Advanced Synthesis of Branched Polyhydroxypyrrolidine Derivatives for Commercial API Production
Advanced Synthesis of Branched Polyhydroxypyrrolidine Derivatives for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic scaffolds, particularly those mimicking natural sugar structures for enzyme inhibition. Patent CN100537584C discloses a groundbreaking methodology for the preparation of branched polyhydroxypyrrolidine derivatives, which serve as critical intermediates in the synthesis of imino sugars and polyhydroxy prolines. These compounds are renowned for their potent glycosidase and glycosyltransferase inhibitory activities, finding applications in treating metabolic disorders such as diabetes, analogous to drugs like Miglitol. The disclosed technology overcomes historical limitations in synthesizing highly substituted pyrrolidine rings by utilizing an iterative nitrone chemistry approach. This strategy enables the precise installation of diverse substituents at the 2 and 5 positions of the pyrrolidine ring, offering unprecedented flexibility in molecular design for drug discovery teams.
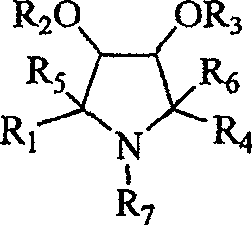
From a supply chain perspective, the ability to access these complex architectures through a general and versatile method represents a significant strategic advantage. The patent outlines a pathway that transforms readily available sugar-derived hydroxylamines into high-value branched structures through oxidation and nucleophilic addition cycles. This capability allows manufacturers to produce a wide library of analogues without developing entirely new synthetic routes for each variant. For procurement managers and R&D directors alike, understanding the nuances of this chemistry is essential for securing a reliable supply of these specialized pharmaceutical intermediates. The following analysis details the technical superiority of this approach and its implications for cost-effective, large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted polyhydroxy pyrroles has been fraught with significant challenges that hindered their widespread adoption in commercial drug manufacturing. Prior art methods, such as those described by Deng et al. in 2001, were severely restricted in scope, often limited to synthesizing only methyl-substituted variants, which failed to address the need for lipophilic modifications required for optimal protein binding. Furthermore, alternative approaches involving the addition of imines to Grignard reagents within a glycosyl ring, as proposed by B.G. Davis in 2002, frequently suffered from unacceptably low yields and poor stereoselectivity. These inefficiencies translate directly into higher production costs and extended lead times, creating bottlenecks for supply chain heads who require consistent, high-volume output. The inability to introduce diverse hydrophobic groups effectively also limits the biological potency of the resulting candidates, forcing R&D teams to screen larger libraries with lower success rates.
The Novel Approach
The methodology presented in CN100537584C revolutionizes this landscape by introducing a modular, iterative synthesis strategy centered on nitrone intermediates. By oxidizing a hydroxylamine to a nitrone and subsequently reacting it with various organometallic reagents, the process allows for the stepwise construction of highly branched structures with exceptional control. This approach is not limited to simple alkyl groups; it accommodates a vast range of substituents including C1-C20 alkyls, aryls, vinyls, and even protected amino acid side chains. The reaction conditions are notably mild, typically proceeding in common organic solvents like tetrahydrofuran or dichloromethane at temperatures ranging from cryogenic -80°C to ambient room temperature. This flexibility ensures that the process can be adapted for different target molecules without requiring hazardous reagents or extreme pressures, thereby enhancing operational safety and reducing the complexity of reactor engineering for commercial scale-up.
Mechanistic Insights into Nitrone-Based Iterative Synthesis
The core of this innovative synthesis lies in the reversible and highly selective chemistry of nitrones. The process initiates with the oxidation of a glycosyl hydroxylamine precursor using mild oxidizing agents such as manganese dioxide, Oxone, or hydrogen peroxide. This transformation generates a reactive nitrone species, which acts as a dipole in subsequent cycloaddition or nucleophilic addition reactions. The stereoselectivity of the addition is governed by the existing chiral centers on the sugar backbone, ensuring that the new substituents are installed with the correct spatial orientation crucial for biological activity. For instance, the addition of organomagnesium or organozinc reagents to the nitrone occurs with high diastereoselectivity, yielding hydroxylamine adducts that can be further manipulated. This mechanistic precision eliminates the need for difficult downstream separations of diastereomers, a common cost driver in fine chemical manufacturing.
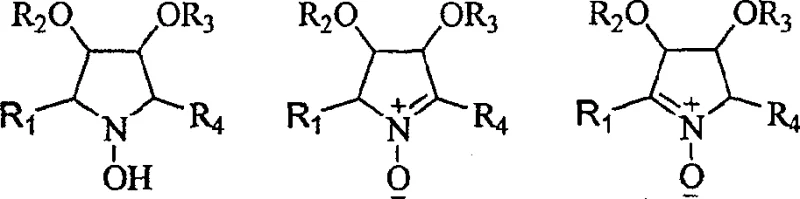
Following the initial addition, the resulting hydroxylamine can be subjected to a second oxidation cycle to regenerate a nitrone, allowing for a second round of nucleophilic attack. This iterative capability is what enables the "branching" described in the patent title. By repeating the oxidation-addition sequence, chemists can build complex quaternary centers and densely functionalized pyrrolidine rings that were previously inaccessible. Furthermore, the protocol includes specific steps for the reduction of the N-O bond using zinc powder or catalytic hydrogenation, converting the hydroxylamine into a stable secondary amine. This amine can then be protected using standard groups like Cbz or Boc, facilitating further functionalization of the side chains without affecting the core ring integrity. Such robustness in the protecting group strategy is vital for multi-step syntheses where orthogonal deprotection is required.
How to Synthesize Branched Polyhydroxypyrrolidine Efficiently
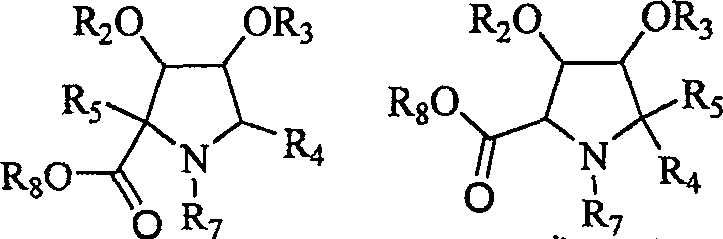
Implementing this synthesis requires careful attention to reaction parameters, particularly during the organometallic addition steps where moisture sensitivity is high. The patent details a systematic progression from hydroxylamine oxidation to nitrone formation, followed by Grignard or organozinc addition at controlled low temperatures to maximize yield and selectivity. Subsequent steps involve the strategic removal of protecting groups, such as trityl or acetonide moieties, under acidic conditions to reveal free hydroxyls or carboxylic acids. For R&D teams looking to replicate or adapt this chemistry, the availability of detailed experimental examples in the patent provides a clear roadmap. The process is designed to be telescoped where possible, minimizing isolation steps and solvent exchanges to improve overall throughput.
- Oxidize the starting hydroxylamine precursor using manganese dioxide or Oxone to generate the reactive nitrone intermediate.
- Perform nucleophilic addition using organometallic reagents such as Grignard or organozinc compounds to introduce diverse alkyl or aryl substituents.
- Execute selective reduction and protection steps, followed by final oxidation to convert hydroxymethyl groups into carboxylic acids for proline analogues.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible benefits in terms of cost stability and supply security. The reliance on commodity chemicals such as manganese dioxide, zinc powder, and common Grignard reagents means that raw material sourcing is not dependent on exotic or single-source suppliers. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Additionally, the high yields reported in the patent examples, often exceeding 70-80% for key steps, imply a significant reduction in waste generation and raw material consumption per kilogram of final product. This efficiency directly correlates to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the use of recyclable solvents contribute to substantial cost savings in utility and waste disposal. By avoiding expensive transition metal catalysts that require rigorous removal to meet ppm-level specifications in APIs, the process simplifies the purification workflow. The ability to perform reactions at or near room temperature for several steps reduces the energy load on heating and cooling systems, further driving down operational expenditures. Moreover, the high stereoselectivity minimizes the loss of valuable chiral starting materials, ensuring that the expensive sugar-derived precursors are converted into product with maximum atom economy.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch quality, a critical factor for maintaining regulatory compliance in API manufacturing. Since the reagents are stable and the reaction endpoints are easily monitored via TLC or HPLC, the risk of batch failure is significantly reduced compared to more finicky enzymatic or biocatalytic processes. This reliability allows supply chain planners to forecast production timelines with greater accuracy, reducing the need for excessive safety stock. The scalability of the method from gram to multi-kilogram scales without significant re-optimization means that capacity can be ramped up quickly to meet sudden spikes in demand from downstream drug developers.
- Scalability and Environmental Compliance: The process aligns well with green chemistry principles by utilizing aqueous workups and avoiding heavy metal contaminants in the final product. The oxidation steps using Oxone or manganese dioxide generate benign byproducts that are easier to treat in standard effluent treatment plants compared to chromium-based oxidants. This environmental compatibility simplifies the permitting process for new manufacturing lines and reduces the long-term liability associated with hazardous waste storage. Furthermore, the modular nature of the synthesis allows for continuous flow processing opportunities, which can further enhance safety and throughput for large-scale commercial production of these complex heterocycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this branched polyhydroxypyrrolidine synthesis technology. These insights are derived directly from the experimental data and scope definitions provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating these intermediates into their own development pipelines. The answers reflect the practical realities of scaling this chemistry while maintaining the high purity standards required for pharmaceutical applications.
Q: What are the advantages of this nitrone-based method over conventional imino sugar synthesis?
A: Unlike conventional methods limited to methyl substitutions or suffering from low yields, this iterative nitrone addition approach allows for the introduction of diverse lipophilic groups (C1-C20 alkyl, aryl, vinyl) with high stereoselectivity and significantly improved yields.
Q: Can this process be scaled for commercial production of glycosidase inhibitors?
A: Yes, the process utilizes mild reaction conditions ranging from -80°C to room temperature and employs common organic solvents like THF and dichloromethane, making it highly suitable for scale-up from laboratory to multi-ton commercial manufacturing.
Q: How does the method control stereochemistry in branched pyrrolidine rings?
A: The method leverages the inherent stereoselectivity of nitrone cycloadditions and nucleophilic attacks on the chiral sugar backbone, ensuring the formation of specific 2,5-disubstituted configurations essential for biological activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Branched Polyhydroxypyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and stereochemical integrity of every batch. Our commitment to quality assurance means that you receive materials that are ready for immediate use in sensitive coupling reactions or biological assays, minimizing your internal validation burden.
We invite you to discuss your specific requirements for branched polyhydroxypyrrolidine derivatives and related imino sugar analogues. Our experts can provide a Customized Cost-Saving Analysis tailored to your target molecule, identifying opportunities to optimize the synthetic route for maximum efficiency. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. We are dedicated to being your strategic partner in overcoming synthetic challenges and accelerating your time to market with cost-effective, high-performance chemical solutions.
