Scalable Synthesis of 19-Norcyclopropa[g]taxanes: A Technical Breakthrough for API Manufacturing
Scalable Synthesis of 19-Norcyclopropa[g]taxanes: A Technical Breakthrough for API Manufacturing
The pharmaceutical industry's relentless pursuit of more efficient routes to complex oncology therapeutics has brought significant attention to the synthesis of taxane intermediates, specifically the 19-norcyclopropa[g]taxane scaffold which serves as a critical precursor for next-generation antimitotic agents. Patent CN1774430A discloses a highly innovative methodology for the preparation of 4,10β-diacetoxy-2α-benzoyloxy-5β,20-epoxy-1,13α-dihydroxy-9-oxo-19-norcyclopropa[g]tax-11-ene, utilizing a unique combination of sulfolane solvent and molecular sieves to drive the critical 7,8-cyclopropanation step. This technical advancement represents a paradigm shift from traditional methods that relied on hazardous alkali metal azides or halides, offering a cleaner, safer, and more controllable reaction profile that is exceptionally well-suited for industrial scale-up. By leveraging the specific solvation properties of sulfolane and the mild basicity of molecular sieves, this process effectively mitigates the formation of persistent vinyl impurities that have historically plagued the purification of these high-value intermediates. For R&D directors and process chemists, understanding the nuances of this solvent system is crucial for optimizing yield and purity in the manufacturing of complex taxane derivatives.
![General structure of the target 19-norcyclopropa[g]taxane intermediate (Formula I)](/insights/img/taxane-intermediate-sulfolane-cyclopropanation-pharma-supplier-20260306120047-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to constructing the 7,8-cyclopropane ring in taxane skeletons, such as those described in earlier patent literature like EP 0673 372, typically necessitated the use of strong nucleophiles or harsh basic conditions involving alkali metal halides such as sodium iodide or potassium fluoride, and occasionally alkali metal azides like sodium azide. These conventional methodologies presented substantial operational challenges, including the generation of toxic waste streams, the requirement for rigorous safety protocols to handle explosive azide species, and significant difficulties in downstream processing due to the emulsification caused by inorganic salts. Furthermore, the use of solvents like acetonitrile or tetrahydrofuran mixtures often resulted in incomplete conversion or the formation of stubborn elimination byproducts, specifically vinyl derivatives, which possess polarity profiles nearly identical to the desired cyclopropane product, thereby necessitating multiple, yield-eroding chromatographic purification steps. The reliance on silica gel in massive excess, as noted in some academic literature, further compounded the economic and environmental burden, rendering such processes impractical for commercial-scale API manufacturing where cost efficiency and throughput are paramount concerns for supply chain stability.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in CN1774430A employs sulfolane (tetramethylene sulfone) as the reaction medium in conjunction with activated 4Å molecular sieves, creating a uniquely favorable environment for the intramolecular cyclization of 7-O-triflate taxane precursors. This method eliminates the need for soluble inorganic bases or toxic azides, replacing them with a heterogeneous solid base that can be easily removed via simple filtration, thus streamlining the workup procedure and significantly reducing the inorganic load in the waste stream. The high boiling point and exceptional thermal stability of sulfolane allow the reaction to proceed at elevated temperatures (around 60°C) without solvent degradation, ensuring complete conversion of the starting material while maintaining the integrity of the sensitive taxane core. Most critically, the inclusion of a controlled amount of water (2-5% by weight) within the sulfolane system introduces a selective hydrolysis mechanism that converts problematic 2-vinyl byproducts into highly polar diol derivatives, which are trivially separated from the target molecule during crystallization, thereby dramatically enhancing the overall purity profile and reducing the reliance on expensive preparative HPLC or flash chromatography.
Mechanistic Insights into Sulfolane-Mediated Cyclopropanation
The core chemical transformation involves an intramolecular nucleophilic substitution where the C-7 oxygen, activated as a trifluoromethanesulfonate (triflate) leaving group, undergoes displacement by the proximal C-8 methylene group to form the strained cyclopropane ring. In this specific protocol, the 7-O-triflate precursor, depicted in Formula (II), serves as the electrophilic substrate, and the reaction is initiated by the weak basic sites present on the surface of the molecular sieves which likely assist in deprotonating or activating the transition state without causing beta-elimination side reactions that are common with stronger bases. The choice of sulfolane is mechanistically significant; as a highly polar aprotic solvent with a high dielectric constant, it effectively stabilizes the developing charge separation in the transition state of the SN2-like cyclization, lowering the activation energy barrier and facilitating the ring closure at moderate temperatures. Unlike lower boiling solvents that might require reflux conditions leading to thermal stress on the molecule, sulfolane provides a stable thermal bath that ensures the reaction kinetics favor cyclization over competing decomposition pathways, resulting in the clean formation of the 19-norcyclopropa[g]taxane skeleton shown in Formula (III).
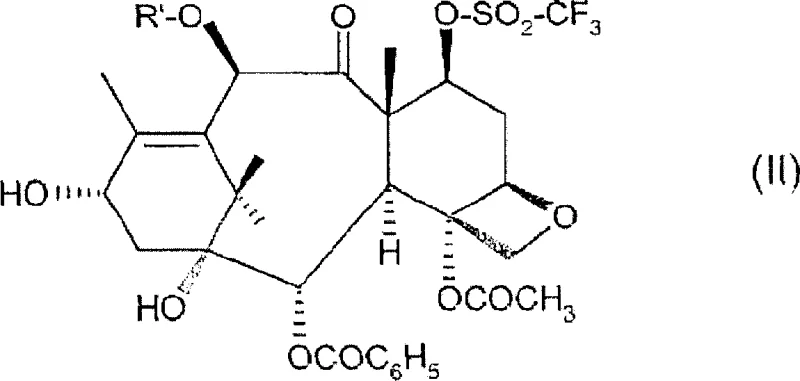
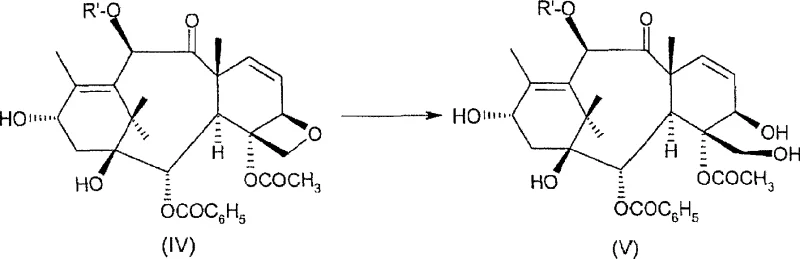
A particularly sophisticated aspect of this mechanism is the management of impurity profiles through solvent engineering, specifically the intentional introduction of water into the sulfolane matrix. During the cyclopropanation, a competing elimination reaction can generate a 2-vinyl byproduct (Formula IV), which is structurally very similar to the desired product and notoriously difficult to remove via standard chromatographic techniques. However, the presence of 2-5% water in the hot sulfolane reaction mixture promotes the hydration of this vinyl double bond, converting the recalcitrant Formula (IV) impurity into a 1,2-diol derivative (Formula V). This chemical modification drastically alters the polarity and hydrogen-bonding capability of the impurity, making it significantly more hydrophilic than the target lipophilic taxane. Consequently, during the subsequent aqueous workup and crystallization steps, the diol byproduct remains in the mother liquor or is washed away, while the desired cyclopropane product precipitates with high purity. This "impurity destruction" strategy via in-situ hydrolysis is a masterclass in process chemistry, turning a potential purification bottleneck into a non-issue through precise control of reaction conditions.
How to Synthesize 19-Norcyclopropa[g]tax-11-ene Efficiently
The implementation of this sulfolane-based cyclopropanation process requires careful attention to solvent quality and reagent activation to ensure reproducible high yields and purity. The procedure begins with the preparation of the reaction mixture by suspending activated 4Å molecular sieves in sulfolane that has been spiked with a precise quantity of water, typically aiming for a 4% weight ratio to optimize the hydrolysis of vinyl impurities without compromising the stability of the triflate starting material. The 7-O-triflate taxane derivative is then added to this suspension, and the mixture is heated to approximately 60°C, a temperature that balances reaction rate with thermal safety, and maintained for several hours until HPLC analysis confirms complete consumption of the starting material. Following the reaction, the mixture is cooled, and the solid molecular sieves are removed via filtration through a celite pad, followed by a thorough washing of the cake with ethyl acetate to recover any adsorbed product. The combined organic filtrate is then subjected to an aqueous workup to remove residual sulfolane and polar byproducts, followed by concentration and crystallization from solvent systems such as ethyl acetate and diisopropyl ether to yield the high-purity intermediate. For a detailed, step-by-step breakdown of the exact quantities, timing, and purification parameters described in the patent examples, please refer to the standardized synthesis guide below.
- React the 7-O-triflate precursor with activated 4Å molecular sieves in sulfolane solvent containing 2-5% water at 60°C.
- Filter the reaction mixture to remove molecular sieves and concentrate the organic phase under reduced pressure.
- Purify the crude product via crystallization using ethyl acetate/diisopropyl ether mixtures or silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this sulfolane-mediated process offers compelling advantages that directly address the pain points of cost volatility and supply continuity in the production of complex pharmaceutical intermediates. By eliminating the need for stoichiometric amounts of alkali metal azides or halides, manufacturers can significantly reduce the costs associated with hazardous material handling, specialized waste disposal, and regulatory compliance, leading to a leaner and more cost-effective production model. The robustness of the reaction in sulfolane, which tolerates a range of temperatures and water contents without catastrophic failure, enhances process reliability and reduces the risk of batch failures that can disrupt supply schedules. Furthermore, the simplified purification workflow, driven by the water-mediated conversion of impurities, reduces the consumption of expensive silica gel and organic solvents typically required for chromatography, thereby lowering the overall cost of goods sold (COGS) and improving the environmental footprint of the manufacturing process. This aligns perfectly with the industry's shift towards greener chemistry and sustainable supply chains, making it an attractive option for long-term partnerships.
- Cost Reduction in Manufacturing: The elimination of toxic and expensive reagents like sodium azide, coupled with the removal of extensive chromatographic purification steps, results in substantial operational cost savings. The ability to use simple crystallization for purification instead of resource-intensive column chromatography drastically reduces solvent consumption and labor hours, directly impacting the bottom line. Additionally, the recovery and reuse of high-boiling sulfolane solvent is technically feasible, further enhancing the economic efficiency of the process over large production campaigns.
- Enhanced Supply Chain Reliability: The use of stable, commercially available reagents such as molecular sieves and sulfolane mitigates the risk of supply disruptions often associated with specialized or regulated chemicals. The process's tolerance to minor variations in reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of out-of-specification results that could delay shipments. This reliability is critical for maintaining the continuous flow of materials required for just-in-time API manufacturing schedules.
- Scalability and Environmental Compliance: The heterogeneous nature of the reaction using solid molecular sieves facilitates easy scale-up from laboratory to pilot and commercial scales without the heat transfer limitations often encountered with homogeneous exothermic reactions. The reduction in hazardous waste generation, particularly the avoidance of heavy metal or azide contamination, simplifies environmental permitting and waste treatment, ensuring compliance with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced taxane synthesis route, providing clarity on its practical application and benefits. These insights are derived directly from the experimental data and claims within the patent documentation, offering a realistic view of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: Why is sulfolane preferred over acetonitrile or DMF for this cyclopropanation?
A: Sulfolane provides a superior solvent environment that minimizes degradation and facilitates the conversion of difficult-to-separate vinyl byproducts into easily removable diol derivatives when controlled amounts of water are present.
Q: What is the role of molecular sieves in this reaction mechanism?
A: Molecular sieves act as a mild, solid-state base that promotes the intramolecular nucleophilic displacement of the triflate group without the harshness or toxicity associated with traditional alkali metal azides or halides.
Q: How does the addition of water improve the impurity profile?
A: Controlled water content (2-5%) hydrolyzes the 2-vinyl byproduct (Formula IV) into a 1,2-diol derivative (Formula V), which possesses significantly different polarity, allowing for easy separation from the desired cyclopropane product during purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 19-Norcyclopropa[g]tax-11-ene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated sulfolane-based cyclopropanation chemistry described in CN1774430A can be executed with precision and consistency at any volume. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of taxane intermediate meets the exacting standards required for oncology API synthesis. Our commitment to quality assurance extends beyond mere compliance; we proactively optimize process parameters to maximize yield and minimize impurities, delivering value that goes beyond the certificate of analysis.
We invite forward-thinking pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthetic technology for their pipeline projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this process can optimize your supply chain economics. We encourage you to reach out today to discuss your project needs,索取 specific COA data for our current inventory, and review our comprehensive route feasibility assessments to ensure your next clinical or commercial campaign is built on a foundation of chemical excellence and supply security.
