Advanced Melatonin Derivative Synthesis for Commercial Pharmaceutical Production
The pharmaceutical and nutraceutical industries have long recognized the therapeutic potential of melatonin, yet its commercial application has been historically constrained by severe physicochemical limitations. Patent CN109627202B introduces a groundbreaking chemical modification strategy that fundamentally alters the solubility profile of this critical hormone. By synthesizing specific melatonin derivatives through a controlled N-alkylation process, the technology achieves a water solubility increase of nearly 700 times compared to the natural compound. This enhancement is not merely a laboratory curiosity but a pivotal advancement for formulators seeking to eliminate toxic organic solvents from final drug products. For R&D Directors and Procurement Managers, this patent represents a viable pathway to higher purity standards and reduced regulatory hurdles associated with solvent residues. The ability to process these molecules in aqueous media opens new avenues for intravenous formulations and rapid-absorption delivery systems that were previously unfeasible with native melatonin.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional handling of natural melatonin in industrial settings is plagued by its inherent lipophilicity and extremely poor water solubility, which necessitates the use of hazardous organic solvents during formulation. This reliance on solvents like ethanol or propylene glycol introduces significant toxicity risks and complicates the purification process, often leaving residual impurities that fail stringent pharmacopeial standards. Furthermore, the low solubility limits the bioavailability and retention time of the active ingredient in the bloodstream, reducing the overall therapeutic efficacy and requiring higher dosages to achieve desired clinical outcomes. From a supply chain perspective, the need for specialized solvent handling and recovery systems increases operational costs and environmental compliance burdens significantly. Manufacturers are frequently forced to compromise on formulation elegance or face costly reformulation efforts to meet safety regulations regarding solvent limits in pediatric and geriatric medications.
The Novel Approach
The innovative methodology outlined in the patent data overcomes these barriers by chemically modifying the indole nitrogen atom to introduce highly hydrophilic functional groups such as sulfonates or carboxylates. This structural transformation allows the derivative to dissolve readily in aqueous environments, thereby eliminating the need for toxic organic co-solvents in the final drug product. The process utilizes readily available reagents like sodium hydride and sultones under mild temperature conditions, ensuring that the reaction is both scalable and safe for commercial production. By shifting the processing medium to water, manufacturers can drastically simplify downstream purification steps and reduce the environmental footprint associated with solvent waste disposal. This approach not only enhances the safety profile of the final medication but also improves the pharmacokinetic properties, ensuring longer retention in the blood and more consistent therapeutic effects for patients.
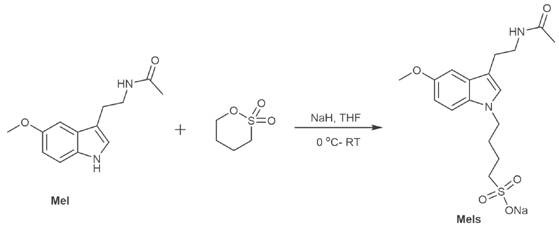
Mechanistic Insights into NaH-Catalyzed N-Alkylation
The core of this synthesis relies on a precise deprotonation mechanism where sodium hydride acts as a strong base to generate a nucleophilic indole anion from the melatonin starting material. This reactive intermediate then undergoes a nucleophilic attack on the electrophilic carbon of the sultone or bromo-ester reactant, forming a stable covalent bond at the nitrogen position. The reaction conditions are meticulously controlled, starting at cryogenic temperatures of -2°C to prevent side reactions and then warming to 20-30°C to drive the conversion to completion over an extended period. This temperature gradient is critical for managing the exothermic nature of the deprotonation and ensuring high selectivity for the N-alkylated product over potential O-alkylation byproducts. The use of thin-layer chromatography for monitoring ensures that the reaction is quenched at the optimal point, minimizing the formation of degradation products and maximizing the yield of the target derivative.
Impurity control is further enhanced by the choice of solvents and workup procedures, which are designed to separate the ionic product from neutral organic impurities effectively. The final purification via column chromatography ensures that the resulting melatonin derivative meets high-purity specifications required for pharmaceutical applications. The structural flexibility of this method allows for the introduction of various chain lengths and functional groups, enabling the tuning of physicochemical properties to match specific delivery requirements. For technical teams, understanding this mechanism is key to scaling the process while maintaining consistent quality and minimizing batch-to-batch variability. The robustness of the chemistry ensures that even at large scales, the reaction proceeds with high fidelity, providing a reliable source of high-quality intermediate for downstream formulation.
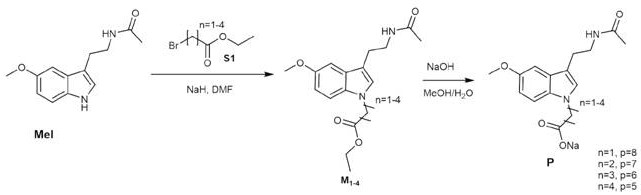
How to Synthesize Melatonin Derivative Efficiently
Implementing this synthesis route requires careful attention to reagent quality and temperature control to ensure reproducible results across different batch sizes. The standardized protocol involves dissolving the starting material in anhydrous tetrahydrofuran and adding the catalyst under an inert atmosphere to prevent moisture interference. Detailed operational parameters regarding stirring rates, addition speeds, and quenching techniques are essential for maintaining safety and product integrity during the exothermic phases. Operators must be trained to monitor the reaction progress closely using analytical methods to determine the exact endpoint for quenching. The following guide outlines the critical steps for executing this synthesis, ensuring that the final product achieves the desired solubility and purity profiles necessary for commercial success.
- Dissolve natural melatonin in anhydrous tetrahydrofuran and cool the mixture to -2°C before adding sodium hydride catalyst.
- Add the specific reactant such as 1-4 butanesultone and maintain stirring while gradually warming the reaction to 20-30°C for 8-12 hours.
- Quench the reaction with ice water, extract the aqueous phase, remove solvents via rotary evaporation, and purify the residue using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain leaders, the adoption of this water-soluble melatonin derivative offers substantial strategic advantages in terms of cost efficiency and operational reliability. The elimination of expensive and hazardous organic solvents from the formulation process translates directly into reduced raw material costs and lower waste management expenses. Additionally, the simplified processing requirements mean that production cycles can be shortened, allowing for faster response times to market demands and reduced inventory holding costs. The enhanced stability and solubility of the derivative also reduce the risk of batch failures due to precipitation or crystallization issues, ensuring a more consistent supply of finished goods. These factors collectively contribute to a more resilient supply chain that is better equipped to handle fluctuations in demand without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The shift to aqueous processing eliminates the need for costly solvent recovery systems and reduces the energy consumption associated with solvent evaporation steps. By removing transition metal catalysts and hazardous reagents from the workflow, the overall cost of goods sold is significantly optimized while maintaining high purity standards. This cost efficiency allows manufacturers to offer competitive pricing without sacrificing margin, making the derivative an attractive option for high-volume generic and over-the-counter products. The reduction in hazardous waste disposal fees further enhances the economic viability of the process, providing a clear financial incentive for adoption.
- Enhanced Supply Chain Reliability: The use of readily available and stable reagents ensures that production is not subject to the volatility of specialized chemical markets. The robustness of the synthesis route means that manufacturing can be easily scaled up or down based on demand forecasts without significant retooling or process validation delays. This flexibility is crucial for maintaining continuity of supply in the face of global disruptions or sudden spikes in consumer demand for sleep aid products. Furthermore, the improved stability of the final derivative reduces the need for specialized storage conditions, simplifying logistics and distribution networks.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, minimizing the generation of hazardous byproducts and reducing the overall environmental footprint of production. This alignment with sustainability goals helps companies meet increasingly strict regulatory requirements and corporate social responsibility targets. The ease of scaling the reaction from laboratory to industrial quantities ensures that supply can grow in tandem with market expansion without encountering technical bottlenecks. This scalability is a key factor for long-term partnerships, as it guarantees that the supplier can support the growing needs of global pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these melatonin derivatives. These answers are derived directly from the patent data and practical manufacturing experience to provide clarity on feasibility and performance. Understanding these details is essential for making informed decisions about integrating this technology into your product pipeline. The responses cover aspects ranging from chemical stability to regulatory compliance, ensuring that all stakeholder concerns are adequately addressed.
Q: How does the new derivative improve upon natural melatonin solubility?
A: The patented modification introduces hydrophilic groups such as sulfonate salts to the indole nitrogen, increasing water solubility by nearly 700 times compared to the native molecule.
Q: What are the primary safety advantages of this synthesis route?
A: By enabling dissolution in water rather than toxic organic solvents, the derivative reduces potential side effects associated with solvent residues in final pharmaceutical formulations.
Q: Is the sleep-aiding efficacy maintained after chemical modification?
A: Biological testing confirms that the derivative retains the physiological function of natural melatonin, with animal models showing a significant prolongation of sleep duration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Melatonin Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless and efficient. Our rigorous QC labs and stringent purity specifications guarantee that every batch of melatonin derivative meets the highest international standards for pharmaceutical intermediates. We understand the critical importance of consistency in active ingredients and have optimized our processes to deliver high-purity melatonin derivative with reliable physicochemical properties. Our team of experts is ready to collaborate with your R&D department to customize the synthesis parameters to fit your specific formulation needs, ensuring optimal performance in your final product.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that highlights how our manufacturing efficiencies can reduce your overall production expenses. Let us help you overcome solubility challenges and bring safer, more effective sleep aid solutions to the market with confidence and speed. Reach out today to discuss how our advanced synthesis capabilities can support your supply chain goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
