Advanced Manufacturing of Novel Spiro Templates for Next-Generation Drug Discovery
Advanced Manufacturing of Novel Spiro Templates for Next-Generation Drug Discovery
The pharmaceutical industry is constantly seeking novel scaffold structures to overcome drug resistance and improve binding affinity, with spirocyclic compounds emerging as a privileged structure in modern medicinal chemistry. Patent CN112552222B discloses a highly efficient preparation method for 2-(2-(tert-butoxycarbonyl)-2-azaspiro[3.4]octan-5-yl)acetic acid, a critical building block for developing new therapeutic agents. This technology addresses the significant bottleneck in accessing diverse spiro templates, which are often hindered by complex synthetic routes and low overall yields in traditional methodologies. By establishing a robust six-step synthesis starting from readily available raw materials, this innovation provides a reliable pathway for generating high-purity intermediates essential for lead optimization campaigns.
For R&D directors and procurement specialists, the ability to source such complex molecular architectures with consistent quality is paramount for accelerating drug discovery timelines. The disclosed method not only optimizes reaction conditions to maximize yield but also ensures that the process is amenable to industrial scaling, thereby reducing the risk associated with late-stage process development. As global demand for specialized pharmaceutical intermediates grows, having access to a validated, patent-protected synthesis route offers a strategic advantage in securing supply chains for next-generation small molecule drugs targeting various disease states.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spirocyclic templates has been plagued by significant challenges, including the reliance on scarce starting materials and multi-step sequences that suffer from cumulative yield losses. Conventional approaches often require harsh reaction conditions, such as extreme temperatures or the use of toxic heavy metal catalysts, which complicate purification and increase environmental disposal costs. Furthermore, many existing literature methods lack the robustness required for kilogram-scale production, leading to batch-to-batch variability that is unacceptable for Good Manufacturing Practice (GMP) environments. The scarcity of reported synthetic routes for specific azaspiro frameworks means that research teams frequently face long lead times when attempting to procure these materials from external vendors.
The Novel Approach
The methodology outlined in patent CN112552222B represents a paradigm shift by utilizing a convergent strategy that builds the spiro core early in the synthesis, ensuring structural integrity throughout subsequent transformations. This novel approach leverages a strategic alkylation and cyclization sequence to construct the 2-azaspiro[3.4]octane skeleton efficiently, followed by a streamlined functionalization process to install the acetic acid side chain. By optimizing each step for operational simplicity and high conversion rates, the process minimizes the formation of difficult-to-remove impurities, thereby reducing the burden on downstream purification units. This results in a cost-effective and scalable manufacturing protocol that significantly enhances the availability of this valuable chemical space for drug discovery programs.
Mechanistic Insights into Spirocyclization and Functionalization
The synthesis begins with the construction of the spirocyclic ketone intermediate (Compound 5), a process that demands precise control over reaction kinetics to achieve the desired ring topology. The initial alkylation of 3-cyanoazetidine with a dihaloalkane sets the stage for ring closure, followed by a Finkelstein reaction where sodium iodide facilitates the substitution of chlorine with iodine, enhancing the leaving group ability for the subsequent cyclization. The critical ring-closing step employs n-butyllithium at cryogenic temperatures (-60°C to -78°C) to generate a lithiated species that undergoes intramolecular nucleophilic attack, forming the strained spiro junction with high fidelity. This low-temperature protocol is essential for suppressing competing elimination reactions and ensuring the formation of the four-membered azetidine ring fused to the cyclopentane system.
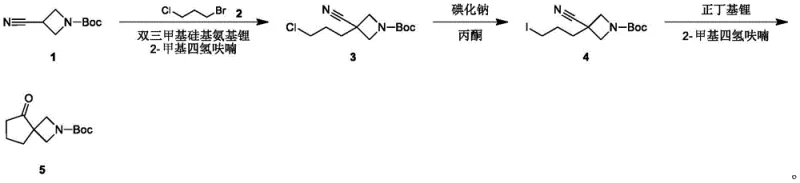
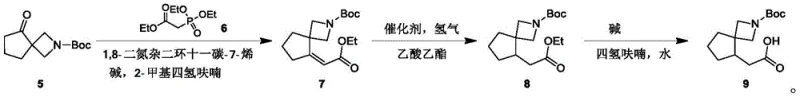
Following the formation of the spiro core, the synthesis proceeds through a Horner-Wadsworth-Emmons (HWE) olefination to introduce the two-carbon acetate side chain. In this key transformation, the spiro-ketone reacts with triethyl phosphonoacetate in the presence of a base such as cesium carbonate and DBU, yielding the alpha,beta-unsaturated ester (Compound 7) with excellent stereocontrol. The resulting alkene is then subjected to catalytic hydrogenation using palladium on carbon under mild pressure (15-25 Psi), which selectively reduces the double bond without affecting the Boc protecting group or the ester functionality. Finally, alkaline hydrolysis cleaves the ethyl ester to reveal the free carboxylic acid, completing the synthesis of the target molecule with high overall purity and yield suitable for biological screening.
How to Synthesize 2-(2-(tert-butoxycarbonyl)-2-azaspiro[3.4]octan-5-yl)acetic acid Efficiently
The patented process offers a clear and reproducible workflow for producing this complex intermediate, breaking down the synthesis into three distinct operational phases that can be easily managed in a pilot or production plant. The first phase focuses on the rigorous construction of the spirocyclic backbone, requiring careful temperature management during the lithiation step to ensure safety and selectivity. The second phase involves the carbon-chain extension via olefination and subsequent saturation, utilizing standard catalytic hydrogenation equipment that is widely available in fine chemical facilities. The final phase is a straightforward hydrolysis that converts the ester precursor into the final acid product, utilizing common aqueous workup procedures to isolate the material in high purity.
- Prepare the spiro-ketone intermediate (Compound 5) via alkylation of 3-cyanoazetidine, followed by Finkelstein reaction and intramolecular cyclization using n-butyllithium.
- Perform Horner-Wadsworth-Emmons olefination between Compound 5 and triethyl phosphonoacetate to generate the unsaturated ester Compound 7.
- Execute catalytic hydrogenation to saturate the double bond, followed by alkaline hydrolysis to yield the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthesis route offers substantial benefits by relying on commodity chemicals and avoiding the need for custom-synthesized, high-cost starting materials. The use of 3-cyanoazetidine derivatives as the primary feedstock ensures a stable supply base, as these materials are produced by multiple manufacturers globally, mitigating the risk of single-source dependency. Additionally, the reaction conditions are designed to be operationally friendly, utilizing solvents like 2-methyltetrahydrofuran and ethyl acetate which are easier to recover and recycle compared to more hazardous alternatives, contributing to lower operational expenditures and a reduced environmental footprint.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by eliminating the need for expensive transition metal catalysts in the carbon-carbon bond-forming steps, relying instead on organophosphorus reagents and basic catalysis. The high yields observed in the hydrogenation and hydrolysis steps minimize material loss, ensuring that the theoretical output is closely matched by actual production volumes. Furthermore, the avoidance of chromatographic purification in favor of crystallization and extraction techniques drastically reduces solvent consumption and processing time, leading to a leaner and more cost-efficient manufacturing profile.
- Enhanced Supply Chain Reliability: By shortening the synthetic route to just six steps from commercially available inputs, the lead time for producing this intermediate is drastically reduced compared to longer, linear syntheses. The robustness of the reaction conditions, particularly the tolerance of the Boc protecting group throughout the sequence, ensures consistent batch quality and reduces the likelihood of production delays caused by failed reactions. This reliability allows supply chain managers to maintain lower safety stock levels while still meeting the demanding delivery schedules of pharmaceutical clients.
- Scalability and Environmental Compliance: The methodology is inherently scalable, with reaction parameters such as temperature and pressure kept within ranges that are easily manageable in large-scale stainless steel reactors. The waste streams generated are primarily aqueous salts and organic solvents that can be treated using standard effluent treatment protocols, aligning with increasingly stringent environmental regulations. The ability to run the hydrogenation step at relatively low pressures further enhances the safety profile of the process, making it suitable for deployment in a wider range of manufacturing facilities without requiring specialized high-pressure infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this spirocyclic intermediate, based on the detailed specifications provided in the patent documentation. Understanding these aspects is crucial for integrating this material into your drug discovery pipeline or manufacturing process. The answers reflect the optimized conditions and safety measures inherent to the described synthetic route.
Q: What are the critical temperature controls for the spirocyclization step?
A: The cyclization step utilizing n-butyllithium requires strict cryogenic conditions, specifically maintaining temperatures between -60°C and -78°C to ensure high regioselectivity and prevent side reactions during the formation of the spiro-core.
Q: How does this method improve supply chain reliability for spiro intermediates?
A: By utilizing commercially available starting materials like 3-cyanoazetidine and avoiding exotic reagents, the process significantly reduces lead times and dependency on custom synthesis, ensuring a more stable and continuous supply for downstream API manufacturing.
Q: Is the final hydrolysis step compatible with large-scale production?
A: Yes, the final hydrolysis operates under mild conditions (15-35°C) using common bases like lithium hydroxide or sodium hydroxide, eliminating the need for hazardous high-pressure or high-temperature reactors, which facilitates safe commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-(tert-butoxycarbonyl)-2-azaspiro[3.4]octan-5-yl)acetic acid Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating complex patent methodologies into commercial reality, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the specific cryogenic and hydrogenation steps required for this spiro compound, ensuring that every batch meets stringent purity specifications required for clinical and commercial applications. With our rigorous QC labs and state-of-the-art manufacturing facilities, we guarantee the supply continuity and quality consistency that global pharmaceutical partners demand for their critical development projects.
We invite you to collaborate with us to secure a stable supply of this high-value intermediate for your upcoming programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your timeline and budget goals effectively.
