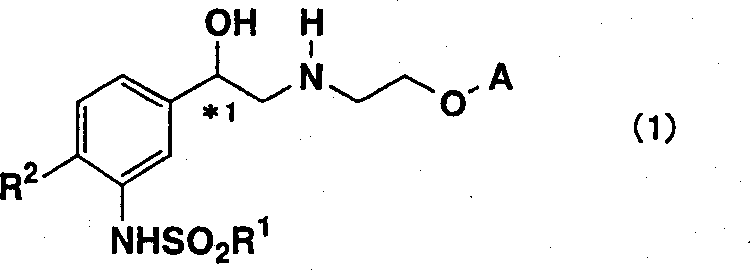 . This new methodology utilizes a transition metal catalyst, specifically a ruthenium complex, to achieve high enantioselectivity without the need for stoichiometric chiral reagents. The process begins with the chlorination of an acetophenone derivative followed by the key asymmetric reduction step to generate a chlorohydrin intermediate. A distinct advantage of this route is the physical nature of the intermediates; compounds such as the chlorohydrin and the subsequent epoxide are described as having excellent crystallinity. This property allows manufacturers to purify these critical intermediates through simple recrystallization rather than chromatography. By eliminating the need for column chromatography, the process drastically reduces solvent usage and processing time. Additionally, the optical purity of the intermediates can be further enhanced through recrystallization, ensuring that the final API meets stringent regulatory standards. This shift from stoichiometric reagents to catalytic systems, combined with crystallization-based purification, represents a paradigm shift towards greener and more economical chemical manufacturing.
. This new methodology utilizes a transition metal catalyst, specifically a ruthenium complex, to achieve high enantioselectivity without the need for stoichiometric chiral reagents. The process begins with the chlorination of an acetophenone derivative followed by the key asymmetric reduction step to generate a chlorohydrin intermediate. A distinct advantage of this route is the physical nature of the intermediates; compounds such as the chlorohydrin and the subsequent epoxide are described as having excellent crystallinity. This property allows manufacturers to purify these critical intermediates through simple recrystallization rather than chromatography. By eliminating the need for column chromatography, the process drastically reduces solvent usage and processing time. Additionally, the optical purity of the intermediates can be further enhanced through recrystallization, ensuring that the final API meets stringent regulatory standards. This shift from stoichiometric reagents to catalytic systems, combined with crystallization-based purification, represents a paradigm shift towards greener and more economical chemical manufacturing.
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Transfer Hydrogenation
The core of this technological advancement lies in the sophisticated mechanism of the asymmetric reduction step. The patent highlights the use of specific ruthenium complexes coordinated with chiral diamine ligands to facilitate the transfer of hydrogen to the prochiral ketone substrate. 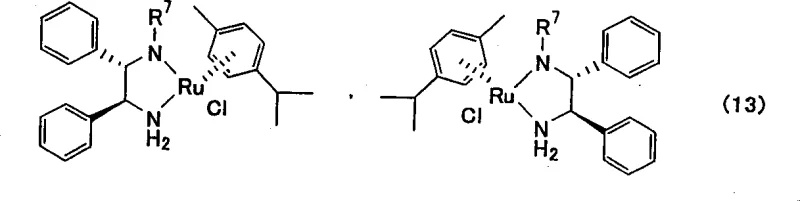 . These catalysts, often referred to as Noyori-type catalysts, operate through a concerted outer-sphere mechanism where the hydride and proton are transferred simultaneously from the metal-ligand framework to the carbonyl oxygen and carbon respectively. This mechanism is highly sensitive to the steric and electronic environment provided by the ligand, which dictates the facial selectivity of the attack and thus the configuration of the resulting alcohol. The patent specifies that catalysts represented by formula (13), containing ligands such as N-(p-toluenesulfonyl)-1,2-diphenylethylenediamine, are particularly effective. The choice of hydrogen donor is also critical, with formic acid/triethylamine azeotropes or isopropanol serving as efficient sources of hydrogen. This catalytic cycle allows for the turnover of thousands of substrate molecules per catalyst molecule, significantly lowering the cost per kilogram of product. Furthermore, the reaction can be conducted under relatively mild conditions, typically between -20°C and room temperature, which enhances safety and reduces energy consumption compared to high-pressure hydrogenation methods.
. These catalysts, often referred to as Noyori-type catalysts, operate through a concerted outer-sphere mechanism where the hydride and proton are transferred simultaneously from the metal-ligand framework to the carbonyl oxygen and carbon respectively. This mechanism is highly sensitive to the steric and electronic environment provided by the ligand, which dictates the facial selectivity of the attack and thus the configuration of the resulting alcohol. The patent specifies that catalysts represented by formula (13), containing ligands such as N-(p-toluenesulfonyl)-1,2-diphenylethylenediamine, are particularly effective. The choice of hydrogen donor is also critical, with formic acid/triethylamine azeotropes or isopropanol serving as efficient sources of hydrogen. This catalytic cycle allows for the turnover of thousands of substrate molecules per catalyst molecule, significantly lowering the cost per kilogram of product. Furthermore, the reaction can be conducted under relatively mild conditions, typically between -20°C and room temperature, which enhances safety and reduces energy consumption compared to high-pressure hydrogenation methods.
Controlling impurities is another critical aspect of the mechanistic design. The formation of the chlorohydrin intermediate is prone to racemization or elimination side reactions if not carefully managed. The patent describes specific protocols to mitigate these risks, such as maintaining low temperatures during the reduction and immediately proceeding to the epoxidation step or purifying via crystallization. The subsequent conversion of the chlorohydrin to the epoxide via base treatment is an intramolecular SN2 reaction that proceeds with inversion of configuration, preserving the stereochemical integrity established in the reduction step. This precise control over stereochemistry throughout the synthetic sequence ensures that the final tricyclic aminoalcohol derivative possesses the desired biological activity. The ability to tune the catalyst ligand structure allows chemists to optimize the enantiomeric excess (ee) for specific substrates, providing a versatile platform for synthesizing various analogues within this chemical class. Such mechanistic robustness is vital for R&D teams aiming to develop stable and reproducible manufacturing processes.
How to Synthesize Tricyclic Aminoalcohol Derivatives Efficiently
The synthesis of these high-value intermediates follows a logical progression designed to maximize yield and purity at every stage. The process initiates with the chlorination of a protected acetophenone using sulfuryl chloride in a mixture of dichloromethane and methanol, yielding an alpha-chloroketone. This intermediate is then subjected to the pivotal asymmetric reduction using the ruthenium catalyst system to afford the chiral chlorohydrin. Following isolation and purification, the chlorohydrin is treated with a base like potassium carbonate to form the corresponding epoxide. The final assembly involves the ring-opening of this epoxide with a functionalized amine, followed by global deprotection to reveal the active pharmacophore.
- Chlorinate the acetophenone precursor using sulfuryl chloride in methanol and dichloromethane to generate the alpha-chloroketone intermediate.
- Perform asymmetric reduction of the chloroketone using a ruthenium-diamine complex catalyst to yield the optically active chlorohydrin with high enantiomeric excess.
- Treat the chlorohydrin with a base such as potassium carbonate to induce intramolecular cyclization, forming the key epoxy intermediate.
- Execute a ring-opening reaction with a protected amino-alcohol followed by deprotection to finalize the tricyclic aminoalcohol derivative structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1360574A offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage is the substantial reduction in manufacturing costs driven by the elimination of expensive chiral auxiliaries and the minimization of purification steps. By replacing stoichiometric reagents with a catalytic system, the raw material cost profile is significantly optimized. Furthermore, the ability to purify intermediates via recrystallization instead of column chromatography translates to massive savings in solvent procurement and waste disposal costs. This efficiency gain allows for a more competitive pricing structure for the final pharmaceutical intermediate, enabling better margin management for downstream drug manufacturers. The simplified workflow also reduces the dependency on specialized chromatography equipment and skilled labor for complex separations, thereby lowering the barrier to entry for contract manufacturing organizations.
Supply chain reliability is another critical factor enhanced by this novel process. The intermediates generated, particularly the chlorohydrin and epoxide compounds, are described as having good crystallinity and stability. Crystalline solids are inherently easier to handle, store, and transport compared to oils or unstable liquids, reducing the risk of degradation during logistics. This stability ensures consistent quality upon delivery and extends the shelf life of inventory, allowing for more flexible production scheduling and buffer stock management. Additionally, the reduced number of reaction steps shortens the overall lead time for production batches. A shorter synthesis cycle means faster turnaround times for customer orders and increased responsiveness to market demand fluctuations. The robustness of the catalytic system also implies a lower risk of batch failure due to reagent variability, ensuring a steady and predictable supply of high-purity materials. These factors collectively contribute to a more resilient supply chain capable of withstanding disruptions.
Scalability and environmental compliance are increasingly important criteria for modern chemical procurement. The process outlined in the patent is inherently scalable due to its reliance on standard unit operations such as crystallization and filtration rather than batch-limited chromatography. The use of catalytic amounts of ruthenium reduces the heavy metal load in the waste stream, simplifying effluent treatment and aiding in meeting stringent environmental regulations. The avoidance of hazardous borane reagents further improves the safety profile of the plant, reducing insurance premiums and regulatory scrutiny. As the pharmaceutical industry moves towards greener chemistry principles, suppliers who can demonstrate such environmental stewardship gain a competitive edge. The combination of cost efficiency, supply stability, and environmental sustainability makes this synthetic route an attractive option for long-term partnerships in the production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of these tricyclic aminoalcohol derivatives. Understanding these details helps stakeholders make informed decisions about integrating this technology into their production pipelines. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation.
Q: How does the new ruthenium-catalyzed method improve upon conventional chiral auxiliary routes?
A: The novel method eliminates the need for expensive and hazardous chiral borane auxiliaries. By utilizing a recyclable ruthenium catalyst for asymmetric transfer hydrogenation, the process significantly reduces raw material costs and simplifies post-reaction workup by avoiding strict anhydrous conditions required by the old method.
Q: What are the purification advantages of the intermediates described in patent CN1360574A?
A: Key intermediates such as the chlorohydrin and epoxide compounds exhibit excellent crystallinity. This physical property allows for purification via simple recrystallization rather than labor-intensive column chromatography, drastically reducing solvent consumption and processing time while enhancing optical purity.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed for industrial efficiency. It features fewer reaction steps compared to prior art, utilizes stable crystalline intermediates that facilitate handling and storage, and employs robust catalytic systems that are amenable to scale-up from kilogram to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tricyclic Aminoalcohol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications. Our technical team has extensively analyzed the methodologies presented in CN1360574A and possesses the expertise to implement this advanced ruthenium-catalyzed route effectively. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory scale to full industrial manufacturing. Our facilities are equipped with state-of-the-art reactors capable of handling sensitive catalytic reactions under controlled atmospheres, and our rigorous QC labs ensure that every batch meets stringent purity specifications. We understand that consistency is key in pharmaceutical supply, and our quality management systems are designed to deliver products with reproducible enantiomeric excess and chemical purity.
We invite you to collaborate with us to leverage these process innovations for your specific drug development needs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how the adoption of this catalytic route can optimize your budget. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner committed to technical excellence, regulatory compliance, and long-term value creation in the pharmaceutical intermediate sector.
