Advanced Epoxide Synthesis Route for High-Purity HIV Protease Inhibitor Intermediates
Advanced Epoxide Synthesis Route for High-Purity HIV Protease Inhibitor Intermediates
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex antiviral agents, particularly those targeting HIV protease. Patent CN1066728C discloses a groundbreaking methodology for producing specific epoxide intermediates, which serve as critical building blocks for potent protease inhibitors like Compound J. This technology represents a significant departure from traditional linear syntheses, offering a streamlined approach that enhances both chemical efficiency and commercial viability. By utilizing a novel sequence involving allylation, halohydrin formation, and base-induced cyclization, manufacturers can achieve superior stereocontrol and yield. For R&D directors and procurement specialists, understanding this patented process is essential for securing a reliable HIV protease inhibitor intermediate supplier capable of meeting rigorous quality standards while optimizing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
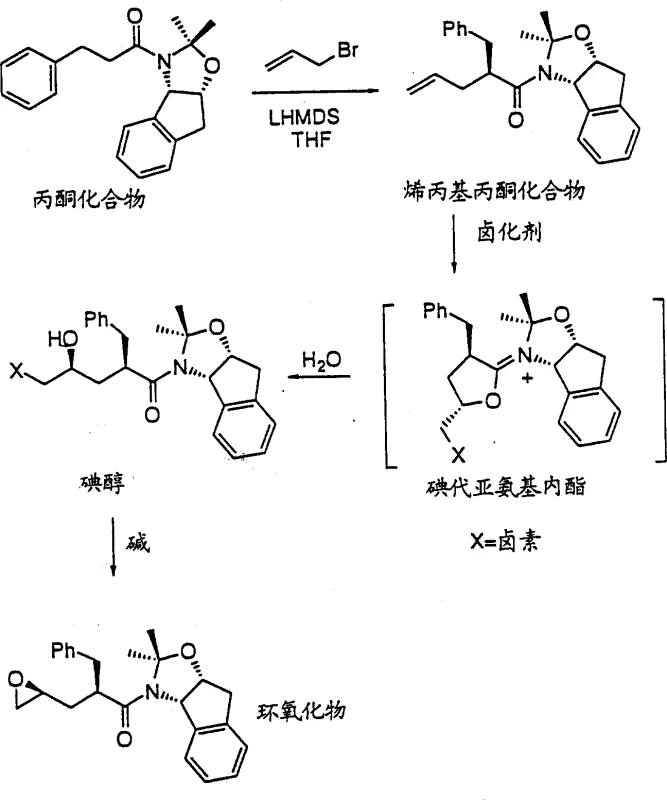
Prior art methods for synthesizing these crucial epoxide intermediates often relied on the reaction of acetonides with electrophilic reagents such as (S)-tosyl glycidyl ester. As illustrated in the reaction schemes of the background art, this conventional approach is fraught with inherent chemical challenges that hinder large-scale production. The primary issue arises from the reactivity of the epoxide product itself; under the highly basic conditions required for the initial coupling, the newly formed epoxide can react further with the acetonide anion. This side reaction leads to the formation of dimeric by-products, significantly reducing the isolated yield of the desired monomeric epoxide. Furthermore, the starting material, (S)-tosyl glycidyl ester, is an expensive chiral reagent that adds substantial cost to the bill of materials. The necessity for multiple recrystallization steps, such as using MTBE after methanol crystallization, to remove these dimeric impurities further complicates the workflow. Consequently, the overall yield from the acetonide starting material typically ranges only between 56% and 61%, making this route economically inefficient for commercial scale-up of complex pharmaceutical intermediates.
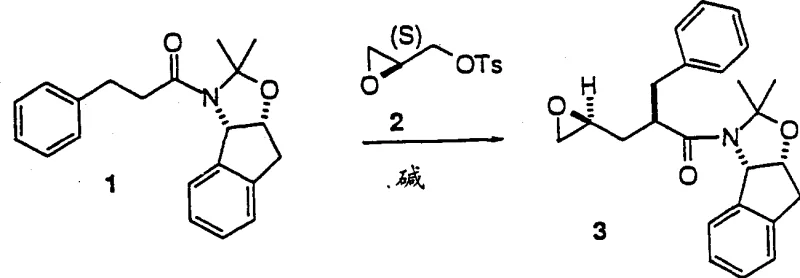
The Novel Approach
In stark contrast to the limitations of the tosylate method, the present invention introduces a robust three-step sequence that circumvents dimerization and utilizes more accessible reagents. The core innovation lies in the formation of a halohydrin intermediate from an allylacetone compound, followed by a base-induced cyclization to close the epoxide ring. This strategy avoids the use of reactive epoxide electrophiles in the early stages, thereby eliminating the risk of double addition by-products. The process begins with the allylation of an acetonide to form an allylacetone compound, which is then subjected to halogenation. Crucially, this halogenation step proceeds through a transient iodo-imino-lactone intermediate, which ensures high diastereoselectivity without substantial hydrolysis of the amide linkage. The final cyclization is triggered by a mild base, cleanly converting the halohydrin into the target epoxide. This novel pathway not only simplifies the purification protocol but also boosts the overall yield to surpass 70% from the acetonide starting material, representing a marked improvement in process efficiency and cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Halohydrin Formation and Cyclization
The success of this synthetic route hinges on the precise control of stereochemistry during the halohydrin formation step. When the allylacetone compound reacts with a halogenating agent, such as N-iodosuccinimide (NIS), in the presence of a weak base aqueous solution, it does not simply undergo random addition. Instead, the reaction likely proceeds through a cyclic iodo-imino-lactone species. This intramolecular participation directs the incoming nucleophile (water or hydroxide) to attack from a specific face, ensuring the formation of the desired stereoisomer with high fidelity. The patent data indicates that this transformation occurs effectively at temperatures ranging from -40°C to 100°C, with preferred conditions around 0°C to 35°C. The use of weak bases like sodium bicarbonate or basic alumina is critical here; they neutralize the acid generated during halogenation without promoting the hydrolysis of the sensitive amide bond or the premature opening of any forming rings. This mechanistic nuance allows for the isolation of the iodohydrin as a stable crystalline solid, which can be purified if necessary before the final cyclization step.
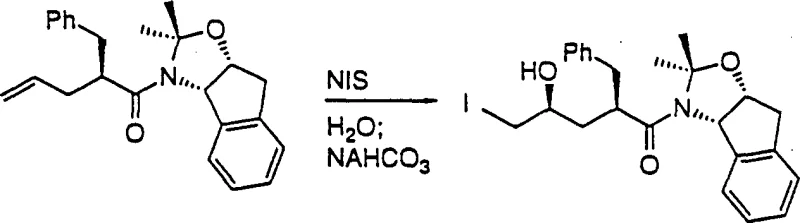
Following the isolation of the halohydrin, the final transformation involves a base-induced cyclization to generate the epoxide ring. This step is chemically straightforward yet vital for establishing the final stereochemical integrity of the molecule. By treating the halohydrin with an alkali metal hydroxide, such as lithium hydroxide or sodium hydroxide, or an alkoxide in a suitable solvent, an intramolecular SN2 reaction is triggered. The alkoxide oxygen attacks the carbon bearing the halogen leaving group, displacing the halide and closing the three-membered epoxide ring. The patent emphasizes that this cyclization can be performed in various solvent systems, including ethers, esters, and alcohols, providing flexibility for process optimization. Importantly, the conditions are mild enough to preserve the other functional groups present in the molecule, such as the acetonide protecting group and the amide linkage. This high level of chemoselectivity ensures that the final epoxide product is obtained with high purity, often exceeding 99% area by HPLC analysis after simple crystallization, thereby minimizing the need for costly chromatographic purification.
How to Synthesize Epoxide Intermediates Efficiently
The synthesis of these high-value epoxide intermediates is designed for operational simplicity and scalability, making it an attractive option for industrial production. The process leverages common organic solvents and reagents that are readily available in the global supply chain, reducing dependency on specialized or exotic chemicals. The initial allylation step utilizes strong bases like LHMDS in THF, a standard procedure in fine chemical synthesis. Subsequent steps involve aqueous workups and crystallizations that are easily adaptable to large reactor volumes. For detailed operational parameters, stoichiometry, and specific temperature profiles required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Perform allylation of the acetonide reactant using a strong base like LHMDS and an allyl halide to generate the allylacetone compound.
- React the allylacetone compound with a halogenating agent such as N-iodosuccinimide (NIS) in the presence of a weak base aqueous solution to form the halohydrin intermediate.
- Treat the isolated halohydrin with an alkali metal hydroxide or alkoxide in a solvent system to induce cyclization and form the final epoxide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic advantages beyond mere chemical elegance. The shift away from expensive chiral electrophiles like tosyl glycidyl ester to commodity reagents like allyl bromide and N-iodosuccinimide fundamentally alters the cost structure of the intermediate. This transition reduces the raw material cost burden and mitigates the risk associated with sourcing niche chiral building blocks. Furthermore, the improved yield profile means that less starting material is required to produce the same amount of final product, directly enhancing material efficiency. The robustness of the crystallization steps described in the patent ensures consistent product quality, which is critical for maintaining supply continuity in the pharmaceutical sector. By minimizing the formation of difficult-to-remove dimeric impurities, the process reduces the load on downstream purification units, leading to faster batch cycles and higher throughput.
- Cost Reduction in Manufacturing: The elimination of expensive chiral reagents and the avoidance of complex chromatographic purification steps lead to significant cost savings. The process utilizes commodity chemicals and achieves higher overall yields, which drastically lowers the cost per kilogram of the final epoxide intermediate. This economic efficiency is further amplified by the ability to recycle solvents like isopropyl acetate and methylcyclohexane, aligning with green chemistry principles and waste reduction goals.
- Enhanced Supply Chain Reliability: Relying on widely available reagents such as N-iodosuccinimide and allyl halides reduces supply chain vulnerability compared to sourcing specialized chiral epoxides. The robust nature of the reaction conditions, which tolerate a range of temperatures and solvent mixtures, ensures that production can continue even with minor variations in raw material quality. This resilience is vital for maintaining uninterrupted supply to downstream API manufacturers who depend on timely delivery of key intermediates.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing standard unit operations like liquid-liquid extraction and crystallization. The absence of heavy metal catalysts or hazardous reagents simplifies waste treatment and regulatory compliance. The high purity achieved through crystallization minimizes solvent consumption for purification, contributing to a lower environmental footprint and facilitating easier approval for commercial manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this epoxide synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: How does this new epoxide synthesis method improve upon conventional tosylate routes?
A: The conventional method using (S)-tosyl glycidyl ester suffers from dimerization by-products and requires expensive starting materials, resulting in lower overall yields (56-61%). The novel halohydrin route avoids these electrophilic issues, achieving over 70% overall yield with simpler purification.
Q: What are the critical reagents for ensuring high diastereoselectivity in this process?
A: The process relies on N-iodosuccinimide (NIS) combined with a weak base like sodium bicarbonate or basic alumina. This specific combination facilitates the formation of the iodo-imino-lactone intermediate, ensuring high diastereoselectivity during the halohydrin formation step without amide hydrolysis.
Q: Is this synthesis route scalable for commercial production of HIV inhibitors?
A: Yes, the patent describes robust conditions using common solvents like isopropyl acetate and THF, along with crystallization steps that avoid chromatography. The elimination of expensive chiral reagents and the use of stable intermediates make it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epoxide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antiviral therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our expertise in handling complex stereochemical transformations allows us to consistently produce epoxide intermediates that adhere to the highest industry standards, providing our partners with the confidence they need to advance their drug development pipelines.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this more efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive pharmaceutical market.
