Advanced Enantioselective Synthesis of (S)-Pregabalin for Commercial API Production
Introduction to Next-Generation Pregabalin Manufacturing
The pharmaceutical industry continuously seeks more efficient pathways for producing high-volume active pharmaceutical ingredients (APIs) like (S)-pregabalin, a critical treatment for neuropathic pain and epilepsy. Patent CN101848905A introduces a groundbreaking enantioselective process that fundamentally shifts the production paradigm from inefficient resolution methods to a streamlined chiral pool synthesis. This technology leverages the inherent chirality of D-mannitol bisacetonide to construct the core pyrrolidinone scaffold with high optical purity, bypassing the need for expensive chiral catalysts or stoichiometric resolving agents. By establishing the stereocenter at the very beginning of the synthesis, the method ensures that optical purity is maintained throughout the entire sequence, offering a robust solution for reliable pharmaceutical intermediate supplier networks aiming to optimize their production capabilities.
The significance of this innovation lies in its ability to address the twin challenges of cost and waste associated with traditional racemic synthesis. Conventional approaches often result in the discard of the unwanted R-enantiomer, capping theoretical yields at 50% and creating substantial environmental burdens. In contrast, this novel route maximizes atom economy and operational efficiency, providing a clear pathway for cost reduction in API manufacturing. For procurement and technical teams evaluating long-term supply strategies, understanding the mechanistic advantages of this patent is essential for securing a competitive edge in the global market for neurological therapeutics.
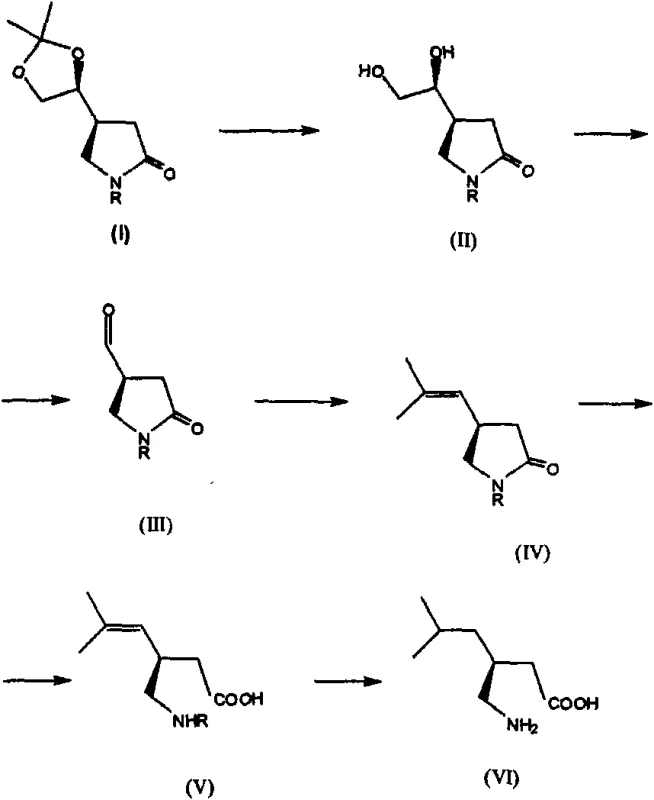
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of (S)-pregabalin has relied heavily on the synthesis of racemic mixtures followed by chiral resolution. Methods such as the malonate synthesis or Hofmann rearrangement typically generate a 1:1 mixture of R and S enantiomers, necessitating the use of chiral resolving agents to isolate the pharmacologically active S-form. This approach is inherently inefficient because it discards or requires complex recycling of the unwanted R-enantiomer, effectively limiting the maximum theoretical yield to 50%. Furthermore, the resolving agents themselves are often expensive, difficult to recover completely, and add significant processing steps to the manufacturing workflow. The reliance on transition metal catalysts in some alternative asymmetric routes, such as aluminum-salen complexes, introduces additional risks regarding heavy metal contamination and high reagent costs, which are detrimental to both economic viability and regulatory compliance in GMP environments.
The Novel Approach
The process described in CN101848905A circumvents these bottlenecks by employing a chiral pool strategy starting from D-mannitol bisacetonide. This abundant, renewable feedstock serves as the source of chirality, transferring its stereochemical information directly to the target molecule without the need for external chiral induction during the key bond-forming steps. The synthesis proceeds through a series of highly selective transformations, including oxidative cleavage, nitro-Michael addition, and cyclization, to generate a key chiral lactam intermediate. This intermediate retains the optical integrity of the starting material, allowing for the subsequent construction of the pregabalin side chain with predictable stereochemistry. By eliminating the resolution step entirely, this method not only doubles the potential yield compared to racemic routes but also simplifies the purification process, resulting in high-purity pregabalin with minimal impurity profiles.
Mechanistic Insights into Chiral Pool Synthesis
The core of this technology rests on the precise manipulation of the D-mannitol derivative to form the pyrrolidin-2-one ring system. The process begins with the oxidative cleavage of D-mannitol bisacetonide using sodium periodate, which generates the corresponding chiral aldehyde. This aldehyde then undergoes a Horner-Wadsworth-Emmons reaction or similar olefination to extend the carbon chain, followed by a conjugate addition of nitromethane. This nitro-Michael addition is a critical stereocontrolling step where the existing chiral center directs the incoming nucleophile to the correct face of the molecule, establishing the relative stereochemistry required for the final API. Subsequent reduction of the nitro group and spontaneous or catalyzed cyclization yields the protected chiral lactam, which serves as the versatile platform for the remainder of the synthesis.
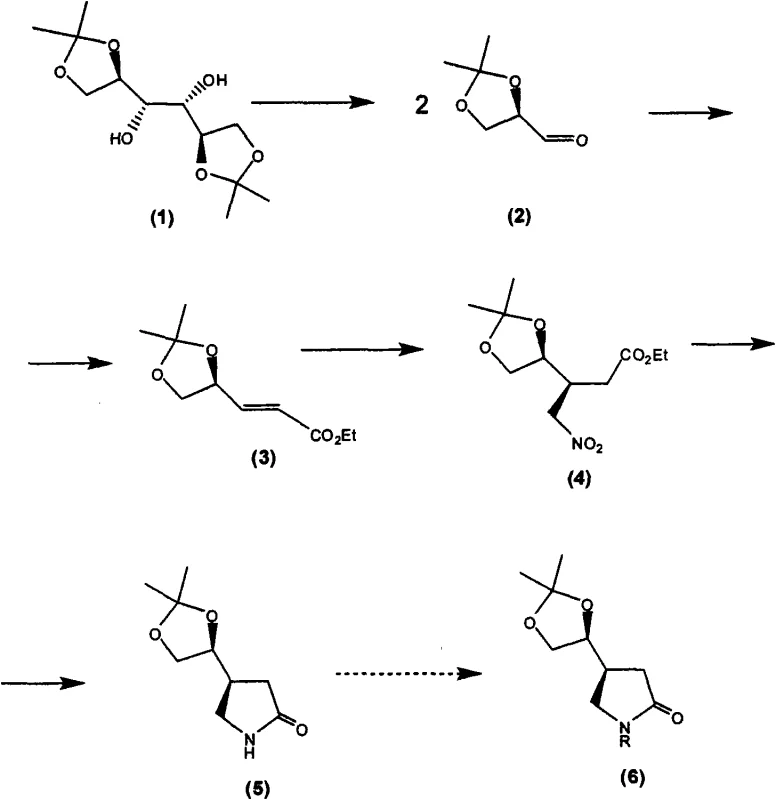
Following the formation of the chiral lactam, the synthetic route involves the strategic removal of the acetonide protecting group to reveal a diol, which is then oxidatively cleaved to an aldehyde. This aldehyde functionality allows for the introduction of the isobutyl side chain via Wittig olefination, creating an unsaturated intermediate. The final stages involve the hydrolytic opening of the lactam ring under basic conditions to form the amino acid backbone, followed by catalytic hydrogenation. This hydrogenation step serves a dual purpose: it reduces the olefinic double bond to the saturated isobutyl group and simultaneously removes any remaining protecting groups if present, delivering the final (S)-pregabalin molecule. The robustness of these reactions ensures that impurity formation is minimized, facilitating easier downstream processing and crystallization.
How to Synthesize (S)-Pregabalin Efficiently
Implementing this synthesis requires careful control of reaction conditions, particularly during the oxidation and hydrogenation steps, to maintain high enantiomeric excess. The protocol outlined in the patent provides a clear roadmap for converting the chiral lactam intermediate into the final API through a sequence of hydrolysis, olefination, and reduction. Detailed standard operating procedures for each transformation, including solvent choices, temperature ranges, and catalyst loadings, are critical for ensuring reproducibility at scale. For technical teams looking to adopt this methodology, the following guide summarizes the critical operational phases involved in the transformation.
- Prepare the chiral starting material (S)-4-((S)-2,2-dimethyl-1,3-dioxolan-4-yl)pyrrolidin-2-one from commercially available D-mannitol bisacetonide via oxidation and cyclization.
- Perform acidic hydrolysis of the dioxolane ring followed by oxidation to generate the key aldehyde intermediate, then utilize Wittig olefination to introduce the isobutyl side chain precursor.
- Execute basic hydrolysis of the lactam ring to open the structure, followed by catalytic hydrogenation to reduce the double bond and yield enantiomerically pure (S)-pregabalin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this enantioselective process offers transformative benefits for supply chain stability and cost management. Traditional resolution-based processes are vulnerable to fluctuations in the cost of resolving agents and the logistical challenges of managing large volumes of waste material. By shifting to a chiral pool synthesis, manufacturers can decouple their production economics from these volatile factors. The use of D-mannitol, a commodity chemical with a stable global supply, ensures that raw material availability remains consistent even during market disruptions. This reliability is paramount for commercial scale-up of complex pharmaceutical intermediates, where continuity of supply is often more valuable than marginal price differences.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps removes the need for expensive resolving agents and the associated processing costs of separating and recycling enantiomers. Additionally, the avoidance of precious metal catalysts like aluminum-salen complexes significantly lowers the bill of materials. The increased theoretical yield, potentially doubling that of racemic routes, means that less raw material is required to produce the same amount of API, driving down the cost per kilogram substantially. These efficiencies compound over large production volumes, resulting in significant margin improvements for the final drug product.
- Enhanced Supply Chain Reliability: Relying on D-mannitol bisacetonide as the chiral source mitigates risks associated with specialized reagent shortages. Since the starting material is derived from common sugars, its supply chain is diversified and resilient. Furthermore, the simplified process flow with fewer unit operations reduces the likelihood of batch failures and production delays. This streamlining allows for faster turnaround times and more predictable delivery schedules, which is crucial for reducing lead time for high-purity pharmaceutical intermediates in a just-in-time manufacturing environment.
- Scalability and Environmental Compliance: The reactions employed in this route, such as periodate oxidation and catalytic hydrogenation, are well-established in industrial chemistry and scale linearly without significant engineering hurdles. The reduction in waste generation, particularly the absence of discarded R-enantiomer salts, aligns with green chemistry principles and reduces the burden on waste treatment facilities. This environmental advantage not only lowers disposal costs but also simplifies regulatory filings related to environmental impact, facilitating smoother approvals for commercial manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios. The answers are derived directly from the technical specifications and advantageous effects described in the patent documentation.
Q: How does this enantioselective process improve yield compared to traditional resolution methods?
A: Traditional methods often synthesize racemic mixtures requiring resolution, which theoretically limits maximum yield to 50% and generates significant waste. This patented enantioselective route utilizes a chiral pool starting material (D-mannitol), ensuring that the stereochemistry is established early and maintained throughout, effectively doubling the theoretical yield potential and eliminating the need for costly chiral resolving agents.
Q: What are the cost advantages of using D-mannitol bisacetonide as a chiral source?
A: D-mannitol bisacetonide is a commercially available, commodity-grade chemical derived from renewable resources. Unlike expensive chiral catalysts (such as aluminum-salen complexes) or stoichiometric chiral auxiliaries required in other routes, this starting material is cost-effective and readily accessible, significantly reducing the raw material cost burden for large-scale API manufacturing.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process relies on robust, standard organic transformations such as oxidation, Wittig olefination, and catalytic hydrogenation. These reactions are well-understood in industrial settings and do not require exotic conditions or sensitive catalysts, making the route highly scalable and suitable for commercial production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Pregabalin Supplier
The technological advancements detailed in CN101848905A represent a significant leap forward in the efficient production of neurological therapeutics. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative pathways and possess the technical expertise to bring them to fruition. As a leading CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are translated into robust industrial realities. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (S)-pregabalin meets the highest global standards for safety and efficacy.
We invite pharmaceutical companies and procurement leaders to collaborate with us to leverage this advanced synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and enhance your competitive position in the global marketplace.
