Advanced Synthesis of Benzothiazolyl Phenyl-Acetic Acid Derivatives for Commercial API Production
Advanced Synthesis of Benzothiazolyl Phenyl-Acetic Acid Derivatives for Commercial API Production
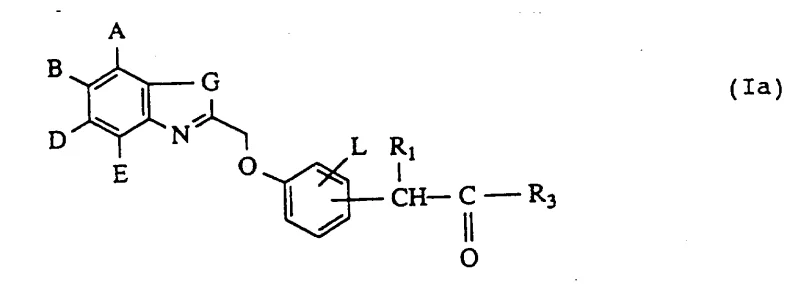
The pharmaceutical landscape continuously demands novel intermediates capable of addressing complex inflammatory pathways, and Patent CN1084166A presents a significant advancement in this domain by disclosing a robust method for preparing substituted (benzothiazolyl- and quinoxalinyl-methoxy)phenyl-acetic acid derivatives. These specific chemical entities serve as potent active compounds in medicine, particularly functioning as leukotriene synthesis inhibitors which are vital for treating respiratory and inflammatory conditions. The technical breakthrough lies in the versatile synthetic route that allows for the precise introduction of heterocyclic moieties onto a phenyl-acetic acid scaffold through controlled etherification and subsequent alkylation steps. For R&D directors and procurement specialists, understanding the underlying chemistry of this patent is crucial because it offers a pathway to high-purity intermediates that can be manufactured with consistent quality. The structural flexibility defined in the patent allows for various substituents on the aromatic rings, enabling the fine-tuning of pharmacological properties while maintaining a scalable synthetic backbone suitable for industrial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing complex heterocyclic ether-linked pharmaceutical intermediates often suffer from significant drawbacks regarding regioselectivity and reaction condition severity. Conventional approaches might rely on harsh acidic conditions or expensive transition metal catalysts that introduce difficult-to-remove impurities into the final product, thereby complicating the purification process and increasing overall production costs. Furthermore, older synthetic routes frequently struggle with the stability of the heterocyclic ring systems under reactive conditions, leading to decomposition or the formation of unwanted by-products that degrade the yield. The lack of modularity in traditional methods also means that changing a substituent often requires a complete redesign of the synthetic route, which is inefficient for rapid drug development cycles. These limitations create bottlenecks in the supply chain, as achieving the necessary purity profiles for regulatory approval becomes increasingly difficult and resource-intensive when using outdated chemical methodologies.
The Novel Approach
The methodology outlined in Patent CN1084166A overcomes these historical challenges by employing a modular strategy centered on the reaction of heterocyclic methyl halides with corresponding phenoxy compounds. This approach utilizes mild basic conditions and common inert solvents, significantly reducing the risk of heterocyclic ring degradation while ensuring high conversion rates. By separating the construction of the ether linkage from the modification of the acetic acid side chain, the process allows for greater control over the final molecular architecture and impurity profile. This decoupling of synthetic steps means that manufacturers can optimize each stage independently, leading to a more robust and reliable production process. The ability to use standard alkali metal bases and readily available halides makes this novel approach not only chemically superior but also economically advantageous for large-scale manufacturing operations seeking to minimize waste and maximize throughput.
Mechanistic Insights into Heterocyclic Etherification and Grignard Alkylation
The core chemical transformation driving this technology is the nucleophilic substitution reaction where a heterocyclic methyl halide acts as the electrophile and a phenoxy compound serves as the nucleophile. This reaction typically proceeds in polar aprotic solvents such as dimethylformamide (DMF) or tetrahydrofuran (THF) in the presence of bases like potassium carbonate or sodium hydride to facilitate the deprotonation of the phenolic hydroxyl group. The mechanism ensures that the oxygen atom of the phenol attacks the methylene carbon attached to the heterocycle, displacing the halogen atom and forming a stable ether bond that links the two distinct aromatic systems. This step is critical because it establishes the core scaffold of the molecule, and the choice of base and solvent directly influences the reaction kinetics and the suppression of side reactions such as elimination or over-alkylation. Understanding this mechanistic detail is essential for process chemists aiming to replicate the high yields reported in the patent examples.
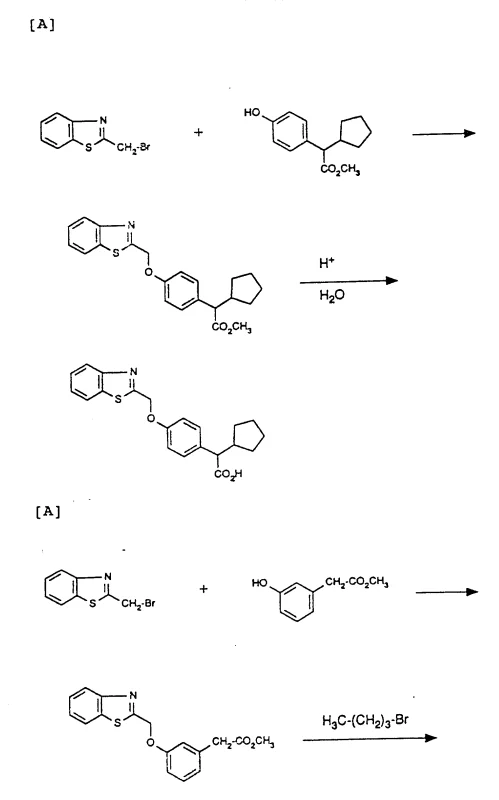
Following the formation of the ether linkage, the synthesis may proceed through divergent pathways depending on the desired final functionality, such as direct alkylation or Grignard addition to a glyoxylic ester intermediate. In the Grignard pathway, an organomagnesium reagent attacks the carbonyl carbon of a glyoxylic ester, introducing complex alkyl or cycloalkyl groups at the alpha position of the acetic acid chain. This step requires strict temperature control, often ranging from -40°C to +25°C, to manage the exothermic nature of the reaction and prevent the decomposition of the sensitive Grignard reagent. The subsequent workup involves careful quenching with aqueous ammonium chloride or dilute acid to protonate the alkoxide intermediate and release the final alcohol or acid product. This level of mechanistic precision allows for the creation of chiral centers and complex steric environments that are often required for high-affinity binding to biological targets like 5-lipoxygenase enzymes.
How to Synthesize Substituted Benzothiazolyl Phenyl-Acetic Acid Efficiently
To successfully implement this synthesis in a commercial setting, one must adhere to a disciplined sequence of unit operations that prioritize safety and reproducibility. The process begins with the preparation of the heterocyclic methyl halide, often achieved through radical bromination of the corresponding methyl-heterocycle using N-bromosuccinimide, followed by the critical etherification step in a dry, inert atmosphere. Detailed standardized synthetic steps see the guide below, which outlines the precise stoichiometry, temperature profiles, and workup procedures necessary to achieve the high purity standards required for pharmaceutical intermediates. Operators must ensure that all solvents are anhydrous during the Grignard addition phase to prevent premature quenching of the reagent, and purification should leverage the solubility differences described in the patent, utilizing recrystallization from methanol or diisopropyl ether to remove trace impurities. Adhering to these protocols ensures that the final product meets the stringent specifications needed for downstream drug substance manufacturing.
- Prepare the heterocyclic methyl halide precursor via bromination of the corresponding methyl-heterocycle using N-bromosuccinimide.
- Perform nucleophilic substitution by reacting the halide with a protected phenoxy compound in an inert solvent like DMF with a base.
- Execute side-chain modification via Grignard addition to glyoxylic esters or hydrolyze the ester group to yield the final carboxylic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the chemical route described in this patent offers substantial benefits regarding raw material availability and process economics. The starting materials, such as benzothiazoles, quinoxalines, and substituted phenols, are commodity chemicals that are widely produced by the global fine chemical industry, ensuring a stable and competitive supply base. Because the synthesis avoids the use of precious metal catalysts like palladium or platinum, which are subject to volatile market pricing and supply constraints, the overall cost of goods sold can be significantly optimized. The reliance on standard unit operations such as filtration, distillation, and crystallization means that the process can be transferred to existing multipurpose manufacturing facilities without requiring specialized equipment investments. This compatibility with standard infrastructure reduces capital expenditure risks and accelerates the timeline from process development to commercial production, providing a clear competitive advantage in the fast-paced pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of stoichiometric inorganic bases drastically lowers the direct material costs associated with production. Furthermore, the high yields reported in the patent examples suggest that raw material utilization is efficient, minimizing the volume of waste generated per kilogram of product. The ability to purify the final compounds through simple recrystallization rather than costly preparative chromatography further contributes to substantial cost savings in the downstream processing stages. These factors combine to create a lean manufacturing profile that is highly attractive for procurement managers focused on long-term margin improvement.
- Enhanced Supply Chain Reliability: Since the synthetic route relies on robust and well-understood chemical transformations, the risk of batch failure due to unpredictable reaction behavior is significantly minimized. The use of common solvents like DMF, THF, and ethyl acetate ensures that supply chain disruptions for specialized reagents are unlikely to halt production. Additionally, the stability of the intermediates allows for flexible scheduling and potential storage of key precursors, providing a buffer against demand fluctuations. This reliability is critical for supply chain heads who must guarantee continuous availability of active pharmaceutical ingredients to meet patient needs without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable because the reaction conditions do not involve extreme pressures or temperatures that would require specialized high-pressure reactors. The waste streams generated are primarily aqueous salts and organic solvents that can be managed through standard wastewater treatment and solvent recovery systems commonly found in modern chemical plants. By avoiding heavy metal contamination, the environmental burden of the process is reduced, simplifying regulatory compliance and disposal logistics. This alignment with green chemistry principles enhances the sustainability profile of the supply chain, which is increasingly important for corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in a production environment. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance for decision-makers. Understanding these details helps clarify the feasibility of adopting this route for specific project requirements and highlights the versatility of the chemical platform.
Q: What are the primary therapeutic applications of these benzothiazolyl derivatives?
A: These compounds function as potent 5-lipoxygenase inhibitors, making them critical intermediates for developing treatments for asthma, bronchitis, inflammatory rheumatosis, and other arachidonic acid metabolic disorders.
Q: How does the patented etherification method improve process scalability?
A: The method utilizes standard nucleophilic substitution conditions with commercially available bases and solvents like DMF or THF, avoiding exotic catalysts and allowing for straightforward scale-up from kilogram to metric ton quantities.
Q: What purity levels can be achieved with the described crystallization methods?
A: The patent describes recrystallization from solvents such as methanol, diisopropyl ether, and ethyl acetate, which typically yields high-purity solids suitable for subsequent pharmaceutical processing without extensive chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Phenyl-Acetic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure required to translate complex patent methodologies like CN1084166A into reliable commercial supply chains. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to full-scale market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of high-purity pharmaceutical intermediates meets your exact requirements. Our commitment to quality assurance means that you can trust us to deliver materials that are consistent, safe, and ready for integration into your final drug products.
We invite you to contact our technical procurement team to discuss how we can support your specific needs for leukotriene inhibitor intermediates. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can benefit your bottom line. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your project timeline, ensuring that your supply chain remains robust and competitive in the global marketplace.
