Advanced Phosgene-Mediated Coupling for Scalable Quinolone Lactam Antibacterial Production
Advanced Phosgene-Mediated Coupling for Scalable Quinolone Lactam Antibacterial Production
The pharmaceutical industry continuously seeks robust synthetic routes for dual-action antibacterial agents, and Patent CN1046523C presents a pivotal advancement in this domain. This intellectual property discloses a highly efficient method for preparing compounds that integrate a quinolone moiety and a beta-lactam moiety, collectively known as Quinolone Lactam Antimicrobials (QLAs). The core innovation lies in a specific coupling strategy that utilizes phosgene to activate the lactam component, forming a reactive chloroformate intermediate which is subsequently coupled with a quinolone derivative. This approach addresses historical challenges in synthesizing these complex hybrid structures, particularly those containing sensitive penem substituents. By establishing a carbamate-containing linking moiety through this activated pathway, the process ensures higher purity and improved overall yields compared to traditional linkage methods. For R&D directors and procurement specialists, understanding this patented methodology is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity QLAs with consistent quality.
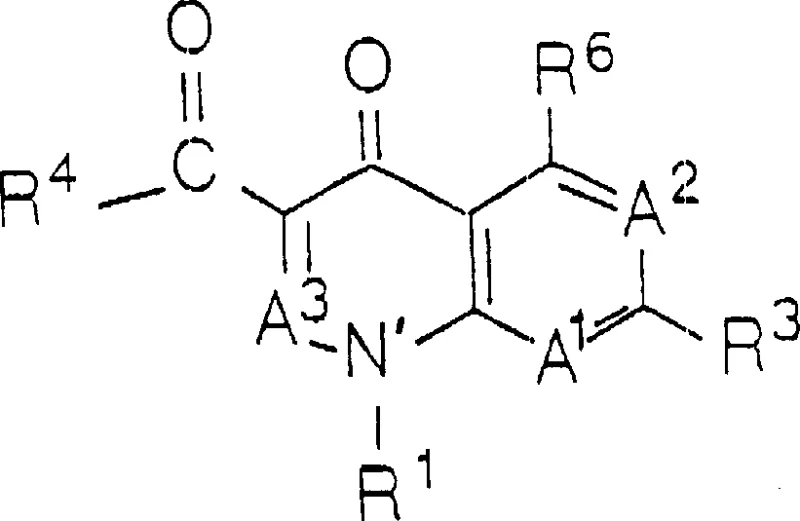
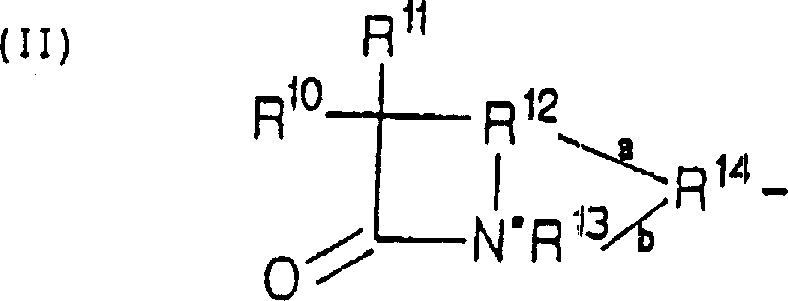
The structural complexity of QLAs requires precise control over the linkage between the two pharmacophores. As illustrated in the general formula, the quinolone portion (Formula I) can vary significantly with substituents at positions R1 through R7, allowing for tuning of antibacterial spectra. Similarly, the beta-lactam portion (Formula II) encompasses a wide range of structures including penems, cephems, and monobactams. The patent specifies that the linking moiety L connects these two distinct units, often through a carbamate bond formed via the reaction of a hydroxyl-bearing lactam with phosgene. This chemical architecture is designed to maximize antimicrobial efficacy while maintaining metabolic stability. The versatility of the R groups allows manufacturers to produce a diverse library of analogs, making this synthetic route a valuable asset for developing next-generation antibiotics. The ability to modify these positions without compromising the integrity of the linkage is a key factor in the commercial viability of this technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of QLAs involved synthesizing protected substituted beta-lactam and quinolone moieties separately before attempting to link them. However, literature and prior art indicate that these conventional connection methods often suffered from generally low yields. This inefficiency was particularly pronounced when preparing QLAs possessing penem substituent moieties, which are chemically sensitive and prone to degradation under harsh coupling conditions. Traditional activation strategies sometimes failed to provide the necessary reactivity without inducing decomposition of the beta-lactam ring, leading to significant material loss and increased production costs. Furthermore, the impurity profiles generated by these older methods were often complex, requiring extensive and costly purification steps to meet pharmaceutical standards. For supply chain heads, these limitations translated into longer lead times and unpredictable availability of high-purity intermediates, creating bottlenecks in the drug development pipeline.
The Novel Approach
The novel approach detailed in Patent CN1046523C overcomes these deficiencies by employing a phosgene-mediated activation strategy. Several attachment methods utilizing phosgene have been found to be useful, enabling the efficient synthesis of QLAs with high yields. The process involves reacting a lactam compound of the formula B-L-H with phosgene to form an intermediate compound of the formula B-L-C(=O)-Cl, where L is oxygen. This chloroformate intermediate is highly reactive towards nucleophilic attack by the quinolone amine or hydroxyl group, facilitating a rapid and clean coupling reaction. Preferably, the coupling step comprises adding a solution containing the quinolone compound to a solution containing the intermediate compound, which helps manage exotherms and control side reactions. This method is particularly effective for sensitive substrates, ensuring that the delicate beta-lactam ring remains intact throughout the synthesis. The result is a streamlined process that significantly reduces waste and improves the economic feasibility of producing these potent antibacterial agents.
Mechanistic Insights into Phosgene-Mediated Carbamate Formation
The mechanistic pathway of this synthesis relies on the formation of a mixed anhydride or chloroformate species which acts as an electrophile. When the lactam alcohol reacts with phosgene, typically in a solvent like dichloromethane or toluene, the hydroxyl group is converted into a good leaving group. This activation is critical because direct esterification or amidation between the unactivated lactam and quinolone would be thermodynamically unfavorable or kinetically slow. The subsequent addition of the quinolone compound, often in its silylated form (where R is Si(R)3) to enhance solubility and nucleophilicity, drives the formation of the carbamate linkage. The reaction is preferably performed at a temperature of from about -80°C to about 0°C. Maintaining this cryogenic environment is essential to suppress competing reactions such as the polymerization of phosgene or the hydrolysis of the activated intermediate. Strict temperature control ensures that the reaction proceeds selectively to form the desired Q-L-B structure without generating significant amounts of symmetric ureas or carbonates.
Impurity control is another critical aspect managed by this mechanism. By generating the chloroformate intermediate in situ and immediately consuming it with the quinolone partner, the process minimizes the residence time of reactive species that could degrade. The patent notes that preferred antimicrobial compounds made by these processes are those where the beta-lactam moiety is a penem, highlighting the method's compatibility with fragile structures. Additionally, the use of protecting groups, such as allyl or silyl esters, prior to the reacting and coupling steps further safeguards the carboxylic acid functionalities present on both moieties. These esters are stable under the coupling conditions but can be readily removed in a subsequent deprotection step using standard reagents like palladium catalysts or fluoride sources. This orthogonal protection strategy ensures that the final product is obtained as the free acid or a specific salt, ready for formulation, with a clean impurity profile that simplifies downstream processing.
How to Synthesize Quinolone Lactam Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the preparation of the lactam intermediate, followed by the activation with phosgene and subsequent coupling with the quinolone component. Detailed operational parameters, such as solvent choices (dichloromethane, THF) and stoichiometry, are optimized to maximize conversion. The examples provided in the patent demonstrate the practical application of this method, showing how to handle the reagents safely and effectively. For instance, the use of a three-necked flask equipped with a low-temperature thermometer and overhead stirrer is recommended to ensure homogeneous mixing and precise thermal regulation. The addition rates of reagents are controlled to maintain the reaction temperature within the specified range of -75°C to -70°C during the critical activation phase. This level of detail underscores the reproducibility of the method, making it an attractive option for contract manufacturing organizations looking to offer cost reduction in API manufacturing services.
- React the lactam compound containing a hydroxyl group with phosgene at low temperatures (-80°C to 0°C) to form a reactive chloroformate intermediate.
- Prepare the quinolone compound, optionally protecting carboxyl groups as esters or using silylated derivatives to enhance nucleophilicity.
- Couple the quinolone solution with the chloroformate intermediate under strict temperature control, followed by deprotection to yield the final active acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this phosgene-mediated synthesis route offers substantial benefits for procurement and supply chain management. The primary advantage lies in the significant improvement in process efficiency and yield. By overcoming the low-yield limitations of conventional methods, this technology reduces the amount of starting material required to produce a given quantity of the final active ingredient. This directly translates to lower raw material costs and reduced waste disposal expenses, contributing to a more sustainable and economical manufacturing process. For procurement managers, this means a more stable pricing structure for the intermediates, as the process is less susceptible to fluctuations in raw material availability due to its higher efficiency. The ability to produce high-purity products with fewer impurities also reduces the burden on quality control laboratories, speeding up the release of batches for clinical or commercial use.
- Cost Reduction in Manufacturing: The elimination of inefficient coupling steps and the improvement in overall yield drastically simplify the production workflow. Since the method avoids the need for multiple protection and deprotection cycles that were common in older routes, the number of unit operations is reduced. This reduction in processing time and resource consumption leads to substantial cost savings. Furthermore, the use of common solvents like dichloromethane and toluene, which are readily available and easily recycled, enhances the economic viability of the process. The high yield specifically noted for penem-containing QLAs means that high-value intermediates are not lost to side reactions, maximizing the return on investment for expensive starting materials.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method contributes to a more reliable supply chain. The process has been demonstrated to be scalable, with examples showing successful execution in reactors ranging from 1-liter to 30-liter volumes. This scalability indicates that the chemistry can be transferred to larger commercial vessels without significant re-optimization, ensuring continuity of supply as demand grows. The use of well-understood reagents like phosgene, handled with appropriate safety protocols, means that the process can be implemented in standard fine chemical facilities. For supply chain heads, this reduces the risk of production delays caused by specialized equipment requirements or unstable reaction conditions, ensuring that critical antibacterial intermediates are available when needed.
- Scalability and Environmental Compliance: The process design inherently supports environmental compliance through waste minimization. Higher yields mean less chemical waste per kilogram of product, aligning with green chemistry principles. The deprotection steps utilize reagents that can be managed within standard waste streams, and the solvents used are compatible with existing recovery systems. The ability to scale up to 30 liters and beyond demonstrates that the thermal management and mixing requirements are achievable in industrial settings. This scalability ensures that the method can meet the demands of commercial scale-up of complex pharmaceutical intermediates without compromising on safety or environmental standards, making it a future-proof choice for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of quinolone lactam antibacterial agents. These answers are derived directly from the technical specifications and examples provided in Patent CN1046523C. Understanding these details is essential for stakeholders evaluating the feasibility of this technology for their specific applications. The information covers critical process parameters, structural scope, and the advantages of the disclosed method over prior art. This transparency helps build confidence in the technical robustness of the synthesis route and its potential for successful commercialization.
Q: What is the key advantage of the phosgene-mediated method for QLAs?
A: The phosgene-mediated method significantly improves yields compared to conventional linkage methods, particularly when preparing QLAs with penem substituent moieties, by forming a stable carbamate-containing linking moiety.
Q: What temperature range is critical for this synthesis?
A: The process steps, including the reaction with phosgene and the subsequent coupling, are preferably performed at a temperature of from about -80°C to about 0°C to maintain intermediate stability and prevent side reactions.
Q: Can this method be scaled for commercial production?
A: Yes, the patent examples demonstrate scalability up to 30-liter reactors, indicating that the process is suitable for commercial scale-up of complex pharmaceutical intermediates with appropriate safety controls for phosgene handling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolone Lactam Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced methodologies like the one described in Patent CN1046523C to deliver superior pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to full-scale manufacturing. We understand the critical importance of stringent purity specifications in the production of antibacterial agents, and our rigorous QC labs are equipped to verify every batch against the highest international standards. By partnering with us, you gain access to a supply chain that prioritizes quality, consistency, and regulatory compliance, mitigating the risks associated with complex chemical synthesis.
We invite you to discuss your specific requirements with our technical procurement team to explore how we can support your drug development goals. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new quinolone lactam derivatives, we are ready to assist. Our commitment to innovation and customer success makes us the ideal partner for sourcing high-quality intermediates. Contact us today to initiate a conversation about optimizing your production strategy and securing a reliable supply of these critical antibacterial components.
