Revolutionizing 10-DAB III Synthesis: A Commercial Scale-Up Guide for Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust pathways to secure critical anticancer precursors, and patent CN1538963A presents a transformative approach to producing 10-deacetylbaccatin III (10-DAB III). This pivotal intermediate serves as the foundational scaffold for the semi-synthesis of paclitaxel and docetaxel, two of the most potent chemotherapy agents globally. The disclosed technology departs from traditional alcoholic solvent systems, introducing a counter-intuitive yet highly effective methodology utilizing acetate solvents such as isobutyl acetate and isopropyl acetate. By leveraging the unique reactivity of hydrazine hydrate within these specific solvent environments, the process achieves rapid selective deesterification of complex taxane mixtures derived from renewable biomass. This innovation addresses long-standing bottlenecks in yield and processing time, offering a compelling value proposition for manufacturers aiming to optimize their supply chains for high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the conversion of taxane-rich biomass extracts into 10-DAB III has relied heavily on alcoholic solvents like methanol or ethanol during the hydrazinolysis step. While chemically feasible, these conventional routes suffer from significant operational inefficiencies that impact overall throughput and cost structures. The reaction kinetics in alcoholic media are notoriously sluggish, often requiring extended reaction times exceeding three hours to reach completion, which ties up reactor capacity and limits production volume. Furthermore, the homogeneity of the reaction mixture in alcohols complicates the downstream workup, necessitating complex extraction sequences and additional solvent swaps to isolate the product. The presence of high levels of total dissolved solids (TDS) in these alcoholic solutions often leads to darker reaction masses and increased impurity profiles, demanding rigorous and costly purification steps to meet the stringent quality standards required for pharmaceutical starting materials.
The Novel Approach
In stark contrast, the methodology outlined in CN1538963A utilizes acetate solvents that fundamentally alter the reaction landscape to favor speed and simplicity. Although one might theoretically expect the ester functionality of the solvent to compete with the taxane substrate for the hydrazine reagent, experimental data demonstrates a surprising acceleration of the desired transformation. The reaction proceeds to completion in approximately 45 minutes at room temperature, representing a drastic reduction in cycle time compared to alcoholic counterparts. Moreover, the interaction between the solvent and hydrazine facilitates the spontaneous formation of a biphasic system, creating a distinct organic layer containing the product and an aqueous layer containing byproducts. This physical separation eliminates the need for extensive extraction protocols, allowing for direct isolation of the organic phase and significantly streamlining the path from crude biomass extract to purified 10-DAB III.
Mechanistic Insights into Selective Hydrazinolysis in Acetate Media
The core chemical breakthrough lies in the selective cleavage of ester functionalities at the C-10 and C-13 positions of the taxane skeleton using hydrazine hydrate. In the context of acetate solvents, the mechanism involves a nucleophilic attack by the hydrazine on the carbonyl carbons of the ester groups attached to the taxane core. Despite the solvent itself possessing ester groups that are susceptible to hydrazinolysis, the reaction conditions are tuned such that the conversion of unwanted taxanes like cephalomannine and 10-deacetyltaxol into 10-DAB III is highly efficient. The steric and electronic environment provided by solvents like isobutyl acetate appears to stabilize the transition state or enhance the local concentration of reactive species, driving the deesterification forward rapidly. This selectivity is crucial because it preserves the integrity of the sensitive oxetane ring and other functional groups essential for the biological activity of the final drug product, ensuring that the resulting intermediate is chemically robust for subsequent synthetic steps.
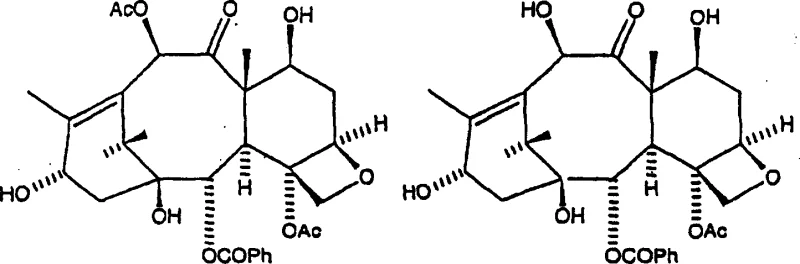
Controlling the impurity profile is another critical aspect of this mechanistic pathway, particularly regarding the formation of stereoisomers. Extended reaction times beyond the optimal window can lead to the generation of 10-DAB V, a 7-position stereoisomer that complicates purification. The accelerated kinetics of the acetate solvent system allow operators to hit the peak conversion point quickly and quench or separate the reaction before significant degradation occurs. Additionally, the biphasic nature of the system aids in impurity management; hydrazine salts and polar byproducts partition preferentially into the aqueous phase, leaving the organic layer enriched with the desired taxane. This inherent purification effect reduces the burden on downstream chromatography or crystallization steps, resulting in a final product with superior HPLC area purity, often exceeding 95%, which is vital for meeting the rigorous specifications of global regulatory bodies.
How to Synthesize 10-DAB III Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and reaction monitoring to maximize yield and purity. The process begins with dissolving the taxane-containing biomass extract in the chosen acetate solvent, followed by the controlled addition of hydrazine hydrate. Operators must maintain the reaction at room temperature and monitor the disappearance of starting materials like 10-deacetyltaxol to determine the precise endpoint. The detailed standardized synthesis steps, including specific concentrations, washing protocols, and crystallization parameters, are provided in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Prepare a biomass extract solution containing taxanes in an acetate solvent such as isobutyl acetate or isopropyl acetate, concentrating to optimal dissolved solid ratios.
- Contact the solution with hydrazine hydrate at room temperature, allowing the mixture to stir for approximately 45 minutes to facilitate selective deesterification at C-10 and C-13 positions.
- Separate the resulting biphasic mixture, wash the organic layer, treat with activated carbon, and crystallize the product using acetonitrile as an anti-solvent to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this acetate-based hydrazinolysis process translates into tangible operational efficiencies and risk mitigation. The primary advantage stems from the drastic simplification of the workflow, which removes multiple unit operations associated with solvent exchange and complex extractions. By reducing the number of processing steps, the facility lowers its consumption of utilities and auxiliary chemicals, directly impacting the variable cost of goods sold. Furthermore, the ability to utilize unpurified or partially purified biomass extracts as feedstock means that manufacturers can source raw materials from a broader range of suppliers without incurring the high costs of pre-purification. This flexibility enhances supply chain resilience, allowing companies to adapt to fluctuations in biomass availability while maintaining consistent output of high-purity 10-DAB III.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and time-consuming purification stages typically required when using alcoholic solvents. Since the reaction naturally separates into two phases, the need for large volumes of extraction solvents and the energy-intensive distillation steps to remove them is significantly diminished. Additionally, the shorter reaction time increases reactor turnover rates, allowing existing infrastructure to produce more batches per year without capital expansion. The reduction in total dissolved solids also minimizes waste generation, lowering the costs associated with hazardous waste disposal and environmental compliance, thereby contributing to a leaner and more sustainable manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing high-quality 10-DAB III is often constrained by the seasonal and geographical limitations of yew biomass harvesting. This technology mitigates those risks by enabling the efficient conversion of lower-grade biomass fractions and side-stream products that were previously considered waste. By unlocking value from these abundant but underutilized resources, suppliers can stabilize inventory levels and reduce dependency on scarce, high-purity natural extracts. The robustness of the chemical process ensures that variations in the raw material composition do not critically impact the final yield, providing buyers with a more predictable and reliable supply of this critical pharmaceutical intermediate throughout the year.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the use of common, industrially available solvents like isobutyl acetate and ethyl acetate, which are already approved for use in pharmaceutical manufacturing. The absence of exotic catalysts or extreme reaction conditions simplifies the engineering requirements for large-scale reactors, reducing the barrier to entry for contract development and manufacturing organizations. From an environmental perspective, the process generates less solvent waste and avoids the use of heavy metals or toxic reagents often found in alternative synthetic routes. This alignment with green chemistry principles not only satisfies regulatory demands but also appeals to end-users who are increasingly prioritizing sustainability in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 10-DAB III using this patented hydrazinolysis method. These answers are derived directly from the experimental data and embodiments described in the patent documentation, providing clarity on reaction specifics, purity outcomes, and scalability potential. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: Why are acetate solvents preferred over alcohols for 10-DAB III hydrazinolysis?
A: Acetate solvents like isobutyl acetate create a biphasic system that simplifies workup and significantly reduce reaction time to about 45 minutes compared to several hours with alcohol solvents, while maintaining high conversion rates.
Q: Can this process handle unpurified biomass extracts?
A: Yes, the method is specifically designed to convert complex mixtures of taxanes found in biomass extracts, including cephalomannine and 10-deacetyltaxol, directly into 10-DAB III without requiring prior HPLC purification.
Q: What is the expected purity of the final 10-DAB III product?
A: Following the described crystallization and purification steps, the process yields 10-DAB III with greater than 95% HPLC area purity, making it suitable for subsequent semi-synthesis of paclitaxel and docetaxel.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-DAB III Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of high-quality taxane intermediates for the global oncology market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering 10-DAB III that meets stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay every batch. By leveraging advanced process technologies like the acetate-based hydrazinolysis method, we can offer competitive pricing without compromising on the quality standards required for GMP manufacturing of life-saving medications.
We invite potential partners to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new analogues, we are ready to provide the data-driven insights you need. Contact us today to explore how our optimized production of 10-DAB III can support your long-term strategic goals and ensure continuity in your pharmaceutical development pipeline.
