Advanced N-Acyl Substituted NCA Technology for Scalable Peptide Intermediate Manufacturing
The pharmaceutical and agrochemical industries constantly seek more efficient pathways for synthesizing complex bioactive molecules, particularly peptides and optically active compounds. Patent CN1388804A introduces a groundbreaking class of amino acid-N-carboxy anhydrides featuring an N-acyl substituent at the nitrogen atom, addressing long-standing challenges in peptide synthesis. This innovation allows for high-yield production of amino acid derivatives without the cumbersome protection and deprotection steps traditionally required. By stabilizing the reactive anhydride moiety through N-acylation, the technology mitigates unwanted polymerization and racemization, offering a robust platform for industrial-scale manufacturing. This report analyzes the technical merits and commercial viability of this novel intermediate for global supply chains.
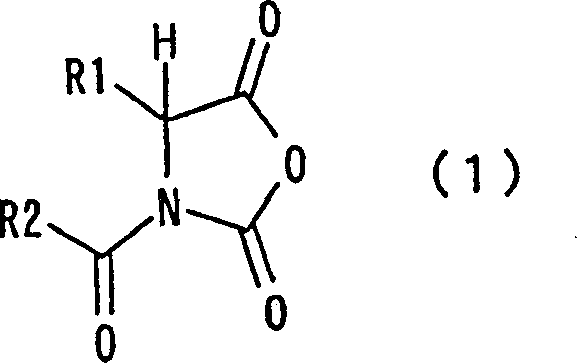
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional peptide synthesis methodologies often rely heavily on urethane-type protecting groups such as tert-butoxycarbonyl (Boc) or benzyloxycarbonyl (Z) to mask amino functionalities. While these groups prevent side reactions, their introduction and subsequent removal necessitate additional synthetic steps, each requiring expensive reagents and rigorous purification protocols. Furthermore, alkyl-type protecting groups can induce racemization, compromising the optical purity essential for pharmaceutical efficacy. The cumulative effect of these multi-step processes significantly inflates production costs and extends lead times, creating bottlenecks for large-scale industrial practices. Consequently, there is an urgent demand for streamlined approaches that maintain stereochemical integrity while reducing operational complexity.
The Novel Approach
The novel approach described in the patent utilizes N-acyl substituted NCAs to bypass the limitations of traditional protecting group strategies. By incorporating an acyl group directly onto the nitrogen atom of the N-carboxy anhydride, the molecule gains inherent stability against polymerization without needing external masking agents. This structural modification enables direct reaction with free amines or amino acids to form diamides efficiently. The process eliminates the distinct protection and deprotection stages, thereby condensing the synthetic route into fewer operational units. This simplification not only enhances overall yield but also reduces the consumption of solvents and reagents, aligning perfectly with modern green chemistry principles and cost-sensitive manufacturing environments.
Mechanistic Insights into N-Acyl Stabilization and Amidation
Mechanistically, the N-acyl substituent plays a critical role in modulating the reactivity of the N-carboxy anhydride ring. In unsubstituted NCAs, the free nitrogen lone pair can initiate unwanted polymerization chains, leading to complex mixtures that are difficult to purify. The electron-withdrawing nature of the acyl group reduces the nucleophilicity of the nitrogen, effectively suppressing these side reactions while preserving the electrophilicity of the carbonyl carbon necessary for amidation. This balance ensures that the reaction proceeds selectively towards the desired amide bond formation. Such control is vital for producing high-purity intermediates where even minor impurities can affect downstream biological activity or regulatory approval status.
Furthermore, the preservation of chiral integrity is a paramount concern in the synthesis of optically active compounds. Traditional activation methods often expose the alpha-carbon to conditions that promote racemization, resulting in a mixture of enantiomers that diminishes therapeutic value. The N-acyl NCA pathway operates under mild conditions that do not compromise the asymmetric center. Experimental data within the patent confirms that the resulting diastereomeric compounds can be obtained as single isomers with high remaining ratios. This capability to maintain stereochemical fidelity without aggressive reagents makes the technology particularly attractive for synthesizing complex peptide drugs where specific spatial configuration dictates function and safety profiles.
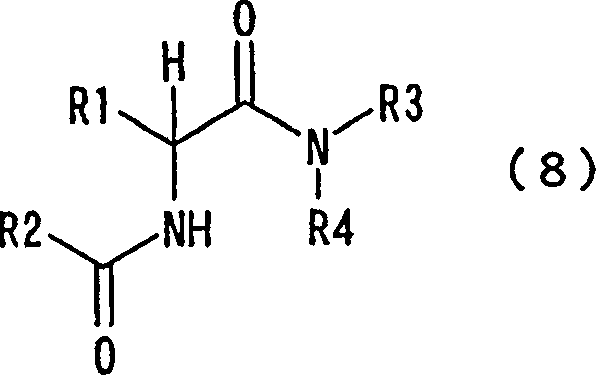
How to Synthesize N-Acyl NCA Efficiently
The synthesis of these valuable intermediates follows a straightforward protocol suitable for scale-up. The process involves reacting an amino acid N-carboxy anhydride with an acyl chloride or acid anhydride in the presence of a condensing agent or amine base. The reaction is typically conducted in inert diluents like ethyl acetate at controlled low temperatures to ensure stability. Detailed standardized synthesis steps see the guide below.
- Dissolve the amino acid N-carboxy anhydride in an inert diluent such as ethyl acetate and cool the solution in an ice bath.
- Add the acyl chloride compound to the mixture, followed by the dropwise addition of an amine base catalyst like 4-dimethylaminopyridine.
- Stir the reaction mixture at low temperature, filter off precipitated salts, and concentrate the filtrate to isolate the pure N-acyl NCA crystals.
Commercial Advantages for Procurement and Supply Chain Teams
This technology offers substantial strategic advantages for procurement and supply chain management by fundamentally simplifying the production workflow. The elimination of protecting group manipulation translates directly into reduced raw material consumption and lower waste disposal costs. By shortening the synthetic sequence, manufacturers can achieve faster throughput and improved asset utilization rates. This efficiency gain is critical for maintaining competitive pricing in the global market for fine chemical intermediates. Additionally, the robustness of the N-acyl NCA intermediates enhances storage stability, reducing the risk of degradation during logistics and warehousing operations.
- Cost Reduction in Manufacturing: The primary economic driver is the removal of separate protection and deprotection unit operations. Each eliminated step saves on labor, energy, and specialized reagents like carbodiimides or uronium salts. This structural simplification allows for significant cost savings in peptide intermediate manufacturing without compromising quality. The process avoids the use of expensive urethane-type protecting groups, further lowering the bill of materials. Overall, the streamlined route offers a more economical pathway for producing high-value amino acid derivatives.
- Enhanced Supply Chain Reliability: Starting materials for this synthesis, such as simple amino acid NCAs and acyl chlorides, are readily available from established chemical suppliers. This accessibility reduces dependency on niche reagents that often suffer from long lead times or supply volatility. The simplified process also lowers the technical barrier for contract manufacturing organizations, expanding the potential supplier base. Consequently, buyers can secure more reliable supply contracts with reduced risk of disruption due to raw material shortages or complex synthesis failures.
- Scalability and Environmental Compliance: The reaction conditions are mild and utilize common organic solvents that are easy to recover and recycle. The absence of heavy metal catalysts or hazardous azide reagents simplifies waste treatment and environmental compliance procedures. This makes the process highly scalable from kilogram to multi-ton production levels without encountering significant safety hurdles. The reduced generation of chemical waste aligns with increasingly stringent environmental regulations, ensuring long-term operational sustainability for manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of N-acyl NCA technology. These answers are derived directly from the patented methodology and its demonstrated benefits in industrial applications. Understanding these aspects helps stakeholders evaluate the feasibility of adopting this route for their specific product portfolios. The information provided ensures clarity on performance metrics and operational requirements.
Q: How does N-acyl substitution prevent polymerization in NCAs?
A: The acyl group reduces the nucleophilicity of the nitrogen atom, preventing it from initiating unwanted polymerization chains while maintaining reactivity towards amines.
Q: Does this method cause racemization of chiral amino acids?
A: No, the reaction conditions are mild and specifically designed to preserve the asymmetric center, ensuring high optical purity of the resulting diamides.
Q: What are the cost benefits compared to Boc/Z protection methods?
A: It eliminates the separate protection and deprotection steps, significantly reducing reagent consumption, solvent use, and overall processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amino Acid N-Carboxy Anhydride Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced peptide synthesis technologies like the N-acyl NCA pathway. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle sensitive anhydride chemistry with stringent purity specifications and rigorous QC labs to ensure every batch meets global regulatory standards. We understand the critical nature of chiral intermediates in drug development and commit to delivering consistent quality.
We invite you to collaborate with us to leverage this cost-effective technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can optimize your supply chain. Let us help you accelerate your time to market with reliable, high-quality pharmaceutical intermediates.
