Advanced Synthesis of Sulfur-Containing Aryl Carbon Glycosides for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking robust and efficient synthetic routes for novel therapeutic agents, particularly in the realm of SGLT2 inhibitors like dapagliflozin and empagliflozin. Patent CN113582956A introduces a groundbreaking methodology for the synthesis of sulfur-containing aryl carbon glycoside compounds, represented by Formula (1). This innovation addresses critical bottlenecks in traditional glycosylation strategies by employing a Pummerer reaction mechanism that bypasses the need for pre-functionalized substrates and transition metal catalysis. For R&D directors and procurement specialists alike, this represents a significant shift towards more sustainable and cost-effective manufacturing processes for high-value API intermediates. The structural versatility of these compounds allows for extensive modification at the R1, R2, and R3 positions, enabling the rapid generation of diverse analog libraries for drug discovery programs.
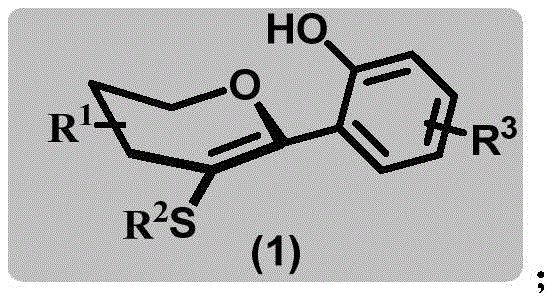
As a reliable pharmaceutical intermediates supplier, understanding the underlying chemistry is crucial for ensuring supply chain stability. The disclosed method utilizes sulfur-containing alkenes (Formula 2) and phenolic compounds (Formula 3) as primary building blocks. By leveraging this specific chemical architecture, manufacturers can access a broad spectrum of derivatives that serve as key precursors for next-generation antidiabetic medications. The ability to construct these complex C-glycosidic bonds directly without protecting group manipulation on the sugar moiety significantly streamlines the synthetic timeline, offering a competitive edge in the fast-paced landscape of metabolic disease treatment development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfur-containing aryl carbon glycosides has been plagued by inefficient multi-step sequences that rely heavily on transition metal catalysis. Traditional approaches often necessitate the pre-functionalization of raw materials, introducing additional synthetic steps that increase both material costs and waste generation. The reliance on precious metal catalysts not only escalates the expense of cost reduction in API manufacturing but also introduces the risk of heavy metal contamination, which requires rigorous and costly purification protocols to meet regulatory standards for pharmaceutical ingredients. Furthermore, these conventional routes frequently suffer from harsh reaction conditions, limited substrate scope, and moderate yields, creating substantial barriers to the commercial scale-up of complex pharmaceutical intermediates. The cumulative effect of these inefficiencies is a prolonged lead time and reduced overall process robustness, which is untenable for large-scale industrial applications.
The Novel Approach
In stark contrast, the methodology outlined in CN113582956A employs a direct Pummerer-type reaction strategy that fundamentally simplifies the synthetic pathway. By reacting sulfur-containing alkenes directly with phenols in the presence of an activating reagent, this novel approach eliminates the need for pre-functionalization and transition metals entirely. This metal-free protocol not only reduces the environmental footprint but also simplifies the downstream processing, as there is no need for specialized metal scavenging resins or complex filtration steps. The reaction proceeds under remarkably mild conditions, typically at 0°C in nitromethane, which preserves the integrity of sensitive functional groups and minimizes side reactions. This efficiency translates directly into higher throughput and improved resource utilization, making it an ideal candidate for reducing lead time for high-purity API precursors in a commercial setting.
Mechanistic Insights into TFAA-Activated Pummerer Reaction
The core of this synthetic breakthrough lies in the activation of the sulfur-containing alkene via a Pummerer-like mechanism facilitated by trifluoroacetic anhydride (TFAA). Upon addition of TFAA to the reaction mixture containing the sulfoxide alkene, an electrophilic sulfonium species is generated in situ. This highly reactive intermediate is then susceptible to nucleophilic attack by the electron-rich aromatic ring of the phenolic compound. The regioselectivity of this attack is governed by the electronic properties of the phenol substituents, allowing for precise control over the substitution pattern on the resulting aryl ring. The use of nitromethane as the solvent is critical, as it stabilizes the charged intermediates and facilitates the smooth progression of the reaction without promoting decomposition pathways often seen in more polar protic solvents.
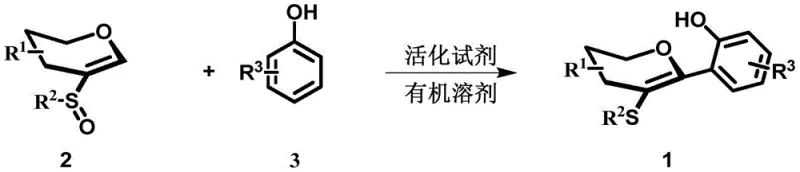
From an impurity control perspective, this mechanism offers distinct advantages. The absence of radical pathways associated with metal catalysis means that the impurity profile is generally cleaner and more predictable. The reaction stoichiometry is carefully optimized, with a preferred molar ratio of alkene to phenol of 1:1.5 and an activator to alkene ratio of 3:1, ensuring complete conversion of the limiting reagent while minimizing the formation of bis-arylated byproducts. The reaction is conducted under an air atmosphere, which further simplifies operational requirements compared to strictly anaerobic conditions. Post-reaction workup involves a straightforward wash with saturated sodium bicarbonate solution to neutralize acidic byproducts, followed by standard extraction and column chromatography, yielding the target high-purity sulfur-containing aryl carbon glycosides with excellent purity profiles suitable for subsequent biological evaluation.
How to Synthesize Sulfur-Containing Aryl Carbon Glycosides Efficiently
Implementing this synthesis route requires careful attention to reagent quality and temperature control to maximize yield and reproducibility. The process begins with the preparation of the sulfur-containing alkene substrate, which is stable and easy to handle, followed by the selection of the appropriate phenolic partner based on the desired R3 substituent. The activation step using TFAA must be performed at low temperatures (0°C) to control the exotherm and prevent degradation of the reactive sulfonium intermediate. Once the reaction is complete, typically after 24 hours, the isolation procedure is designed to be scalable, utilizing common organic solvents and standard separation techniques. For detailed operational parameters and specific embodiment data, please refer to the standardized synthesis guide below.
- Combine sulfur-containing alkene (Formula 2) and phenolic compound (Formula 3) in nitromethane solvent with a molar ratio of 1: 1.5.
- Add trifluoroacetic anhydride (TFAA) as the activating reagent at a molar ratio of 3: 1 relative to the alkene, maintaining the temperature at 0°C.
- Stir the reaction mixture under air atmosphere for 24 hours, followed by aqueous workup and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere chemical novelty. The elimination of transition metal catalysts removes a significant cost driver and supply chain vulnerability, as the availability and price of precious metals can be volatile. Furthermore, the simplified workflow reduces the number of unit operations required, leading to lower labor costs and reduced equipment occupancy time. This efficiency is crucial for maintaining competitive pricing structures in the generic and branded pharmaceutical markets. The robustness of the reaction conditions also implies a lower risk of batch failure, ensuring consistent supply continuity for downstream API manufacturing partners who rely on timely delivery of key intermediates.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the avoidance of pre-functionalization steps significantly lower the raw material costs associated with production. Additionally, the simplified purification process reduces the consumption of specialized resins and solvents, contributing to substantial cost savings in the overall manufacturing budget. The high atom economy of the direct coupling reaction ensures that a greater proportion of input materials are converted into valuable product, minimizing waste disposal costs and enhancing the overall economic viability of the process.
- Enhanced Supply Chain Reliability: The starting materials, including the sulfur-containing alkenes and various substituted phenols, are commercially available and inexpensive, reducing the risk of supply disruptions caused by scarce reagents. The ability to run the reaction under air atmosphere eliminates the need for complex inert gas infrastructure, allowing for greater flexibility in manufacturing site selection and operation. This accessibility ensures that production schedules can be maintained reliably, even in the face of global logistical challenges, providing a stable foundation for long-term supply agreements.
- Scalability and Environmental Compliance: The mild reaction conditions and lack of toxic heavy metals make this process inherently safer and more environmentally friendly, aligning with increasingly stringent global regulatory standards for green chemistry. The straightforward workup procedure generates less hazardous waste, simplifying compliance with environmental protection regulations and reducing the burden on waste treatment facilities. These factors collectively facilitate a smoother path to commercial scale-up, allowing manufacturers to rapidly increase production volumes from pilot scale to multi-ton annual capacity without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production pipelines. The answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented method utilizes a Pummerer reaction strategy activated by trifluoroacetic anhydride (TFAA), completely eliminating the need for expensive transition metal catalysts and avoiding heavy metal residue issues.
Q: What is the typical yield range for this reaction?
A: According to the patent data, the reaction consistently achieves yields of 42% and above, with many specific embodiments demonstrating yields exceeding 60-70% under optimized conditions.
Q: Can this method be scaled for industrial production?
A: Yes, the process operates under mild conditions (0°C) and uses readily available raw materials without stringent inert gas requirements, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfur-Containing Aryl Carbon Glycosides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Pummerer-based synthesis route for the production of next-generation SGLT2 inhibitor intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market-ready supply is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch conforms to the highest industry standards for pharmaceutical intermediates. Our state-of-the-art facilities are equipped to handle the specific solvent and temperature requirements of this chemistry, providing a secure and compliant environment for your proprietary projects.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and accelerate your time to market. Let us be your partner in turning this innovative chemistry into a commercial reality.
