Advanced One-Pot Synthesis of Beta-Ketoesters for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced One-Pot Synthesis of Beta-Ketoesters for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for critical building blocks, particularly those serving as precursors for potent antibacterial agents. Patent CN1308309C discloses a groundbreaking methodology for the preparation of beta-ketoester compounds, which serve as indispensable intermediates in the synthesis of quinoline antibiotics such as Ciprofloxacin and Levofloxacin. This technology represents a paradigm shift from traditional multi-step sequences to a streamlined one-pot process, leveraging a modified Blaise reaction mechanism. By addressing the inherent inefficiencies of prior art, this innovation offers a pathway to high-purity pharmaceutical intermediates with enhanced economic viability. The strategic implementation of in-situ metal activation not only simplifies the operational workflow but also aligns with modern green chemistry principles by minimizing waste generation. For global procurement teams, understanding the technical nuances of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
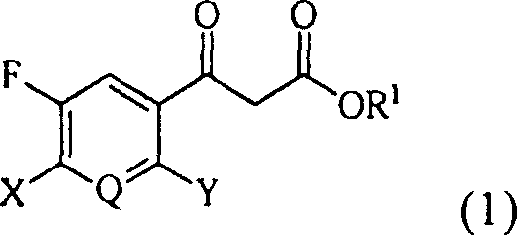
Historically, the synthesis of formula (1) beta-ketoester compounds relied on a cumbersome three-step sequence that introduced significant operational complexity and cost burdens to the manufacturing process. The conventional route typically begins with the hydrolysis of a nitrile group to a carboxylic acid, followed by conversion to an acid chloride using thionyl chloride, and finally condensation with a malonate derivative. Each of these discrete steps necessitates isolation and purification, often requiring energy-intensive distillation or chromatographic techniques that are ill-suited for large-scale production. Furthermore, the use of stoichiometric amounts of activating agents and the handling of reactive acid chloride intermediates pose substantial safety risks and environmental challenges. The cumulative yield loss across three distinct stages often results in suboptimal overall efficiency, driving up the cost of goods sold and creating bottlenecks in the supply chain for high-purity OLED material and API precursors.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a modified Blaise reaction to achieve the transformation in a single reactor vessel, thereby eliminating the need for intermediate isolation. This method employs metal zinc activated in-situ by a catalytic amount of organic acid, which facilitates the direct coupling of the nitrile substrate with an alpha-haloacetate. By collapsing the synthesis into a one-pot operation, the process drastically reduces solvent consumption, labor hours, and equipment occupancy time. The elimination of the acid chloride formation step removes the requirement for hazardous thionyl chloride, enhancing the overall safety profile of the manufacturing facility. This streamlined workflow ensures cost reduction in electronic chemical manufacturing and pharmaceutical sectors by maximizing throughput while minimizing the footprint of chemical waste, making it an ideal candidate for commercial scale-up of complex polymer additives and fine chemicals.
Mechanistic Insights into In-Situ Zinc Activation and Blaise Reaction
The core innovation of this technology lies in the mechanistic modification of the classic Blaise reaction, specifically regarding the activation of the zinc metal reagent. In traditional protocols, zinc requires a pre-activation step involving acid washing, filtration, and drying, which is laborious and introduces variability in reactivity. The patented method circumvents this by introducing a catalytic quantity of organic acids, such as methanesulfonic acid, directly into the reaction mixture containing zinc dust and solvent. This induces immediate surface activation of the zinc, generating the reactive organozinc species necessary for nucleophilic attack on the nitrile group without the need for separate preprocessing. This in-situ activation ensures consistent reproducibility batch-to-batch, a critical factor for R&D directors focusing on purity and impurity profiles. The reaction proceeds through the formation of an enaminoester intermediate, which is subsequently hydrolyzed under acidic conditions to yield the target beta-ketoester structure with high regioselectivity.
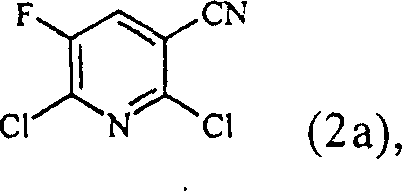
Impurity control is inherently superior in this one-pot system due to the reduced number of unit operations and the minimization of side reactions associated with intermediate handling. The use of equimolar to twofold excess of zinc, compared to the five to fifteenfold excess required in older methods, significantly lowers the load of metal residues in the crude product. This reduction in metal content simplifies the downstream purification process, as there is less unreacted zinc to decompose during the acid quench, thereby reducing the evolution of hydrogen gas. The hydrolysis step is carefully controlled at low temperatures, typically between 0°C and 10°C, to prevent degradation of the sensitive beta-ketoester moiety. Consequently, the final product can often be isolated via simple crystallization or filtration, achieving stringent purity specifications without the need for silica gel chromatography, which is a major advantage for reducing lead time for high-purity pharmaceutical intermediates.
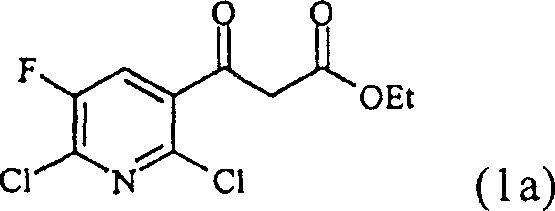
How to Synthesize Ethyl 3-(2,6-dichloro-5-fluoro-3-pyridyl)-3-oxopropionate Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety while maintaining operational simplicity. The process begins with the suspension of zinc powder in a suitable solvent such as tetrahydrofuran, followed by the addition of the catalytic organic acid activator under reflux conditions. Once the zinc is activated, the nitrile starting material is introduced, followed by the controlled dropwise addition of the alpha-haloacetate reagent over a period of one to two hours to manage exothermicity. After the coupling reaction is complete, the mixture is cooled, and aqueous acid is added to hydrolyze the intermediate, followed by a controlled warming period to ensure complete conversion. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, temperature profiles, and workup procedures validated in the patent examples to ensure successful technology transfer.
- Activate zinc powder in solvent with catalytic organic acid under reflux.
- Add nitrile compound followed by dropwise addition of alkyl alpha-haloacetate.
- Hydrolyze the intermediate enaminoester with aqueous acid and purify via filtration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers transformative benefits for procurement managers and supply chain heads tasked with optimizing costs and ensuring continuity. The consolidation of three chemical steps into a single one-pot reaction fundamentally alters the cost structure by reducing raw material consumption, utility usage, and waste disposal fees. The elimination of column chromatography, a technique that is notoriously difficult to scale and expensive to operate, allows for the use of standard filtration equipment, thereby lowering capital expenditure requirements for production facilities. Furthermore, the reduced usage of zinc metal and alpha-haloacetates translates directly into substantial cost savings on raw material procurement, as these reagents represent a significant portion of the variable costs in the conventional route. These efficiencies collectively contribute to a more resilient supply chain capable of withstanding market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The primary driver of cost efficiency in this process is the dramatic reduction in reagent stoichiometry and the removal of intermediate isolation steps. By lowering the zinc requirement from a large excess to near-equimolar amounts, the process minimizes the cost of metal reagents and the associated costs of disposing of zinc salts. Additionally, avoiding the use of thionyl chloride and the subsequent neutralization of acidic by-products reduces the consumption of auxiliary chemicals and scrubbing resources. The ability to purify the final product through crystallization rather than chromatography eliminates the high costs associated with silica gel and large volumes of eluent solvents. These factors combine to create a manufacturing process that is significantly more economical than legacy methods, offering competitive pricing for downstream customers.
- Enhanced Supply Chain Reliability: Operational simplicity is a key determinant of supply chain reliability, and this one-pot method excels by reducing the number of potential failure points in the production line. Fewer unit operations mean less equipment is required, reducing the risk of mechanical breakdowns and maintenance downtime that can disrupt delivery schedules. The robustness of the in-situ activation method ensures consistent reaction performance even with variations in raw material quality, providing a stable output of high-purity intermediates. This reliability allows suppliers to offer shorter lead times and more accurate delivery forecasts, which is critical for pharmaceutical clients managing tight production schedules for active ingredients. Consequently, partners can rely on a steady flow of materials without the volatility often associated with complex multi-step syntheses.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently designed for large-scale application by avoiding techniques that do not translate well from lab to plant. The absence of chromatographic purification removes a major bottleneck to scaling, allowing production volumes to be increased simply by enlarging reactor size without changing the fundamental workflow. Moreover, the reduced generation of chemical waste and hazardous gases aligns with increasingly stringent environmental regulations, minimizing the regulatory burden on manufacturing sites. The lower hydrogen evolution during workup enhances plant safety, reducing insurance costs and permitting complexities. This alignment with green chemistry principles ensures long-term sustainability and compliance, safeguarding the supply chain against future regulatory shifts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-ketoester synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and feasibility of the method for potential manufacturing partners. Understanding these details is crucial for evaluating the technology's fit within existing production capabilities and supply chain strategies. The answers provided reflect the specific improvements in safety, cost, and efficiency documented in the intellectual property.
Q: How does this process improve upon conventional three-step methods?
A: This method consolidates hydrolysis, acylation, and condensation into a single one-pot reaction, eliminating the need for isolating unstable acid chloride intermediates and significantly reducing processing time.
Q: What are the safety benefits of the in-situ zinc activation?
A: By reducing the molar excess of zinc metal required from 5-15 equivalents down to 1-2 equivalents, the generation of hazardous hydrogen gas during the subsequent acid hydrolysis step is drastically minimized.
Q: Is column chromatography required for purification?
A: No, the process is designed for industrial scalability, allowing the final product to be purified through simple filtration and washing with cold ethanol, avoiding expensive and slow chromatographic separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Ketoester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN1308309C are translated into reality with precision. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for antibiotic intermediates. We are committed to delivering high-quality beta-ketoester compounds that empower our clients to accelerate their drug development timelines while maintaining cost efficiency.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments that demonstrate how we can optimize your supply chain for beta-ketoester intermediates. Let us be your trusted partner in transforming complex chemical challenges into commercial successes.
