Advanced Low-Valent Titanium Coupling for High-Purity Pharmaceutical Intermediates Manufacturing
Introduction to Next-Generation Intermediate Synthesis
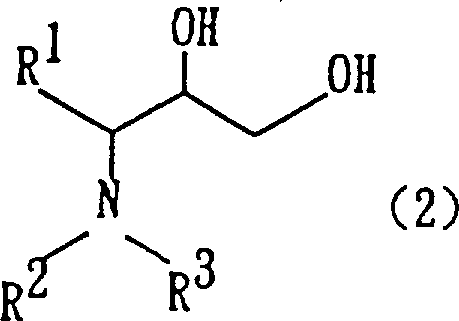
The pharmaceutical industry continuously seeks robust and economically viable pathways for synthesizing complex chiral building blocks, particularly those serving as critical precursors for antiretroviral therapies. Patent CN1116424A discloses a groundbreaking preparation method for 3-amino-2-hydroxy-1-propanol derivatives, represented by general formula (2), which are indispensable intermediates in the production of HIV protease inhibitors. This technology represents a significant paradigm shift from traditional azide-based or vanadium-mediated processes, introducing a highly efficient hetero-coupling reaction facilitated by low-valent titanium. By leveraging in situ generated titanium species from titanium tetrachloride and zinc, this method achieves superior stereocontrol and operational safety. For global procurement teams and R&D directors, understanding this mechanistic advancement is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials with reduced environmental impact and optimized cost structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-amino-2-hydroxy-1-propanol derivatives has relied on methodologies fraught with significant technical and economic drawbacks. One prominent conventional route involves the use of L-ascorbic acid or D-erythorbic acid as starting materials, necessitating the formation of hazardous azide compounds. This approach not only demands high-cost reagents but also involves a multitude of reaction steps, increasing the cumulative yield loss and operational complexity. Another known method utilizes vanadium trichloride as an indispensable reductant in coupling reactions; however, vanadium reagents are notoriously expensive and pose challenges in waste management. Furthermore, when synthesizing specific derivatives like N-tosylvaline exclusives, these older methods often suffer from poor stereoselectivity, leading to difficult separations of diastereomers and compromising the overall purity profile required for sensitive API manufacturing processes.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a low-valent titanium system to drive a direct hetero-coupling between protected amino aldehyde derivatives and formaldehyde sources. This methodology drastically simplifies the synthetic sequence by eliminating the need for dangerous azide intermediates and costly transition metal reductants like vanadium. The reaction proceeds under relatively mild conditions, typically ranging from -50°C to 0°C, using abundant and inexpensive metals such as zinc or magnesium to reduce titanium tetrachloride. This innovation not only shortens the reaction timeline but also inherently favors the formation of the desired erythro isomer, thereby streamlining downstream purification. For supply chain stakeholders, this translates to a more resilient production model with fewer bottlenecks and a significantly reduced dependency on volatile specialty chemical markets.
Mechanistic Insights into Low-Valent Titanium Mediated Coupling
The core of this technological breakthrough lies in the generation of reactive low-valent titanium species within the reaction medium. When titanium tetrachloride (TiCl4) is treated with a reducing metal like zinc (Zn) in aprotic solvents such as tetrahydrofuran (THF), it forms a highly reactive titanium complex capable of mediating carbon-carbon bond formation. The process begins with the activation of the formaldehyde source, which can be paraformaldehyde, trioxane, or aqueous formaldehyde, used in molar excess (typically 10 to 20 equivalents) to drive the equilibrium forward. The protected amino aldehyde derivative, shown in Formula (1), then undergoes a nucleophilic attack facilitated by the titanium center. This mechanism is distinct from radical-based couplings, offering a more controlled environment that preserves the integrity of sensitive functional groups while establishing the critical 1,2-diol motif with high fidelity.
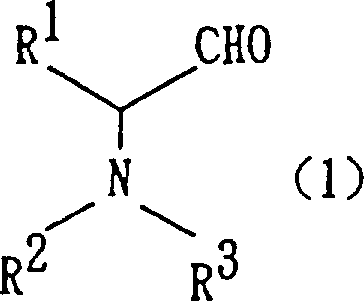
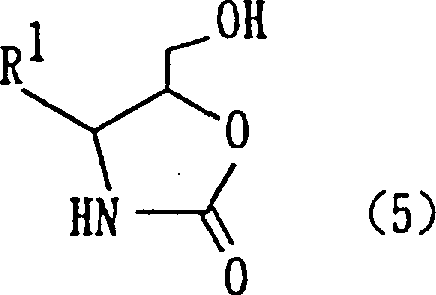
Furthermore, the stereoselectivity of this reaction is a critical parameter for R&D teams focusing on chiral drug synthesis. When optically active amino aldehydes are employed, the reaction predominantly yields the erythro configurational isomer of the 3-amino-2-hydroxy-1-propanol derivative. This stereochemical outcome is governed by the specific coordination geometry of the titanium intermediate and the steric bulk of the protecting groups, such as benzyloxycarbonyl or tert-butoxycarbonyl. Following the coupling, the resulting diol intermediate (Formula 4) can be seamlessly converted into 4-substituted-5-hydroxymethyloxazolidinone derivatives (Formula 5) via base-induced intramolecular cyclization. This versatility allows manufacturers to produce a wide array of downstream scaffolds from a single robust platform, enhancing the utility of the process for diverse pharmaceutical intermediate portfolios.
How to Synthesize 3-Amino-2-Hydroxy-1-Propanol Derivatives Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and stereoselectivity. The process typically involves generating the low-valent titanium species at sub-zero temperatures before introducing the aldehyde substrate. Detailed operational protocols regarding reagent addition rates, temperature ramping, and quenching procedures are essential for reproducibility. The standardized synthesis steps outlined below provide a framework for laboratory and pilot-scale execution, ensuring that the critical quality attributes of the final product are consistently met.
- Generate low-valent titanium in situ by reacting TiCl4 with Zinc powder in THF at temperatures between -50°C and 0°C.
- Add a formaldehyde source, such as paraformaldehyde (10-20 molar equivalents), to the reaction mixture containing the low-valent titanium species.
- Gradually introduce the protected amino aldehyde derivative (Formula 1) to the mixture, maintaining controlled temperatures to ensure stereoselective formation of the erythro-diol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this low-valent titanium coupling technology offers substantial strategic benefits for procurement managers and supply chain heads. By transitioning away from azide chemistry and expensive vanadium reductants, manufacturers can achieve significant cost reductions in raw material procurement. The use of commodity chemicals like zinc powder and titanium tetrachloride stabilizes the supply chain against the volatility associated with specialty reagents. Moreover, the simplified workup procedure, which involves basic filtration and extraction, reduces solvent consumption and waste treatment costs. This efficiency directly contributes to cost reduction in pharmaceutical intermediate manufacturing, allowing for more competitive pricing models without compromising on quality or regulatory compliance standards.
- Cost Reduction in Manufacturing: The elimination of high-cost reagents such as vanadium trichloride and hazardous azides fundamentally alters the cost structure of production. By utilizing inexpensive zinc and titanium tetrachloride, the direct material costs are drastically lowered. Additionally, the improved stereoselectivity reduces the burden on chiral separation processes, which are often the most expensive unit operations in fine chemical synthesis. This cumulative effect results in a leaner manufacturing process that delivers substantial economic value to the end customer.
- Enhanced Supply Chain Reliability: Reliance on readily available industrial commodities like zinc and THF ensures a stable supply of raw materials, mitigating the risk of production stoppages due to reagent shortages. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up, ensuring that delivery timelines for high-purity pharmaceutical intermediates are met consistently. This reliability is paramount for downstream API manufacturers who operate on tight Just-In-Time inventory models and cannot afford disruptions in their critical raw material flow.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-kilogram scales in the patent examples. The absence of toxic azide intermediates simplifies environmental, health, and safety (EHS) compliance, reducing the regulatory burden and facility upgrade costs associated with handling explosive or highly toxic substances. This green chemistry advantage facilitates smoother technology transfer to commercial-scale plants, ensuring long-term commercial scale-up of complex pharmaceutical intermediates is both feasible and sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this titanium-mediated synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and scope of the technology for potential partners. Understanding these details is key to evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of using low-valent titanium over traditional vanadium methods?
A: The low-valent titanium method eliminates the need for expensive and hazardous vanadium trichloride reductants. It offers superior stereoselectivity, predominantly yielding the erythro isomer required for HIV protease inhibitors, and utilizes readily available zinc and titanium tetrachloride, significantly reducing raw material costs and supply chain risks.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process avoids the formation of toxic azide intermediates found in conventional routes. The reaction conditions allow for straightforward workup procedures involving filtration and extraction, which effectively remove inorganic titanium salts and zinc residues, resulting in a cleaner crude product that requires less intensive purification to meet stringent pharmaceutical purity specifications.
Q: Can this synthesis route be scaled for commercial production of oxazolidinone derivatives?
A: Yes, the pathway is highly scalable. The subsequent cyclization of the diol intermediate to form oxazolidinone derivatives uses common bases like sodium hydride or potassium tert-butoxide under mild conditions. This two-step sequence (coupling followed by cyclization) is robust and adaptable to large-scale reactors, ensuring consistent supply continuity for downstream API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-2-Hydroxy-1-Propanol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 3-amino-2-hydroxy-1-propanol derivatives meets the exacting standards required by global regulatory bodies. Our commitment to excellence ensures that your supply chain remains robust, compliant, and efficient.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and optimize your manufacturing costs.
