Revolutionizing Cephalosporin Synthesis: High-Yield Coupling and Scalable Intermediate Production
Revolutionizing Cephalosporin Synthesis: High-Yield Coupling and Scalable Intermediate Production
The pharmaceutical landscape is constantly evolving, driven by the need for more efficient, cost-effective, and high-purity synthetic routes for critical antibiotics. Patent CN1243755C introduces a groundbreaking methodology for the preparation of 3-cyclic ether-substituted cephalosporins, specifically targeting the synthesis of advanced intermediates that serve as the backbone for next-generation antibacterial agents. This technology leverages a novel zwitterionic coupling strategy that bypasses many of the cumbersome protection and deprotection steps associated with conventional cephalosporin manufacturing. By utilizing a specific 3-tetrahydrofuran substitution pattern, the process not only enhances the stability of the beta-lactam ring but also improves the pharmacokinetic profile of the final active pharmaceutical ingredient. For R&D directors and process chemists, this patent represents a significant leap forward in optimizing the impurity profile and overall yield of complex cephem derivatives.
At the core of this innovation is the ability to directly couple a zwitterionic cephem nucleus with an activated side chain under mild aqueous-organic conditions. Unlike traditional methods that often require anhydrous environments and hazardous silylating agents, this approach utilizes a biphasic or homogeneous system involving acetone and water. The result is a robust process that delivers the target carboxylic acid or carboxylate salt in high crystalline purity. This is particularly vital for supply chain managers who require consistent quality across large batches. The patent details specific embodiments where the leaving group on the side chain precursor is optimized—such as diethyl phosphorothioate—to facilitate rapid acylation without compromising the delicate stereochemistry at the C-6 and C-7 positions of the cephem ring system.
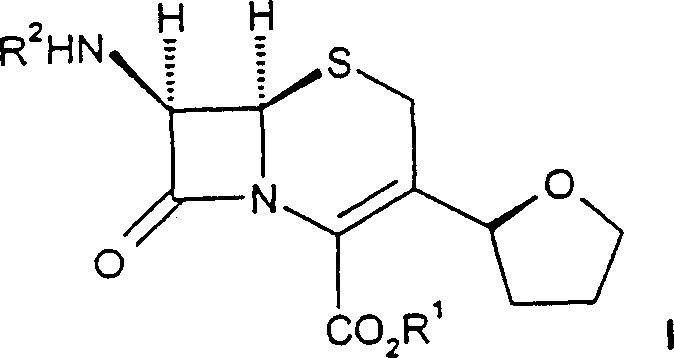
The structural versatility offered by this patent allows for the incorporation of various aminothiazole glyoxylate derivatives, enabling the production of a wide spectrum of cephalosporin analogs. The presence of the cyclic ether moiety at the 3-position is known to confer enhanced metabolic stability and improved parenteral absorption characteristics. By mastering the synthesis of these specific intermediates, manufacturers can secure a competitive edge in the global market for generic and proprietary antibiotics. The technical depth of CN1243755C provides a clear roadmap for transitioning from laboratory-scale discovery to commercial-scale production, addressing key pain points such as solvent recovery, waste minimization, and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for 3-substituted cephalosporins have long been plagued by inefficiencies that drive up costs and complicate supply chains. Conventional methods often rely on the use of silyl protecting groups to mask the carboxylic acid functionality during the coupling reaction. This necessitates additional reaction steps for silylation and subsequent desilylation, each introducing potential yield losses and impurity generation. Furthermore, these processes frequently require strictly anhydrous conditions and low temperatures, demanding specialized equipment and increasing energy consumption. The use of halogenated solvents in large volumes also poses significant environmental and regulatory challenges, complicating waste disposal and increasing the carbon footprint of the manufacturing process.
Another critical drawback of older methodologies is the difficulty in controlling stereochemistry during the acylation step. Racemization at the alpha-carbon of the side chain can lead to the formation of inactive or even toxic isomers, requiring costly and time-consuming purification steps such as preparative HPLC or repeated recrystallizations. Additionally, the isolation of the final product from conventional reaction mixtures is often problematic, leading to oils or amorphous solids that are difficult to handle and characterize. These issues collectively result in longer lead times, higher production costs, and a less reliable supply of high-quality intermediates for downstream API synthesis, creating bottlenecks for pharmaceutical companies aiming to bring products to market quickly.
The Novel Approach
The methodology disclosed in CN1243755C offers a transformative solution by utilizing zwitterionic intermediates that eliminate the need for carboxylic acid protection. This innovative approach allows for direct coupling in a mixed solvent system of acetone and water, typically in a ratio of 1.3:1, which significantly simplifies the reaction setup and workup procedure. By operating at near-neutral pH levels (pH 7-7.5) and moderate temperatures ranging from 20°C to 30°C, the process minimizes the degradation of the sensitive beta-lactam ring. The use of sodium hydroxide as a base facilitates the formation of the reactive nucleophile in situ, ensuring rapid and complete conversion of the starting materials without the need for exotic or hazardous reagents.
Moreover, this novel route promotes the formation of highly crystalline products directly from the reaction mixture upon the addition of anti-solvents like acetone. This crystalline nature is a major advantage for purification, as it allows for the effective exclusion of impurities through simple filtration and washing, rather than complex chromatographic separations. The patent highlights that this method can achieve yields of approximately 80% with exceptional enantiomeric purity, exceeding 96% e.e. in preferred embodiments. Such high fidelity in stereochemical control ensures that the resulting intermediates meet the stringent quality standards required for regulatory approval. This streamlined process not only reduces the number of unit operations but also enhances the overall safety and sustainability of the manufacturing workflow.
Mechanistic Insights into Zwitterionic Coupling and Side Chain Activation
The success of this synthetic strategy hinges on the unique reactivity of the zwitterionic cephem nucleus (Formula II). In this state, the molecule possesses both a positively charged nitrogen within the dihydrothiazine ring and a negatively charged carboxylate group. This internal salt structure enhances the solubility of the cephem in polar media while maintaining sufficient nucleophilicity at the 7-amino position to attack the activated side chain. The reaction mechanism involves the nucleophilic displacement of a leaving group (L) on the side chain precursor (Formula III) by the 7-amino group of the zwitterion. The choice of leaving group is critical; the patent identifies diethyl phosphorothioate and activated esters as particularly effective, providing a balance between reactivity and stability that prevents premature hydrolysis of the side chain activator.
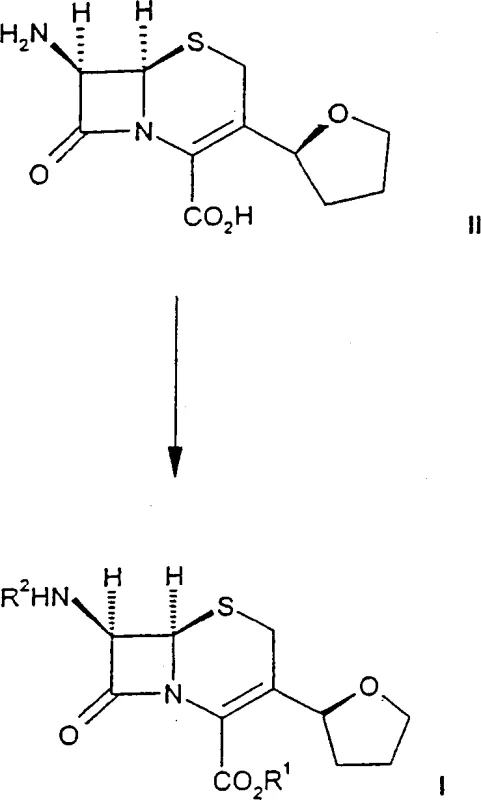
Stereochemical integrity is preserved through the use of Z-isomers of the oxime ether side chain precursors. The patent emphasizes that the OA2 group in Formula III should preferably be in the cis position relative to the amide bond. This geometric configuration is essential for the biological activity of the final cephalosporin. The mild reaction conditions prevent the isomerization of the Z-oxime to the less active E-isomer, a common side reaction in harsher acidic or basic environments. Furthermore, the solvent system plays a dual role: acetone acts as an organic co-solvent to dissolve the organic reactants, while water facilitates the ionization of the zwitterion and the base. This synergistic effect creates an optimal microenvironment for the coupling reaction to proceed with high specificity and minimal byproduct formation.
Impurity control is inherently built into the mechanism of this process. By avoiding the use of silylating agents, the formation of silyl-related byproducts is completely eliminated. Additionally, the precipitation step serves as a powerful purification tool. As the product crystallizes out of the acetone-water mixture, soluble impurities such as unreacted side chain acids and inorganic salts remain in the mother liquor. This phenomenon, known as crystallization-induced diastereomeric purification, further enhances the optical purity of the isolated solid. The result is an intermediate that requires minimal downstream processing before being converted into the final active pharmaceutical ingredient, thereby reducing the overall cost of goods and accelerating the time to market for new antibiotic formulations.
How to Synthesize 3-Cyclic Ether Cephalosporin Intermediates Efficiently
The practical implementation of this patented technology involves a sequence of well-defined steps that can be readily adapted for industrial scale-up. The process begins with the preparation of the key zwitterionic intermediate, which can be generated from protected precursors via mild deprotection methods. Following this, the coupling reaction is executed under controlled pH and temperature conditions to maximize yield and purity. The detailed标准化 synthesis steps provided below outline the specific reagents, stoichiometry, and operational parameters required to reproduce the high-quality results described in the patent documentation. Adhering to these guidelines ensures consistency and reliability in production.
- Prepare the zwitterionic cephem core (Formula II) via deprotection of the p-nitrobenzyl ester using sodium dithionite in acetone-water.
- Activate the side chain acid (Formula III) using diethyl phosphorothioate or mixed anhydride methods in situ.
- Couple the activated side chain with the zwitterion in a 1.3: 1 acetone-water mixture at 20-30°C, adjusting pH to 7-7.5 with sodium hydroxide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1243755C offers substantial strategic benefits beyond mere technical superiority. The simplification of the synthetic route translates directly into cost reduction in cephalosporin intermediates manufacturing. By eliminating multiple protection and deprotection steps, the process reduces the consumption of raw materials, solvents, and utilities. The ability to use water as a primary solvent component significantly lowers solvent procurement costs and simplifies waste treatment protocols. Furthermore, the high yield and crystalline nature of the product reduce the need for extensive purification, leading to shorter cycle times and increased throughput per reactor volume. These efficiencies cumulatively contribute to a more competitive pricing structure for the final intermediates.
- Cost Reduction in Manufacturing: The elimination of silylation reagents and the reduction in solvent usage drastically lower the variable costs associated with production. The process operates at ambient temperatures, removing the need for energy-intensive cooling or heating systems. Additionally, the high recovery rates achieved through efficient precipitation minimize material loss, ensuring that a greater proportion of input raw materials are converted into saleable product. This lean manufacturing approach allows for significant margin improvement and provides flexibility in pricing strategies for long-term supply contracts.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures consistent supply continuity. The reagents used, such as acetone, sodium hydroxide, and standard side chain acids, are commodity chemicals with stable global availability. This reduces the risk of supply disruptions caused by the scarcity of specialized or hazardous reagents. Moreover, the stability of the intermediates produced allows for safer storage and transportation, mitigating the risks associated with the degradation of sensitive pharmaceutical materials during logistics. This reliability is crucial for maintaining uninterrupted API production schedules.
- Scalability and Environmental Compliance: The methodology is inherently scalable, having been demonstrated effectively from gram to kilogram scales in the patent examples. The use of aqueous systems aligns with green chemistry principles, reducing the environmental impact of manufacturing operations. This compliance with environmental regulations facilitates smoother permitting processes and enhances the corporate social responsibility profile of the manufacturing entity. The ability to scale up without significant process re-engineering ensures that supply can be rapidly ramped up to meet surging market demand for critical antibiotics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cephalosporin synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a reliable foundation for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the key advantages of the zwitterionic coupling method described in CN1243755C?
A: The zwitterionic coupling method eliminates the need for silylation protection steps often required in traditional cephalosporin synthesis. This results in a more direct route with fewer unit operations, significantly reducing solvent consumption and processing time while maintaining high stereochemical integrity at the C-7 position.
Q: How does this process ensure high enantiomeric purity?
A: The process utilizes highly purified starting materials and mild reaction conditions (20-30°C) that minimize racemization risks. The patent specifies that the resulting compounds achieve an enantiomeric or diastereomeric purity of 96% to 100%, ensuring consistent biological activity and regulatory compliance for downstream API manufacturing.
Q: Is this methodology scalable for commercial production?
A: Yes, the methodology is designed for scalability. It employs common industrial solvents like acetone and water, avoids cryogenic temperatures, and utilizes robust precipitation techniques for isolation. These factors make it highly suitable for large-scale manufacturing ranging from pilot batches to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyclic Ether Cephalosporin Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antibiotics. Our team of expert chemists has extensively analyzed the technology disclosed in CN1243755C and possesses the capability to implement these advanced coupling methods with precision. We offer comprehensive CDMO services that cover the entire lifecycle of your project, from process optimization to commercial manufacturing. Our facilities are equipped to handle complex synthetic pathways, ensuring that we can deliver extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to meeting stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your antibiotic development programs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to innovation and excellence. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your next project. Let us help you optimize your supply chain and accelerate your path to market with superior cephalosporin intermediates.
