Advanced Benzyne-Mediated Cyclization for Commercial Scale-Up of Complex Indole Intermediates
Advanced Benzyne-Mediated Cyclization for Commercial Scale-Up of Complex Indole Intermediates
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing privileged scaffolds, particularly indole derivatives which serve as the core structure for numerous bioactive molecules. Patent CN101503387B introduces a groundbreaking preparation method for 3-aryl substituted indoles containing an ester group at the 2-position, addressing critical bottlenecks in traditional synthetic routes. This technology leverages a benzyne-mediated [3+2] cycloaddition strategy that operates under remarkably mild conditions, typically between 40°C and 80°C, avoiding the harsh thermal or acidic environments often required by legacy methods. For R&D directors and process chemists, this represents a significant leap forward in controlling reaction selectivity and minimizing degradation pathways. By utilizing o-trimethylsilyl phenyl trifluoromethanesulfonate as a benzyne precursor alongside 2-azido-3-aryl-acrylic acid esters, the process achieves high conversion rates with exceptional operational simplicity. The implications for supply chain stability are profound, as the reliance on exotic catalysts is eliminated in favor of readily available reagents like cesium fluoride and triphenylphosphine. This report analyzes the technical merits and commercial viability of this novel approach, positioning it as a preferred route for the reliable pharmaceutical intermediate supplier seeking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indole nucleus has relied heavily on classical transformations such as the Fischer indole synthesis or the Hemetsberger-Knittel reaction, both of which present significant challenges for modern process chemistry. The Fischer synthesis, while versatile, often necessitates strongly acidic conditions and high temperatures that can be incompatible with sensitive functional groups like esters or halogens found in advanced intermediates. Furthermore, the Hemetsberger-Knittel indole synthesis, which involves the thermal decomposition of 2-azido-3-aryl-acrylates, typically requires refluxing in high-boiling solvents such as xylene or chlorobenzene to drive the nitrogen extrusion and cyclization. 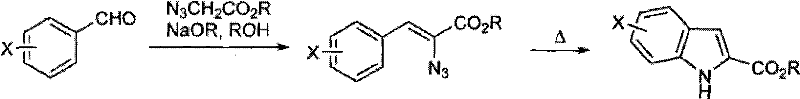 These harsh thermal conditions frequently lead to the formation of complex impurity profiles, including polymerized by-products and decomposed starting materials, which complicate downstream purification and reduce overall yield. Additionally, the safety hazards associated with heating organic azides to high temperatures cannot be overstated, posing risks for commercial scale-up of complex heterocycles. The inability to fine-tune the reaction environment often results in batch-to-batch variability, creating uncertainty for procurement managers who require consistent quality and predictable lead times. Consequently, there is a pressing need for alternative methodologies that can deliver high-purity indole derivatives without compromising safety or operational efficiency.
These harsh thermal conditions frequently lead to the formation of complex impurity profiles, including polymerized by-products and decomposed starting materials, which complicate downstream purification and reduce overall yield. Additionally, the safety hazards associated with heating organic azides to high temperatures cannot be overstated, posing risks for commercial scale-up of complex heterocycles. The inability to fine-tune the reaction environment often results in batch-to-batch variability, creating uncertainty for procurement managers who require consistent quality and predictable lead times. Consequently, there is a pressing need for alternative methodologies that can deliver high-purity indole derivatives without compromising safety or operational efficiency.
The Novel Approach
In stark contrast to these thermal methods, the technology disclosed in CN101503387B utilizes a sophisticated benzyne interception strategy that proceeds under significantly milder conditions, typically ranging from 40°C to 80°C. This novel approach involves the in situ generation of benzyne from o-trimethylsilyl phenyl triflate using cesium fluoride as a fluoride source, which then undergoes a concerted [3+2] cycloaddition with the 2-azido-3-aryl-acrylic acid ester dipole. 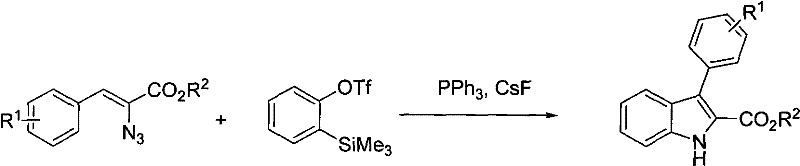 This mechanistic shift allows for precise control over the reaction kinetics, effectively suppressing the thermal decomposition pathways that plague traditional azide chemistry. The use of a mixed solvent system comprising toluene and acetonitrile further enhances solubility and reaction homogeneity, facilitating efficient heat transfer and mass transport during the exothermic addition phase. From a manufacturing perspective, this translates to a drastic simplification of the process workflow, where the reaction can be completed within 3 to 8 hours followed by a straightforward cold filtration to remove triphenylphosphine oxide. The ability to operate at lower temperatures not only improves safety but also preserves the integrity of sensitive ester functionalities, ensuring that the final product meets stringent purity specifications required for API manufacturing. This methodology exemplifies how modern organic synthesis can overcome historical limitations to deliver superior process economics and reliability.
This mechanistic shift allows for precise control over the reaction kinetics, effectively suppressing the thermal decomposition pathways that plague traditional azide chemistry. The use of a mixed solvent system comprising toluene and acetonitrile further enhances solubility and reaction homogeneity, facilitating efficient heat transfer and mass transport during the exothermic addition phase. From a manufacturing perspective, this translates to a drastic simplification of the process workflow, where the reaction can be completed within 3 to 8 hours followed by a straightforward cold filtration to remove triphenylphosphine oxide. The ability to operate at lower temperatures not only improves safety but also preserves the integrity of sensitive ester functionalities, ensuring that the final product meets stringent purity specifications required for API manufacturing. This methodology exemplifies how modern organic synthesis can overcome historical limitations to deliver superior process economics and reliability.
Mechanistic Insights into Benzyne-Mediated Cycloaddition
The core of this technological advancement lies in the elegant generation and trapping of the benzyne intermediate, a highly reactive species that serves as the electrophilic partner in the cyclization cascade. Upon addition of cesium fluoride to the reaction mixture, the fluoride ion selectively attacks the silicon atom of the o-trimethylsilyl phenyl triflate, triggering the elimination of the triflate group and the formation of the strained benzyne triple bond. This transient intermediate is immediately intercepted by the 1,3-dipolar character of the azido group on the acrylic acid ester derivative. The resulting cycloaddition forms a triazoline intermediate which rapidly extrudes nitrogen gas to yield the aromatic indole system. This concerted mechanism is highly favorable entropically and enthalpically under the specified conditions, driving the reaction to completion with minimal energy input. For the R&D team, understanding this mechanism is crucial for troubleshooting and optimization, as it highlights the importance of maintaining anhydrous conditions to prevent benzyne quenching by moisture. The stoichiometry of the reagents, specifically the 1:1 molar ratio of triphenylphosphine to the azido-ester, plays a subtle yet vital role in stabilizing the reaction environment and scavenging potential radical species that could lead to oligomerization.
Impurity control in this process is inherently superior due to the specificity of the benzyne-azide interaction and the mild thermal profile. Unlike acid-catalyzed rearrangements which can promote skeletal isomerizations or ester hydrolysis, this neutral cyclization leaves the ester moiety at the 2-position intact. The primary by-product, triphenylphosphine oxide, is a solid that precipitates out of the solution upon cooling, allowing for its removal via simple filtration before concentration. This physical separation step significantly reduces the burden on chromatographic purification, thereby increasing the overall throughput and reducing solvent consumption. Furthermore, the absence of transition metal catalysts eliminates the risk of heavy metal contamination, a critical parameter for regulatory compliance in pharmaceutical production. The robustness of this mechanism against various substituents on the aryl ring (such as methyl, chloro, or nitro groups) demonstrates its versatility, making it a powerful tool for generating diverse libraries of indole analogs for drug discovery programs. This level of chemical precision ensures that the final active pharmaceutical ingredient maintains a clean impurity profile, facilitating faster regulatory approval and market entry.
How to Synthesize 3-Aryl-2-Carboxyindole Efficiently
The practical implementation of this synthesis route is designed for ease of execution, requiring standard laboratory equipment and commonly available reagents. The process begins by dissolving the 2-azido-3-aryl-acrylic acid ester and the benzyne precursor in a optimized mixture of toluene and acetonitrile, ensuring complete solubilization prior to reaction initiation. This solution is then added dropwise over a period of 30 minutes to a stirred suspension of cesium fluoride and triphenylphosphine, maintaining the temperature strictly within the 40°C to 80°C window to control the rate of benzyne generation.
- Dissolve 2-azido-3-aryl-acrylic acid ester and o-trimethylsilyl phenyl triflate in a mixed solvent of toluene and acetonitrile.
- Add the solution dropwise to a reaction vessel containing cesium fluoride and triphenylphosphine in toluene/acetonitrile at 40-80°C.
- Maintain temperature for 3-8 hours, filter cold, concentrate filtrate, and purify via column chromatography to obtain the target indole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of expensive transition metal catalysts, such as palladium or rhodium complexes often used in cross-coupling approaches to similar structures, results in substantial cost savings on raw material acquisition. Moreover, the simplified workup procedure, which relies on filtration rather than complex aqueous extractions or scavenger treatments, drastically reduces processing time and labor costs. The use of commodity solvents like toluene and acetonitrile ensures that supply chain disruptions are minimized, as these materials are globally sourced and readily available in bulk quantities. This reliability is essential for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream API manufacturers. Additionally, the high atom economy of the cycloaddition reaction minimizes waste generation, aligning with increasingly stringent environmental regulations and reducing disposal costs associated with hazardous chemical waste.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for precious metal catalysts and their associated removal resins, which are often major cost drivers in fine chemical synthesis. The mild reaction conditions also translate to lower energy consumption for heating and cooling, further enhancing the overall economic efficiency of the manufacturing campaign. By simplifying the purification train through the precipitation of triphenylphosphine oxide, the yield loss typically associated with multiple chromatographic steps is avoided, maximizing the output per batch. These cumulative efficiencies allow for a more competitive pricing structure for the final high-purity indole derivatives, providing a strategic advantage in the marketplace.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents such as o-trimethylsilyl phenyl triflate and cesium fluoride mitigates the risk of supply interruptions caused by the instability of diazo compounds or sensitive organometallics. The robustness of the reaction to minor variations in temperature and stoichiometry ensures consistent batch quality, reducing the frequency of failed batches and the need for reprocessing. This predictability enables supply chain planners to forecast inventory levels with greater accuracy and reduce safety stock requirements. Furthermore, the scalability of the process from gram to kilogram scale has been demonstrated without significant modification, ensuring a seamless transition from pilot plant to commercial production facilities.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies the environmental permitting process and reduces the regulatory burden associated with wastewater treatment and residue analysis. The solvent system used is amenable to recycling and recovery, supporting sustainable manufacturing practices and reducing the overall carbon footprint of the synthesis. The safe handling of azides at moderate temperatures minimizes occupational health and safety risks, fostering a safer working environment for plant operators. These factors collectively contribute to a more resilient and sustainable supply chain capable of adapting to evolving market demands and regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology, providing clarity for stakeholders evaluating its adoption.
Q: What are the primary advantages of this benzyne-mediated method over traditional Fischer indole synthesis?
A: Unlike Fischer synthesis which requires strong acids and specific ketone substrates, this method utilizes mild neutral conditions (40-80°C) and tolerates a wide range of functional groups including esters and halogens, resulting in higher purity and fewer side reactions.
Q: How does this process impact impurity profiles for pharmaceutical applications?
A: The reaction generates triphenylphosphine oxide as the main by-product, which is easily removed via cold filtration. The mild temperature profile prevents thermal decomposition of the sensitive azido-intermediate, ensuring a cleaner crude product and simplified downstream purification.
Q: Is this synthetic route suitable for large-scale manufacturing?
A: Yes, the process uses commercially available solvents (toluene, acetonitrile) and reagents. The exothermic nature is manageable via controlled dropwise addition, and the absence of expensive transition metal catalysts makes it highly economically viable for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl-2-Carboxyindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates that adhere to the most rigorous standards of purity and consistency. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We leverage our state-of-the-art rigorous QC labs to verify that every batch of 3-aryl-2-carboxyindole meets stringent purity specifications, guaranteeing performance in your downstream synthetic applications. Our commitment to excellence extends beyond mere compliance; we actively collaborate with our partners to optimize processes for maximum yield and minimal environmental impact, embodying the principles of green chemistry in every operation.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain to drive value and efficiency. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your production volume and quality requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your competitive position in the global pharmaceutical market. Let us be your trusted partner in navigating the complexities of modern chemical manufacturing.
