Advanced Manufacturing of Azacycloalkanoylaminothiazole Intermediates for Oncology Drug Development
Advanced Manufacturing of Azacycloalkanoylaminothiazole Intermediates for Oncology Drug Development
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic scaffolds, particularly those serving as potent kinase inhibitors. Patent CN1227250C discloses a groundbreaking methodology for the preparation of 5-(2-oxazolylalkylthio)-2-azacycloalkanoylaminothiazoles, a class of compounds exhibiting significant efficacy as cyclin-dependent kinase (cdks) inhibitors. These molecules are critical in the development of therapeutics for proliferative diseases, including various forms of cancer such as bladder, breast, colon, and lung malignancies. The disclosed process represents a paradigm shift from traditional synthetic routes by addressing key safety and scalability bottlenecks inherent in earlier methodologies. By introducing a novel sequence involving quaternary ammonium salt intermediates and optimized cyclization conditions, this technology enables the production of high-purity pharmaceutical intermediates suitable for commercial scale-up. For R&D directors and procurement specialists, understanding the nuances of this patented route is essential for securing a reliable supply chain for next-generation oncology drugs.
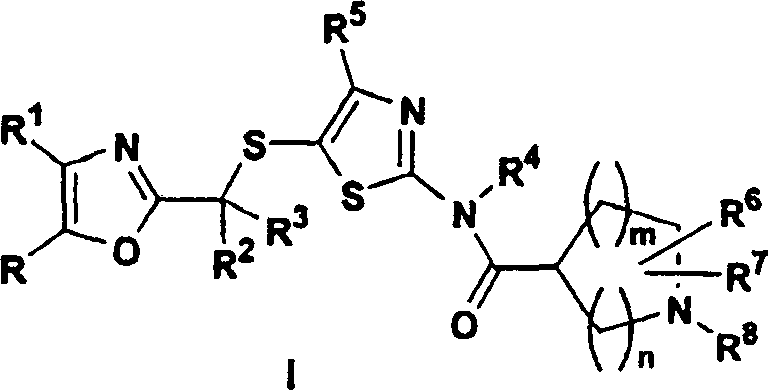
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques, specifically those described in WO 9924416 and U.S. Patent No. 6,040,321, relied heavily on the use of hazardous alpha-diazoketones and 5-acetylthio-2-acetylaminothiazole starting materials. These conventional pathways presented severe industrial limitations, primarily due to the intrinsic instability and safety risks associated with diazo compounds, which pose explosion hazards during large-scale handling. Furthermore, the purification of the resulting intermediates often necessitated expensive and time-consuming chromatographic separation techniques, which are economically unviable for multi-kilogram or ton-scale manufacturing. The reliance on such complex purification steps not only inflated the cost of goods sold but also introduced significant variability in batch-to-batch consistency. Additionally, the formation of oxazole rings using traditional dehydrating agents like phosphorus oxychloride generated substantial amounts of acidic waste, complicating environmental compliance and waste management protocols for chemical manufacturers.
The Novel Approach
The innovative process detailed in CN1227250C circumvents these historical challenges by employing a safer and more efficient synthetic strategy centered on the formation of a quaternary ammonium salt intermediate. Instead of utilizing dangerous diazo precursors, the method reacts an alpha-substituted ketone, such as an alpha-halopinacolone, with hexamethylenetetramine to generate a stable quaternary salt, which is subsequently hydrolyzed to the desired alpha-aminoketone. This modification eliminates the need for hazardous azide or diazo chemistry, drastically improving operational safety profiles. Moreover, the process facilitates the isolation of intermediates through crystallization rather than chromatography, ensuring high purity levels while minimizing solvent consumption. The strategic use of Burgess' reagent for the cyclodehydration step further enhances the workflow by allowing for easy aqueous workup, thereby streamlining the overall production timeline and reducing the environmental footprint associated with acidic waste disposal.
Mechanistic Insights into Oxazole Cyclization and Thiazole Coupling
A critical aspect of this synthesis lies in the efficient construction of the oxazole ring, a key structural motif required for biological activity. The patent elucidates a mechanism where an alpha-aminoketone is first acylated with an alpha-haloacyl halide to form an amide intermediate. Subsequent cyclodehydration transforms this linear amide into the cyclic 2-oxazolylalkyl derivative. Unlike traditional methods that employ harsh phosphorus-based dehydrating agents which create difficult-to-remove byproducts, this invention优选 (prefers) the use of Burgess' reagent, chemically known as (methoxycarbonylsulfamoyl)triethylammonium hydroxide. This reagent promotes mild and selective dehydration, preventing side reactions that could compromise the integrity of the sensitive thioether linkage. The mechanistic advantage here is twofold: it preserves the stereochemical integrity of chiral centers if present, and it generates byproducts that are easily separated during the aqueous quench, leading to significantly higher isolated yields of the oxazole intermediate.
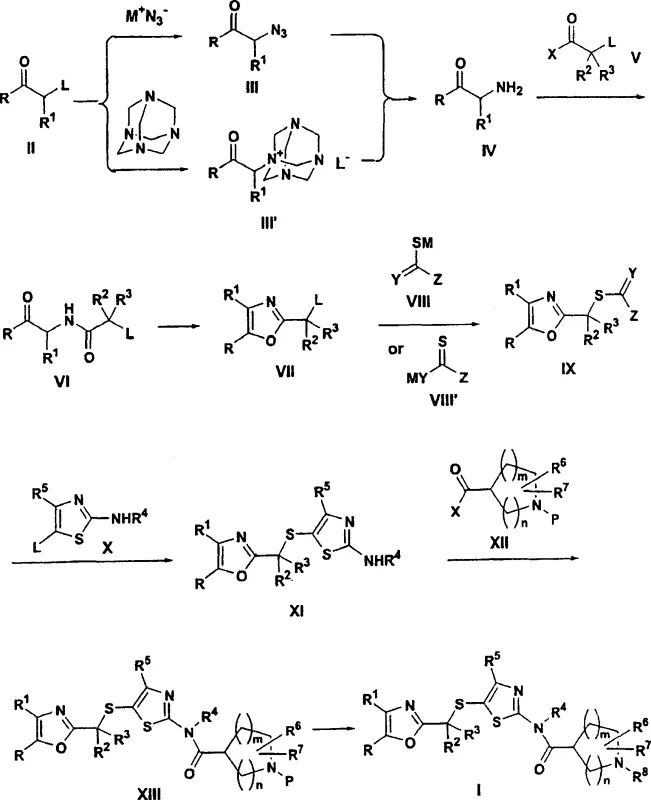
Following the formation of the oxazole core, the process involves a nucleophilic substitution to introduce the sulfur bridge, connecting the oxazole moiety to the thiazole ring system. The 2-oxazolylalkyl halide reacts with a sulfur-containing reagent, such as thiourea or a thioacid salt, to generate a 2-oxazolylalkylsulfide intermediate. This sulfide then undergoes a coupling reaction with a 5-halo-2-aminothiazole derivative under basic conditions. The final assembly of the pharmacophore is achieved through an amide coupling between the resulting amino-thiazole and a protected azacycloalkanoic acid derivative. This modular approach allows for significant flexibility in varying the R groups, enabling medicinal chemists to rapidly generate analog libraries for structure-activity relationship (SAR) studies while maintaining a consistent and scalable backbone synthesis for the most promising candidates.
How to Synthesize 5-(2-oxazolylalkylthio)-2-azacycloalkanoylaminothiazoles Efficiently
The execution of this synthetic route requires precise control over reaction parameters to maximize yield and purity at every stage. The process begins with the conversion of an alpha-haloketone to a quaternary ammonium salt using hexamethylenetetramine in a ketone solvent like acetone, followed by acid hydrolysis to release the alpha-aminoketone. This amine is then acylated and cyclized to form the oxazole ring, which is subsequently converted to a sulfide and coupled with the thiazole fragment. The final steps involve coupling with the azacycloalkanoic acid and deprotection to yield the target molecule. While the general chemistry is robust, specific attention must be paid to the choice of dehydrating agents and the purification of the quaternary salt intermediate to ensure optimal throughput. The detailed standardized synthesis steps see the guide below.
- React alpha-substituted ketone with hexamethylenetetramine to form a quaternary ammonium salt, followed by acid hydrolysis to yield the alpha-aminoketone.
- Acylate the aminoketone with an alpha-haloacyl halide and perform cyclodehydration using Burgess reagent to form the 2-oxazolylalkyl derivative.
- Convert the oxazole derivative to a sulfide using thiourea, couple with 5-halo-2-aminothiazole, and finally acylate with a protected azacycloalkanoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible benefits regarding cost stability and supply continuity. By eliminating the need for hazardous diazo compounds, the process reduces the regulatory burden and insurance costs associated with handling explosive materials, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. The shift from chromatographic purification to crystallization-based isolation significantly lowers solvent usage and waste disposal costs, enhancing the overall economic viability of the production campaign. Furthermore, the use of commercially available and inexpensive reagents like hexamethylenetetramine and Burgess' reagent ensures that raw material sourcing remains stable and unaffected by niche supply chain disruptions. This reliability is crucial for maintaining uninterrupted production schedules for critical oncology therapeutics.
- Cost Reduction in Manufacturing: The elimination of expensive chromatographic purification steps and hazardous reagents leads to substantial cost savings. By utilizing crystallization for isolation, the process minimizes solvent consumption and reduces the time required for downstream processing. The avoidance of transition metal catalysts or rare reagents further optimizes the bill of materials, making the final API intermediate more price-competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetone, ethanol, and hexamethylenetetramine ensures a robust supply chain that is less susceptible to geopolitical or logistical volatility. Since the process avoids specialized, hard-to-source precursors like alpha-diazoketones, manufacturers can secure long-term contracts for raw materials with greater confidence. This stability translates into reduced lead times for high-purity pharmaceutical intermediates, allowing drug developers to accelerate their clinical trial timelines with predictable material availability.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, moving seamlessly from laboratory benchtop to multi-ton production. The use of Burgess' reagent and aqueous workups simplifies waste treatment, aligning with stringent environmental regulations regarding acidic and organic waste discharge. This eco-friendly profile not only mitigates regulatory risks but also enhances the corporate sustainability metrics of the manufacturing partner, a key consideration for modern pharmaceutical companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the new route. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing manufacturing portfolios.
Q: Why is the hexamethylenetetramine route preferred over the azide route for this synthesis?
A: The patent highlights that while azide reduction is possible, safety considerations and economic factors make the hexamethylenetetramine pathway superior for large-scale operations, offering yields above 90% without hazardous reagents.
Q: How does this process improve purification compared to prior art methods?
A: Unlike previous methods requiring expensive chromatographic separation, this novel process utilizes crystallization and standard extraction techniques, significantly simplifying downstream processing and reducing solvent waste.
Q: What specific dehydration agent is recommended for the oxazole ring closure?
A: The invention preferably utilizes Burgess' reagent ((methoxycarbonylsulfamoyl)triethylammonium hydroxide) instead of traditional phosphorus oxyhalides, facilitating easier product isolation from aqueous workups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azacycloalkanoylaminothiazole Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN1227250C are executed with precision and efficiency. Our state-of-the-art facilities are equipped to handle the specific requirements of heterocyclic chemistry, including the safe handling of halogenated intermediates and the rigorous purification needed for oncology drugs. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of azacycloalkanoylaminothiazole intermediate meets the highest international standards for pharmaceutical applications. Our commitment to quality ensures that your drug development pipeline remains uninterrupted by supply inconsistencies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation kinase inhibitor projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your path to market while optimizing your overall production costs.
