Advanced Selective Synthesis of Z-Isomer 3-(2-Substituted-Vinyl) Cephalosporins for Commercial Scale
Introduction to Stereoselective Cephalosporin Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for synthesizing beta-lactam antibiotics with precise stereochemical control, particularly for third-generation cephalosporins where biological activity is strictly dependent on geometric configuration. Patent CN1107679C discloses a groundbreaking selective production method for the Z-isomer of 3-(2-substituted-vinyl) cephalosporins, addressing a long-standing challenge in antibiotic synthesis. The core innovation lies in the manipulation of reaction media and thermal conditions during the Wittig olefination step, which is critical for installing the vinyl side chain at the 3-position of the cephem nucleus. This technology enables the direct generation of the therapeutically active cis-isomer, exemplified by structures such as Cefditoren, with exceptional purity levels that bypass traditional purification bottlenecks.
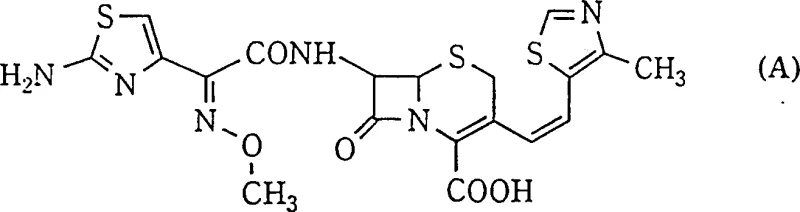
For R&D directors and process chemists, the significance of this patent extends beyond mere yield improvements; it represents a fundamental shift in how stereoselectivity is managed in complex heterocyclic systems. The Z-configuration, where the cephem ring and the thiazole substituent are arranged cis across the double bond, is essential for optimal antibacterial potency against Gram-negative bacteria. Conventional methods often struggle to differentiate between the Z and E isomers during synthesis, leading to mixtures that are notoriously difficult to separate due to similar physicochemical properties. By leveraging the specific solvent interactions and kinetic controls detailed in this intellectual property, manufacturers can secure a reliable cephalosporin intermediate supplier pathway that guarantees high optical purity from the reaction vessel, minimizing downstream processing risks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-vinyl cephalosporins via the Wittig reaction has been plagued by poor stereoselectivity when conducted under standard ambient conditions. Prior art documents, including Japanese Patent Publication No. 3-64503 and European Patent EP0175610, describe processes where the reaction is typically performed in dichloromethane or dichloromethane-water mixtures at room temperature. Under these conventional parameters, the resulting reaction product is invariably a mixture of the desired Z-isomer and the undesired E-isomer, often in ratios as unfavorable as 4.7:1 or even lower. This lack of selectivity creates a massive burden on the purification infrastructure, as the E-isomer acts as a persistent impurity that co-elutes or co-crystallizes with the target molecule.
Furthermore, the separation of these geometric isomers is technically arduous and economically inefficient. Literature indicates that even advanced chromatographic techniques struggle to resolve the Z and E isomers effectively, often requiring multiple passes over non-ionic porous resin columns or repeated fractional crystallizations. Each additional purification step introduces potential yield losses, increases solvent waste, and extends the overall production cycle time. For procurement managers focused on cost reduction in pharmaceutical manufacturing, these inefficiencies translate directly into higher COGS (Cost of Goods Sold) and reduced throughput. The necessity to discard or recycle significant portions of the E-isomer further exacerbates the material cost, making the conventional room-temperature Wittig approach unsustainable for large-scale commercial production of high-value antibiotics.
The Novel Approach
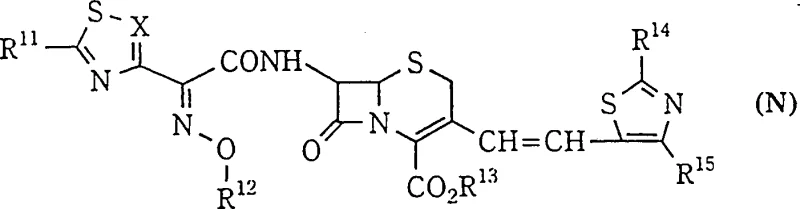
The methodology outlined in CN1107679C offers a transformative solution by redefining the reaction environment to favor the thermodynamic or kinetic formation of the Z-isomer exclusively. The inventors discovered that by employing a mixed solvent system composed of a chlorinated hydrocarbon and a lower alkanol in a specific volume ratio ranging from 1:3 to 1:0.25, coupled with a reaction temperature maintained at +5°C or lower, the selectivity profile shifts dramatically. Ideally, operating within the range of -10°C to -30°C, and most preferably between -18°C and -23°C, suppresses the formation of the E-isomer to trace levels. Experimental data within the patent demonstrates Z:E ratios exceeding 45:1 under these optimized conditions, a stark contrast to the near-equimolar mixtures obtained at room temperature.
This novel approach eliminates the dependency on complex chromatographic separations, allowing the high-purity Z-isomer to be isolated directly via crystallization from the concentrated reaction mixture. The addition of methanol or butyl acetate post-reaction facilitates the precipitation of the target compound while leaving the minute quantities of E-isomer in the mother liquor. For supply chain heads concerned with the commercial scale-up of complex beta-lactams, this simplification is invaluable. It reduces the number of unit operations, minimizes equipment occupancy time, and ensures a consistent supply of high-purity API intermediates. The ability to tune the solvent ratio, specifically favoring mixtures like chloroform and n-propanol at 1:0.4, provides a robust and reproducible protocol that can be easily transferred from pilot plant to multi-ton manufacturing scales without loss of fidelity.
Mechanistic Insights into Low-Temperature Stereoselective Wittig Olefination
The enhanced stereoselectivity observed in this process can be attributed to the intricate interplay between solvent polarity, hydrogen bonding capabilities of the alkanol, and the reduced thermal energy available for isomerization. In the Wittig reaction mechanism involving the phosphoranyl ylide derived from the 3-cephem nucleus, the transition state leading to the Z-alkene is stabilized by the specific solvation shell provided by the lower alkanol component. At sub-zero temperatures, the kinetic pathway favoring the cis-arrangement of the bulky cephem ring and the thiazole moiety becomes dominant. The reduced thermal agitation prevents the equilibration towards the more thermodynamically stable E-isomer, which is often favored at higher temperatures due to steric relief. This kinetic trapping of the Z-configuration is crucial for maintaining the structural integrity required for biological activity.
Moreover, the choice of solvent components plays a pivotal role in modulating the reactivity of the ylide intermediate. The chlorinated hydrocarbon ensures adequate solubility of the lipophilic cephem substrate, while the lower alkanol, such as n-propanol, likely participates in stabilizing the oxaphosphetane intermediate through hydrogen bonding interactions. This stabilization lowers the activation energy for the Z-selective pathway. From an impurity control perspective, this mechanism inherently limits the generation of the E-isomer at the source rather than attempting to remove it post-formation. This "prevention over cure" strategy is highly valued by quality assurance teams, as it reduces the risk of genotoxic impurities or hard-to-remove structural analogs persisting in the final drug substance. The result is a cleaner reaction profile that aligns perfectly with stringent regulatory requirements for antibiotic manufacturing.
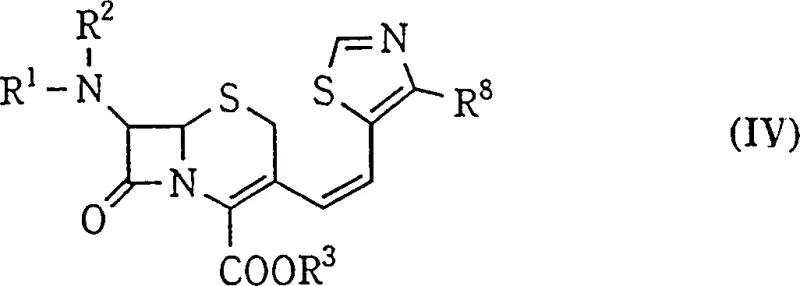
How to Synthesize Z-Isomer Cephalosporin Intermediates Efficiently
Implementing this selective synthesis route requires precise control over reaction parameters, starting from the preparation of the phosphonium salt precursor to the final crystallization step. The process begins with the conversion of a 3-halomethyl-3-cephem compound into the corresponding phosphoranyl ylide, typically using triphenylphosphine and a base in a biphasic system. Once the ylide is generated, the critical phase involves the careful adjustment of the solvent composition and temperature before the introduction of the aldehyde reactant. Maintaining the thermal window between -10°C and -30°C throughout the addition and stirring period is non-negotiable for achieving the reported high selectivity ratios. Detailed standardized synthetic steps see the guide below.
- Prepare the phosphoranyl ylide precursor by reacting a 3-halomethyl-3-cephem compound with triphenylphosphine and a base in a biphasic system, followed by separation of the organic layer containing the ylide.
- Cool the ylide solution to between -5°C and -50°C in a mixed solvent of chlorinated hydrocarbon and lower alkanol (volume ratio 1: 3 to 1:0.25), then add the thiazole-5-carbaldehyde reactant.
- Maintain the low reaction temperature for 12-20 hours to ensure high Z-isomer selectivity, followed by washing, concentration, and crystallization from methanol or butyl acetate to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this low-temperature Wittig protocol offers substantial operational benefits that extend well beyond the laboratory bench. The primary advantage lies in the drastic simplification of the downstream processing workflow. By achieving Z:E ratios that allow for direct crystallization, the need for expensive and time-consuming column chromatography is completely eliminated. This removal of a major purification bottleneck translates into significant cost reduction in pharmaceutical manufacturing, as it reduces solvent consumption, minimizes waste disposal costs, and lowers the capital expenditure required for separation equipment. The process efficiency gains allow for faster batch turnover, enhancing the overall agility of the production facility.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps results in a leaner manufacturing process with fewer unit operations. Without the need for large silica gel columns or extensive recycling of mother liquors to recover minor isomers, the consumption of consumables and solvents is drastically reduced. Furthermore, the higher effective yield of the desired Z-isomer means that less starting material is wasted on generating inactive byproducts. This efficiency drives down the variable cost per kilogram of the intermediate, providing a competitive pricing advantage in the global API market while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: Simplifying the synthesis route inherently reduces the risk of production delays and batch failures. Chromatography is often a source of variability and scaling challenges; removing it creates a more robust and predictable manufacturing process. The use of common, commercially available solvents like dichloromethane, chloroform, and n-propanol ensures that raw material sourcing remains stable and unaffected by niche supply constraints. This reliability is critical for meeting the rigorous delivery schedules of multinational pharmaceutical clients who depend on uninterrupted supplies of key antibiotic intermediates for their own formulation lines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor configurations capable of precise temperature control and efficient mixing. The reduction in solvent volume and the avoidance of solid adsorbents like silica gel significantly decrease the environmental footprint of the manufacturing process. Less hazardous waste is generated, simplifying compliance with increasingly strict environmental regulations regarding solvent emissions and waste disposal. This green chemistry aspect not only mitigates regulatory risk but also aligns with the sustainability goals of modern pharmaceutical companies, making the supply chain more resilient and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is low temperature critical for Z-isomer selectivity in this Wittig reaction?
A: According to patent CN1107679C, conducting the Wittig reaction at temperatures below +5°C, ideally between -10°C and -30°C, drastically suppresses the formation of the unwanted E-isomer. At room temperature, the E-isomer predominates or forms in significant quantities, requiring difficult chromatographic separation.
Q: What solvent system is required to achieve high purity without chromatography?
A: The process utilizes a specific mixed solvent system comprising a chlorinated hydrocarbon (such as dichloromethane or chloroform) and a lower alkanol (preferably n-propanol). The volume ratio must be strictly controlled between 1:3 and 1:0.25 to optimize stereoselectivity and facilitate direct crystallization.
Q: How does this method improve supply chain reliability for cephalosporin APIs?
A: By eliminating the need for complex column chromatography to separate Z and E isomers, the method simplifies the manufacturing workflow. This allows for direct crystallization of the high-purity Z-isomer, significantly reducing processing time, solvent consumption, and equipment bottlenecks associated with purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefditoren Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity in the production of life-saving cephalosporin antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate low-temperature conditions required for this Z-selective Wittig reaction are maintained with absolute precision at an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced HPLC capabilities to verify Z:E ratios and impurity profiles for every batch, guaranteeing that our clients receive intermediates that meet the highest global pharmacopoeial standards.
We invite pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology for their API pipelines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this selective route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition to a more cost-effective and reliable supply of high-purity cephalosporin intermediates.
