Advanced Catalytic Hydrogenation for High-Purity Cyclopropylalanine Derivatives Manufacturing
The pharmaceutical industry continuously demands efficient pathways for synthesizing chiral amino acids, which serve as critical building blocks for potent therapeutic agents. Patent CN1568304A discloses a groundbreaking preparation method for enantiomerically enriched cyclopropylalanine derivatives, addressing the longstanding challenge of achieving high optical purity without compromising the structural integrity of the cyclopropyl ring. This technology leverages a sophisticated transition metal-catalyzed asymmetric hydrogenation strategy, utilizing substantially enantiomerically pure bis-phosphine catalysts to convert specific enamide precursors into valuable amino acid derivatives. The significance of this innovation lies in its ability to produce L-cyclopropylalanine-containing peptides, which have been identified as effective cysteine protease inhibitors for treating parasitic diseases, thereby offering a reliable pharmaceutical intermediates supplier solution for advanced drug development pipelines. 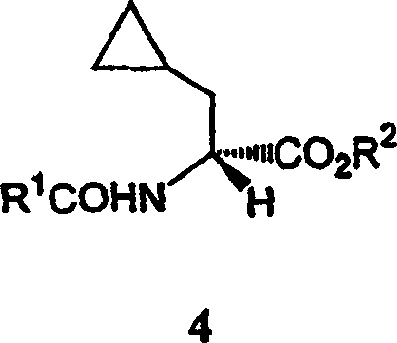
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of racemic cyclopropylalanine has been documented through various routes, such as the reaction of carbon monoxide and hydrogen with cyclopropanemethanol in the presence of cobalt catalysts, or the hydrolysis of diethyl cyclopropylmethyl(formamido)malonate. However, these traditional methods fundamentally fail to provide enantiomerically enriched products directly, necessitating cumbersome resolution steps that drastically reduce overall efficiency. Furthermore, existing chemoenzymatic syntheses starting from racemic mixtures often involve excessive synthetic operations, frequently exceeding six steps, and suffer from significant yield losses, particularly in the final deprotection stages where yields may drop to merely 50 percent. Another conventional approach involves the asymmetric alkylation of glycine amide enolates using stoichiometric amounts of expensive chiral auxiliaries like pseudoephedrine derivatives, which not only inflates raw material costs but also generates substantial chemical waste due to the requirement for auxiliary removal and recovery.
The Novel Approach
In stark contrast to these inefficient legacy processes, the novel approach detailed in the patent introduces a streamlined catalytic asymmetric hydrogenation methodology that fundamentally transforms the manufacturing landscape for these complex molecules. By employing a transition metal catalyst system containing substantially enantiomerically pure bisphosphine ligands, this method achieves high enantioselectivity using only catalytic quantities of the chiral source, rather than stoichiometric amounts. The process allows for the direct conversion of readily available enamides into the desired alpha-amino acid derivatives with exceptional control over stereochemistry. 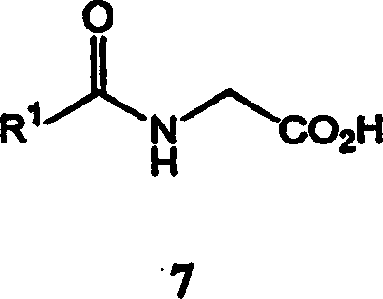
Crucially, this innovative route circumvents the common pitfall of cyclopropyl ring hydrogenolysis, a side reaction that typically plagues transition metal catalysis involving strained rings, thereby ensuring the preservation of the pharmacologically active cyclopropyl moiety throughout the synthesis.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough resides in the precise selection of the catalyst system, which typically comprises a transition metal such as rhodium, ruthenium, or iridium coordinated with a chiral bisphosphine ligand. The patent highlights the efficacy of ligands such as 1,2-bis-(2,5-dialkylphospholano)benzene (DuPHOS) and novel phosphinometallocene-aminophosphines featuring a ferrocenyl backbone. These ligands create a highly defined chiral environment around the metal center, directing the addition of hydrogen to the enamide double bond with extreme facial selectivity. The mechanism involves the formation of an active catalyst species where the metal coordinates to the olefinic bond of the enamide substrate, followed by oxidative addition of hydrogen and migratory insertion, ultimately yielding the saturated amino acid derivative with the desired configuration. 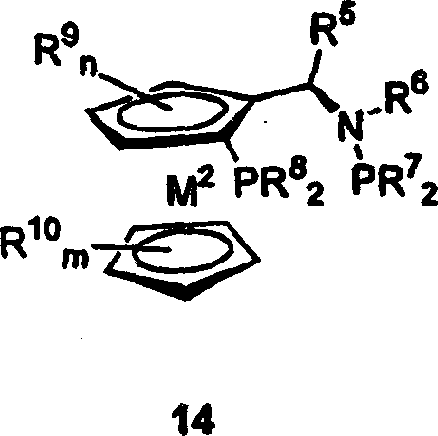
Furthermore, the impurity profile is rigorously controlled by the specificity of the catalyst; the use of these specialized ligands suppresses the thermodynamic drive towards ring-opening hydrogenolysis, which is a known vulnerability of cyclopropyl groups under reducing conditions. This mechanistic fidelity ensures that the final product maintains high chemical purity and enantiomeric excess, often exceeding 98 percent ee as demonstrated in the experimental examples, thus meeting the stringent quality requirements for high-purity pharmaceutical intermediates.
How to Synthesize Cyclopropylalanine Derivatives Efficiently
The patent outlines multiple viable synthetic routes, including a robust three-step process and a concise two-step alternative, both designed to maximize yield and operational simplicity for industrial applications. The three-step sequence begins with the condensation of cyclopropanecarboxaldehyde with an N-acylglycine to form an azlactone, followed by ring opening with an alcohol to generate the enamide, and concludes with the pivotal asymmetric hydrogenation step. Alternatively, the two-step route utilizes a Horner-Emmons reaction between cyclopropanecarboxaldehyde and a substituted phosphoryl glycine to directly access the enamide intermediate. Detailed standardized synthesis steps see the guide below.
- React cyclopropanecarboxaldehyde with N-acylglycine in the presence of carboxylic acid anhydride and base to form an azlactone intermediate.
- Contact the azlactone with an alcohol, optionally with alkali metal alkoxide, to generate the corresponding enamide ester.
- Hydrogenate the enamide under pressure using a transition metal catalyst complexed with a substantially enantiomerically pure bisphosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology presents a compelling value proposition centered on cost optimization and supply reliability. By shifting from stoichiometric chiral auxiliaries to catalytic systems, the process significantly reduces the consumption of expensive chiral reagents, leading to substantial cost savings in raw material procurement. Additionally, the elimination of multiple protection and deprotection steps inherent in older chemoenzymatic routes simplifies the manufacturing workflow, reducing labor costs and processing time while minimizing solvent usage and waste disposal expenses.
- Cost Reduction in Manufacturing: The transition to a catalytic asymmetric hydrogenation process eliminates the need for costly stoichiometric chiral auxiliaries and the associated waste streams, directly lowering the cost of goods sold. The ability to use common, commercially available starting materials like cyclopropanecarboxaldehyde and N-acylglycines further enhances economic efficiency by avoiding specialized, high-cost precursors. Moreover, the high conversion rates and selectivity observed in the examples suggest minimal need for expensive purification chromatography, allowing for more economical crystallization-based isolation methods.
- Enhanced Supply Chain Reliability: The reliance on robust chemical catalysis rather than sensitive enzymatic processes improves the resilience of the supply chain against biological variability and storage constraints. The starting materials required for this synthesis are commodity chemicals with stable global availability, reducing the risk of supply disruptions associated with niche biocatalysts or complex chiral pool resources. This stability ensures consistent lead times for high-purity pharmaceutical intermediates, enabling downstream drug manufacturers to plan their production schedules with greater confidence.
- Scalability and Environmental Compliance: The reaction conditions described, operating at moderate temperatures and hydrogen pressures compatible with standard industrial reactors, facilitate seamless commercial scale-up of complex pharmaceutical intermediates from pilot to multi-ton production. The process generates fewer byproducts and avoids the heavy metal waste often associated with stoichiometric organometallic reagents, aligning with increasingly strict environmental regulations and sustainability goals. This green chemistry profile not only reduces regulatory compliance burdens but also enhances the corporate social responsibility standing of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations regarding the implementation of this patented synthesis technology, derived directly from the detailed specifications and experimental data provided in the intellectual property documentation. These insights are intended to clarify the operational feasibility and strategic benefits for potential partners evaluating this manufacturing route.
Q: How does this process prevent the hydrogenolysis of the cyclopropyl ring?
A: The process utilizes specific transition metal catalysts, such as rhodium or ruthenium complexed with chiral bisphosphine ligands like DuPHOS or ferrocenyl aminophosphines, which facilitate asymmetric hydrogenation of the enamide double bond without cleaving the sensitive cyclopropyl C-C bonds.
Q: What are the advantages of this catalytic method over chemoenzymatic synthesis?
A: Unlike chemoenzymatic routes that often require multiple steps and suffer from lower yields in the final deprotection stage, this catalytic hydrogenation approach uses catalytic amounts of chiral reagents, offers higher atom economy, and allows for the direct production of both R and S enantiomers from common starting materials.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the reaction conditions described, including moderate temperatures and hydrogen pressures ranging from ambient to 20.7 barg, are compatible with standard industrial hydrogenation equipment, facilitating reliable commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropylalanine Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic and patented research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the catalytic hydrogenation parameters described in CN1568304A to ensure stringent purity specifications and consistent enantiomeric excess are met for every batch. With rigorous QC labs and state-of-the-art hydrogenation facilities, we are uniquely positioned to deliver high-purity pharmaceutical intermediates that meet the exacting standards of global regulatory bodies.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. Contact us today to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments that demonstrate the tangible economic and technical advantages of partnering with us for your cyclopropylalanine derivative supply requirements.
