Advanced Manufacturing of Polycyclic Carbamoyl Pyridone Compounds for HIV Treatment
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical antiviral agents, particularly those targeting Human Immunodeficiency Virus (HIV) replication. Patent CN114394983A discloses a comprehensive suite of methodologies for the synthesis of polycyclic carbamoyl pyridone compounds, which serve as potent HIV-1 integrase inhibitors. This intellectual property represents a significant advancement in the field of antiretroviral therapy, offering multiple convergent pathways to construct the complex polycyclic core essential for biological activity. For global procurement teams and technical directors, understanding the nuances of this patent is crucial for securing a reliable supply of high-purity pharmaceutical intermediates. The disclosed methods emphasize modularity, allowing manufacturers to adapt the synthesis based on raw material availability and specific purity requirements. By leveraging these advanced synthetic strategies, stakeholders can mitigate supply chain risks associated with single-route dependencies. The technology described herein provides a foundation for producing clinical-grade materials with consistent quality attributes, addressing the stringent regulatory standards required for HIV medication manufacturing.
Furthermore, the patent details specific embodiments that optimize reaction conditions to enhance overall process efficiency. The ability to synthesize these compounds through various general routes (Scheme I through Scheme VI) offers flexibility in process development, enabling chemists to select the most cost-effective and environmentally benign pathway. This adaptability is paramount for long-term commercial viability, as it allows for continuous improvement and optimization without necessitating a complete overhaul of the manufacturing process. For organizations aiming to establish themselves as a reliable polycyclic carbamoyl pyridone supplier, mastering these synthetic techniques is a strategic imperative. The integration of enzymatic resolution steps further underscores the commitment to producing enantiomerically pure substances, which is a critical quality attribute for drug safety and efficacy. Consequently, this patent serves as a vital resource for driving innovation in the production of next-generation antiretroviral therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches to complex polycyclic heterocycles often suffer from low overall yields, excessive step counts, and the reliance on hazardous reagents that complicate waste management and increase operational costs. Conventional routes frequently require harsh reaction conditions, such as extreme temperatures or pressures, which can degrade sensitive intermediates and lead to the formation of difficult-to-remove impurities. Moreover, achieving high enantiomeric excess in traditional synthesis often necessitates chiral chromatography or the use of expensive chiral auxiliaries, both of which significantly impact the cost of goods sold and extend production timelines. These inefficiencies create bottlenecks in the supply chain, making it challenging to meet the growing global demand for antiviral medications. Additionally, the use of stoichiometric amounts of heavy metal catalysts in older methodologies poses environmental challenges and requires rigorous purification steps to ensure residual metal levels comply with regulatory limits. Such limitations hinder the ability to scale production efficiently and economically.
The Novel Approach
In contrast, the methodologies outlined in patent CN114394983A introduce a streamlined and versatile approach that addresses many of the shortcomings associated with conventional synthesis. By utilizing a modular strategy that builds the polycyclic core through efficient cyclization and condensation reactions, the novel approach reduces the number of isolation steps and minimizes material loss. The incorporation of enzymatic resolution techniques provides a sustainable alternative to chemical resolution, offering high selectivity under mild conditions while reducing the environmental footprint of the manufacturing process. This innovation facilitates cost reduction in pharmaceutical intermediates manufacturing by lowering reagent consumption and simplifying downstream processing. Furthermore, the patent describes routes that utilize readily available starting materials, enhancing supply chain resilience and reducing dependency on specialized precursors. The ability to produce high-purity polycyclic carbamoyl pyridone compounds through these optimized pathways ensures that manufacturers can deliver consistent quality at scale. This technological leap forward supports the commercial scale-up of complex pharmaceutical additives and intermediates, aligning with industry goals for sustainability and efficiency.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Enzymatic Resolution
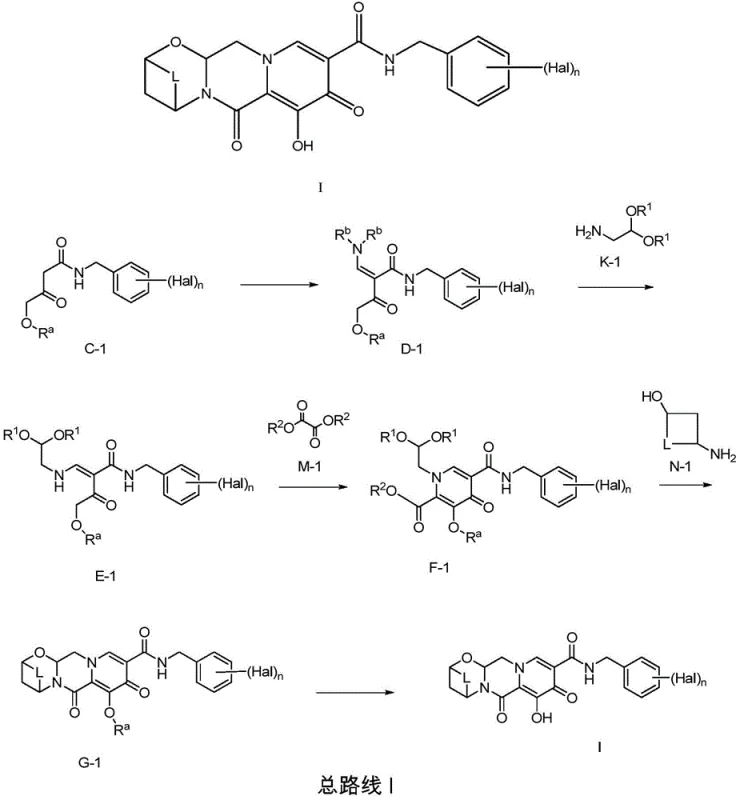
The core of the synthetic innovation lies in the precise construction of the pyridone ring system and the subsequent fusion with the polycyclic framework. The cyclization of intermediate E-1 to form F-1 is a pivotal transformation that establishes the central heterocyclic core. This reaction typically involves the use of a base, such as sodium methoxide, in an alcoholic solvent, promoting intramolecular condensation. The mechanistic pathway involves the deprotonation of an active methylene group, followed by nucleophilic attack on an ester functionality, leading to ring closure. Careful control of reaction temperature and base strength is essential to prevent over-reaction or decomposition of the sensitive enamine moiety. The patent specifies conditions that favor the formation of the desired regioisomer, ensuring that the resulting scaffold possesses the correct substitution pattern for subsequent biological activity. This level of mechanistic control is critical for maintaining batch-to-batch consistency and meeting stringent purity specifications required for clinical applications.
Another critical aspect of the process is the establishment of chirality, which is achieved through enzymatic resolution of racemic intermediates. The patent details the use of lipases, such as Novozyme 435 or CAL-B, to selectively acylate or hydrolyze one enantiomer of a racemic mixture. This biocatalytic approach exploits the stereospecificity of enzymes to differentiate between enantiomers, allowing for the isolation of the desired optical isomer with high enantiomeric excess. The mechanism involves the binding of the substrate to the enzyme's active site, where specific amino acid residues facilitate the transfer of an acyl group or the hydrolysis of an ester bond in a stereo-selective manner. This method avoids the use of toxic heavy metals and harsh chemical reagents often associated with asymmetric synthesis. By integrating this green chemistry principle, the process not only enhances product purity but also aligns with modern environmental compliance standards. The resulting enantiomerically enriched intermediates are then carried forward in the synthesis, ensuring that the final active pharmaceutical ingredient meets the necessary stereochemical requirements for efficacy and safety.
How to Synthesize Polycyclic Carbamoyl Pyridone Efficiently
The synthesis of these complex molecules requires a systematic approach that integrates chemical transformation with biocatalytic precision. The process begins with the preparation of key acyclic precursors, which are then subjected to cyclization to form the core heterocyclic structure. Subsequent functionalization steps introduce the necessary side chains and stereochemical features. Detailed standardized synthetic steps are provided in the guide below to ensure reproducibility and quality control across different manufacturing sites. Adhering to these protocols is essential for achieving the high yields and purity levels demanded by the pharmaceutical industry.
- Perform acylation and amidation of Meldrum's acid to provide intermediate C-1 using appropriate catalysts and acylating reagents.
- React C-1 with alkylated formamide acetal and K-1 to form enamine E-1, followed by cyclization with M-1 to generate F-1.
- Condense F-1 with N-1 to form G-1, followed by deprotection to yield the final polycyclic carbamoyl pyridone compound of Formula I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the synthetic routes described in this patent offers substantial strategic benefits beyond mere technical feasibility. The streamlined nature of the process directly translates to enhanced operational efficiency, reducing the time and resources required to bring materials from bench scale to commercial production. By minimizing the number of unit operations and utilizing robust reaction conditions, manufacturers can achieve significant cost savings in manufacturing without compromising on quality. The reliance on commercially available starting materials mitigates the risk of raw material shortages, ensuring a stable and continuous supply of critical intermediates. This reliability is crucial for maintaining uninterrupted production schedules for life-saving antiretroviral therapies. Furthermore, the elimination of complex purification steps reduces solvent consumption and waste generation, contributing to a more sustainable manufacturing footprint.
- Cost Reduction in Manufacturing: The implementation of enzymatic resolution and efficient cyclization steps eliminates the need for expensive chiral columns and reduces the consumption of stoichiometric reagents. This qualitative shift in process design leads to substantial cost savings by lowering raw material expenses and reducing waste disposal costs. The simplified workflow also decreases labor hours and energy consumption associated with extended reaction times and multiple isolation steps. Consequently, the overall cost of goods is optimized, allowing for more competitive pricing in the global market. These efficiencies are achieved through intelligent process design rather than compromising on quality, ensuring that cost reductions are sustainable and long-term.
- Enhanced Supply Chain Reliability: The versatility of the disclosed synthetic routes allows for the substitution of certain reagents or solvents based on regional availability, reducing dependency on single-source suppliers. This flexibility enhances supply chain resilience, protecting against disruptions caused by geopolitical events or logistical bottlenecks. The use of stable intermediates that can be stored for extended periods further buffers the supply chain against demand fluctuations. By establishing a robust manufacturing network based on these adaptable protocols, organizations can guarantee reducing lead time for high-purity pharmaceutical intermediates and ensure timely delivery to customers. This reliability fosters stronger partnerships with downstream pharmaceutical companies and strengthens market position.
- Scalability and Environmental Compliance: The processes described are inherently scalable, having been designed with commercial production in mind from the outset. The avoidance of hazardous reagents and the use of greener biocatalytic steps align with increasingly strict environmental regulations worldwide. This compliance reduces the regulatory burden and potential liabilities associated with hazardous waste management. The ability to scale up without significant process re-engineering accelerates time-to-market for new drug formulations. Additionally, the reduced environmental impact enhances the corporate social responsibility profile of the manufacturer, appealing to eco-conscious stakeholders and investors. This combination of scalability and sustainability positions the technology as a leader in modern pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of these critical intermediates. The answers are derived directly from the technical specifications and embodiments detailed within the patent documentation. Understanding these aspects helps stakeholders make informed decisions regarding procurement and process adoption. The information provided reflects the current state of the art in synthetic methodology for this class of compounds.
Q: What are the key advantages of the enzymatic resolution step in this synthesis?
A: The enzymatic resolution step utilizing lipases such as Novozyme 435 allows for the production of enantiomerically enriched intermediates with high optical purity, avoiding the need for complex chiral chromatography and reducing waste associated with traditional resolution methods.
Q: How does this patent address the scalability of HIV integrase inhibitor intermediates?
A: The patent outlines multiple general routes (I-VI) that utilize commercially available starting materials and standard reaction conditions, facilitating scale-up from laboratory to commercial production without requiring exotic reagents or extreme pressure conditions.
Q: What impurity control mechanisms are embedded in the cyclization steps?
A: The cyclization of E-1 to F-1 is controlled through precise temperature management and base selection, minimizing side reactions and ensuring the formation of the desired pyridone core with high structural integrity and minimal byproduct formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polycyclic Carbamoyl Pyridone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of advanced pharmaceutical intermediates for the global fight against HIV. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch. Our facility is equipped to handle complex synthetic challenges, including the enzymatic resolution and cyclization steps outlined in patent CN114394983A. By partnering with us, you gain access to a supply chain that prioritizes quality, reliability, and regulatory compliance above all else.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our capabilities align with your project goals. Request a Customized Cost-Saving Analysis to understand how our optimized processes can benefit your bottom line. We are ready to provide specific COA data and route feasibility assessments to support your development timeline. Let us be your trusted partner in bringing life-saving medications to patients worldwide through superior manufacturing excellence and unwavering commitment to quality.
