Industrial Scale Synthesis of Chiral Cycloalkylamines: A Technical Breakthrough for Pharma Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies for the synthesis of chiral building blocks, particularly diastereomerically enriched compounds that serve as critical precursors for active ingredients. Patent CN1989097A introduces a transformative process for the preparation of these compounds, addressing long-standing inefficiencies in stereoselective synthesis. This innovation centers on the reaction between a specific cycloalkyl ketone and an enantiomerically enriched amine to form an intermediate, which is subsequently reduced to yield a highly pure diastereomer. For R&D Directors and Procurement Managers, this patent represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. By shifting away from fragile laboratory-scale methods to a robust industrial protocol, manufacturers can secure a more reliable source of complex chiral amines while mitigating the risks associated with traditional synthetic routes.
The core of this technological advancement lies in its ability to bypass the limitations of earlier methods, such as those described by Pedrosa et al., which relied heavily on expensive and sensitive reagents. The new approach facilitates the commercial scale-up of complex polymer additives and pharmaceutical intermediates by simplifying the reaction conditions and improving overall yield stability. As a leading entity in fine chemical manufacturing, understanding the nuances of this patent allows us to offer superior cost reduction in electronic chemical manufacturing and pharma sectors alike. The following analysis details the mechanistic advantages and commercial implications of adopting this streamlined synthesis pathway for your next project.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of enantiomerically pure cycloalkylamines has been plagued by significant operational challenges that hinder large-scale production. Traditional methods, such as the one disclosed by Pedrosa, depend on the stereoselective ring opening of chiral 1,3-oxazolidines using Grignard reagents or organoaluminum reagents. These reagents are notoriously sensitive to air and moisture, requiring stringent inert atmosphere conditions that drastically increase operational complexity and infrastructure costs. Furthermore, the reliance on expensive chiral auxiliaries, such as (-)-δ-benzylaminocarbinol, necessitates elaborate recovery processes to maintain economic viability. The inability to easily recycle these auxiliaries, combined with the low tolerance for environmental variations, renders such methods unsuitable for the rigorous demands of modern industrial manufacturing. Additionally, the purification of the resulting products often requires chromatography, a technique that is not only costly but also difficult to scale, leading to lower overall yields and inconsistent batch quality.
The Novel Approach
In stark contrast, the process outlined in CN1989097A offers a streamlined alternative that is inherently designed for industrial suitability. This novel approach eliminates the need for air- and moisture-sensitive reagents, replacing them with stable and commercially available starting materials. By reacting a cycloalkyl ketone with an enantiomerically enriched amine, such as phenylethylamine, the process forms an imine intermediate that can be directly reduced without isolation. This telescoping of steps significantly reduces processing time and solvent consumption, leading to substantial cost savings in raw material procurement. Moreover, the method avoids the use of expensive chiral auxiliaries that require recovery, instead utilizing the chiral amine as a temporary directing group that can be removed in a final hydrogenolysis step. This strategic design ensures that the process remains less complex and more robust, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where consistency and reliability are paramount.
Mechanistic Insights into Diastereoselective Imine Reduction
The chemical elegance of this process is rooted in the stereoselective formation and reduction of the imine intermediate. The reaction begins with the condensation of a ketone, where R1 is a cycloalkyl group, with a chiral amine of Formula II. 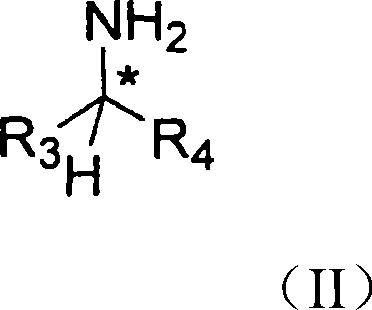 . In this structure, the chiral center denoted by the asterisk plays a crucial role in inducing diastereoselectivity during the subsequent reduction. The steric bulk of the substituents R3 and R4, typically a methyl and a phenyl group respectively, creates a defined chiral environment around the nitrogen atom. When this amine reacts with the ketone to form the imine of Formula III, the existing chirality biases the approach of the reducing agent.
. In this structure, the chiral center denoted by the asterisk plays a crucial role in inducing diastereoselectivity during the subsequent reduction. The steric bulk of the substituents R3 and R4, typically a methyl and a phenyl group respectively, creates a defined chiral environment around the nitrogen atom. When this amine reacts with the ketone to form the imine of Formula III, the existing chirality biases the approach of the reducing agent. 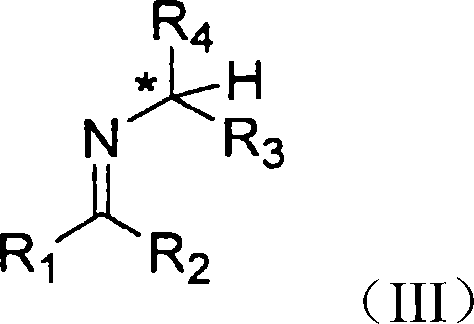 . This bias ensures that the hydride attack occurs preferentially from one face of the imine double bond, resulting in a diastereomerically enriched product. The use of reducing agents like sodium borohydride or catalytic hydrogenation with palladium further enhances this selectivity, allowing for the precise control of stereochemistry without the need for cryogenic temperatures or exotic catalysts.
. This bias ensures that the hydride attack occurs preferentially from one face of the imine double bond, resulting in a diastereomerically enriched product. The use of reducing agents like sodium borohydride or catalytic hydrogenation with palladium further enhances this selectivity, allowing for the precise control of stereochemistry without the need for cryogenic temperatures or exotic catalysts.
Impurity control is another critical aspect where this mechanism excels, providing R&D teams with confidence in the purity profile of the final API intermediate. The process leverages the physical properties of the diastereomeric salts formed during the workup. Unlike free bases which are often oily and difficult to purify, the salts of the Formula IV compounds with acids such as hydrochloric acid can be readily crystallized. This crystallization step acts as a powerful purification tool, effectively rejecting minor diastereomers and other impurities from the crystal lattice. The patent data indicates that a single recrystallization can boost the diastereomeric excess to at least 95%, and often up to 98%. This high level of purity is achieved without the need for preparative chromatography, which is a major bottleneck in traditional synthesis. For quality assurance teams, this means a more consistent impurity spectrum and a reduced risk of genotoxic impurities carrying through to the final drug substance, aligning perfectly with stringent regulatory requirements for high-purity OLED material and pharmaceutical standards.
How to Synthesize Chiral Cycloalkylamines Efficiently
To implement this synthesis route effectively, operators must adhere to specific parameters regarding solvent selection and temperature control to maximize yield and selectivity. The process typically involves contacting the ketone and chiral amine in a solvent that forms an azeotrope with water, such as toluene or isopropyl acetate, to drive the imine formation to completion.
- Condense a cycloalkyl ketone with an enantiomerically enriched amine like phenylethylamine in a solvent such as toluene.
- Reduce the resulting imine intermediate directly using sodium borohydride or catalytic hydrogenation.
- Purify the diastereomeric salt via recrystallization to achieve high enantiomeric excess before hydrogenolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible improvements in cost structure and supply reliability. The elimination of sensitive reagents and complex purification steps directly addresses the pain points of volatility in raw material pricing and production delays. By simplifying the synthetic route, manufacturers can reduce the number of unit operations, which in turn lowers energy consumption and waste generation. This efficiency gain is critical for maintaining competitive pricing in the global market for fine chemical intermediates. Furthermore, the robustness of the chemistry ensures that production schedules are less likely to be disrupted by environmental factors or reagent instability, providing a more predictable supply timeline for downstream customers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the avoidance of expensive chiral auxiliaries and the elimination of chromatographic purification. In traditional methods, the cost of the chiral auxiliary and the losses incurred during its recovery can constitute a significant portion of the total manufacturing cost. By using a chiral amine that serves as a removable directing group, the process sidesteps these expenses entirely. Additionally, the ability to telescope the imine formation and reduction steps without isolating the intermediate reduces solvent usage and labor hours. These cumulative efficiencies result in a drastically simplified cost model, allowing for significant cost savings that can be passed on to clients or reinvested in further process optimization. The qualitative improvement in cost efficiency makes this route highly attractive for high-volume production scenarios.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialty reagents that have limited suppliers or long lead times. This patented method utilizes commodity chemicals such as cycloalkyl ketones and phenylethylamine, which are widely available from multiple global sources. The robustness of the reaction conditions, which do not require inert atmospheres or cryogenic cooling, further reduces the risk of batch failures due to equipment malfunction or operator error. This resilience ensures a steady flow of materials, reducing lead time for high-purity pharmaceutical intermediates and allowing manufacturers to respond more agilely to market demand fluctuations. The decreased dependency on fragile reagents translates to a more secure and stable supply chain for critical drug substances.
- Scalability and Environmental Compliance: Scaling a chemical process from the laboratory to the plant floor often reveals hidden challenges related to heat transfer and mixing, particularly with exothermic reactions involving sensitive reagents. The chemistry described in this patent is inherently scalable due to its mild reaction conditions and the use of standard solvents. The reduction step, whether using sodium borohydride or catalytic hydrogenation, can be safely managed in large reactors with established safety protocols. Furthermore, the avoidance of chromatography significantly reduces the volume of organic waste generated, aligning with increasingly strict environmental regulations. The ability to purify via crystallization rather than column chromatography minimizes solvent waste and simplifies wastewater treatment, contributing to a greener manufacturing footprint and easier compliance with environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is this method superior to the Pedrosa process for industrial production?
A: Unlike the Pedrosa process which relies on air-sensitive Grignard reagents and expensive chiral auxiliaries requiring recovery, this patented method utilizes robust reagents and avoids complex purification steps like chromatography.
Q: What level of stereochemical purity can be achieved with this route?
A: Through a single recrystallization step of the acid salt, the process consistently achieves diastereomeric excesses of at least 95%, often reaching 98%, ensuring high-quality intermediates.
Q: Is this synthesis suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for industrial suitability, avoiding moisture-sensitive reagents and utilizing common solvents like toluene and isopropyl acetate for easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Cycloalkylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this process are fully realized in practice. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that monitor every batch for diastereomeric and enantiomeric excess. Our infrastructure is designed to handle the specific requirements of chiral synthesis, including dedicated lines for hydrogenation and crystallization, guaranteeing the consistency and quality required by top-tier pharmaceutical companies.
We invite you to collaborate with us to leverage this advanced synthesis route for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral intermediates can enhance your supply chain efficiency and product quality.
