Advanced Manufacturing of N-Methyl-N-(2-(4-Hydroxyphenyl)Ethyl)-2-Bromo-5-Hydroxy-4-Methoxybenzamide for Alzheimer’s Therapy
The pharmaceutical industry's relentless pursuit of effective treatments for neurodegenerative disorders has placed significant emphasis on the efficient production of Galantamine, a potent acetylcholinesterase inhibitor approved for the treatment of Alzheimer's disease. Central to the commercial viability of this active pharmaceutical ingredient (API) is the robust and economical synthesis of its key precursors. Patent CN101674835A discloses a groundbreaking methodology for preparing N-methyl-N-(2-(4-hydroxyphenyl)ethyl)-2-bromo-5-hydroxy-4-methoxybenzamide, a critical intermediate often referred to as compound C2. This technical disclosure represents a paradigm shift from traditional multi-step protective group strategies to a streamlined, direct coupling approach. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for optimizing supply chains and reducing the cost of goods sold (COGS) in the competitive neurological drug market. The innovation lies not merely in a new chemical transformation but in a holistic re-engineering of the synthetic route that addresses long-standing purity and yield challenges inherent in legacy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the target benzamide intermediate C2 has relied on a convoluted nine-step sequence that heavily utilizes benzyl ether protecting groups to mask phenolic hydroxyl functionalities during the construction of the molecular framework. As illustrated in the prior art scheme, this conventional pathway necessitates the initial protection of tyramine derivatives followed by separate functionalization of the benzoic acid moiety, culminating in a final deprotection step using harsh hydrobromic acid conditions. 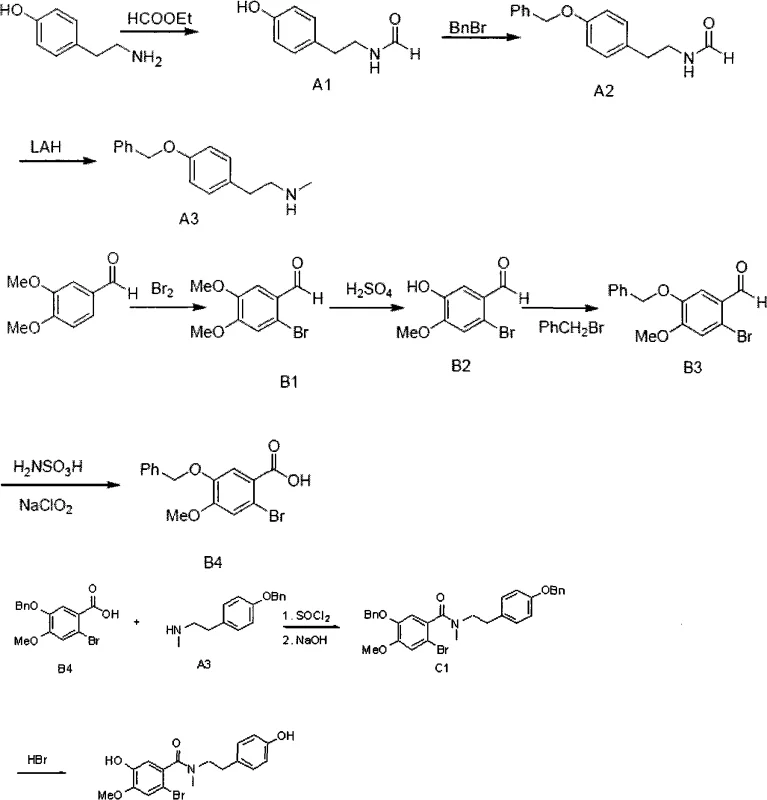 The reliance on benzyl groups introduces a critical vulnerability in the manufacturing process: the potential for benzyl migration. During the final acidic deprotection phase, the benzyl group can migrate from the oxygen atom to the aromatic ring, generating a structurally similar impurity known as C3. This specific byproduct possesses physicochemical properties nearly identical to the desired product, making its removal via standard crystallization or chromatography exceptionally difficult and costly. Furthermore, the nine-step linear sequence inherently accumulates yield losses at each stage, resulting in a low overall throughput that negatively impacts the economic feasibility of large-scale production. The use of stoichiometric amounts of strong reducing agents like lithium aluminum hydride and hazardous alkylating agents further complicates the safety profile and waste management requirements of the conventional process.
The reliance on benzyl groups introduces a critical vulnerability in the manufacturing process: the potential for benzyl migration. During the final acidic deprotection phase, the benzyl group can migrate from the oxygen atom to the aromatic ring, generating a structurally similar impurity known as C3. This specific byproduct possesses physicochemical properties nearly identical to the desired product, making its removal via standard crystallization or chromatography exceptionally difficult and costly. Furthermore, the nine-step linear sequence inherently accumulates yield losses at each stage, resulting in a low overall throughput that negatively impacts the economic feasibility of large-scale production. The use of stoichiometric amounts of strong reducing agents like lithium aluminum hydride and hazardous alkylating agents further complicates the safety profile and waste management requirements of the conventional process.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel process described in the patent achieves the synthesis of compound C2 through a direct condensation of two fully deprotected building blocks: 2-bromo-5-hydroxy-4-methoxybenzoic acid (B13) and N-methyl-N-(2-(4-hydroxyphenyl)ethyl)amine (A12). This strategic simplification eliminates the need for any protecting group chemistry, thereby removing the entire risk profile associated with benzyl migration and the subsequent purification nightmares. By bypassing the protection-deprotection cycle, the synthetic route is drastically shortened, reducing the number of unit operations and the total volume of solvents required. The direct coupling strategy leverages the differential reactivity of the functional groups, allowing for selective amide bond formation in the presence of free phenolic hydroxyls under controlled basic conditions. This approach not only enhances the chemical purity of the final intermediate by avoiding the formation of migration byproducts but also significantly improves the mass balance of the overall process. For supply chain managers, this translates to a more resilient manufacturing protocol that is less susceptible to bottlenecks caused by complex purification steps or the handling of hazardous reagents required for protection and deprotection.
Mechanistic Insights into Direct Amide Coupling
The core chemical innovation of this process revolves around the activation of the carboxylic acid functionality in compound B13 to facilitate nucleophilic attack by the secondary amine A12. The preferred embodiment utilizes thionyl chloride (SOCl2) as the activating agent to convert the carboxylic acid into the corresponding acid chloride in situ. This activation step is typically conducted in a polar aprotic or moderately polar organic solvent such as ethyl acetate or methyl isobutyl ketone at elevated temperatures ranging from 45°C to 50°C. The formation of the acid chloride is a critical equilibrium that must be driven to completion, often aided by catalytic amounts of dimethylformamide (DMF) which generates the highly reactive Vilsmeier-Haack intermediate. Once the acid chloride is formed, excess thionyl chloride is removed under vacuum to prevent side reactions and ensure a clean coupling environment. The resulting activated species is then dissolved in a solvent compatible with the subsequent coupling step, such as dichloromethane, ready for reaction with the amine component.
The coupling reaction itself is a delicate balance of kinetics and thermodynamics, requiring precise control over pH and temperature to maximize yield and minimize hydrolysis of the activated acid. The amine component, typically supplied as a stable hydrobromide salt (A12·HBr), is neutralized in situ using a base such as sodium hydroxide or sodium carbonate. This neutralization generates the free amine nucleophile which immediately attacks the electrophilic carbonyl carbon of the acid chloride. The reaction is preferably conducted at low temperatures, specifically between 0°C and 5°C, to suppress exothermic runaway and prevent the hydrolysis of the acid chloride by water present in the biphasic system or generated during neutralization. 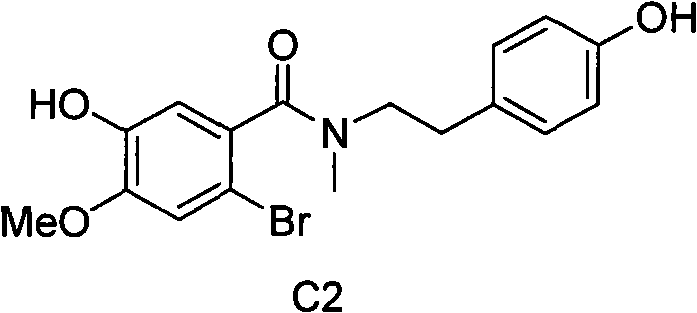 The use of a biphasic solvent system, such as dichloromethane and methanol with aqueous base, facilitates the transfer of the ionic amine salt into the organic phase where the reaction occurs. Following the coupling, the reaction mixture is subjected to pH adjustment and crystallization protocols that leverage the solubility differences between the product and any unreacted starting materials, ensuring high purity without the need for column chromatography. This mechanistic elegance allows for the production of high-purity pharmaceutical intermediates suitable for direct conversion into Galantamine.
The use of a biphasic solvent system, such as dichloromethane and methanol with aqueous base, facilitates the transfer of the ionic amine salt into the organic phase where the reaction occurs. Following the coupling, the reaction mixture is subjected to pH adjustment and crystallization protocols that leverage the solubility differences between the product and any unreacted starting materials, ensuring high purity without the need for column chromatography. This mechanistic elegance allows for the production of high-purity pharmaceutical intermediates suitable for direct conversion into Galantamine.
How to Synthesize N-Methyl-N-(2-(4-Hydroxyphenyl)Ethyl)-2-Bromo-5-Hydroxy-4-Methoxybenzamide Efficiently
Implementing this novel synthetic route requires strict adherence to the activation and coupling parameters defined in the patent to ensure reproducibility and safety. The process begins with the preparation of the acid chloride from 2-bromo-5-hydroxy-4-methoxybenzoic acid, followed by the separate preparation of the free amine from its salt form. The convergence of these two streams must be managed carefully to maintain the reaction temperature within the optimal window. Detailed standardized operating procedures regarding reagent addition rates, stirring speeds, and quenching protocols are essential for successful technology transfer from the laboratory to the pilot plant. The following guide outlines the critical operational phases for executing this synthesis effectively.
- Activate 2-bromo-5-hydroxy-4-methoxybenzoic acid (B13) using thionyl chloride in an organic solvent like ethyl acetate at 45-50°C to form the acid chloride.
- Prepare the amine component by neutralizing N-methyl-N-(2-(4-hydroxyphenyl)ethyl)amine HBr salt (A12HBr) with a base such as sodium hydroxide in methanol.
- Couple the activated acid chloride with the free amine in a biphasic or organic solvent system at controlled low temperatures (0-5°C) followed by pH adjustment and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this protective group-free synthesis offers tangible economic and logistical benefits that extend beyond simple yield improvements. The elimination of benzyl bromide, a potent lachrymator and alkylating agent, from the bill of materials significantly reduces the regulatory burden and safety costs associated with handling hazardous chemicals. Furthermore, removing the lithium aluminum hydride reduction step eliminates the need for specialized equipment capable of handling pyrophoric materials and the complex workup procedures required to safely quench such reactions. These changes collectively lower the barrier to entry for contract manufacturing organizations (CMOs) and reduce the overall cost of manufacturing (COM) by simplifying the facility requirements and waste treatment protocols. The streamlined nature of the process also意味着 a shorter production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The most significant driver of cost savings in this new process is the drastic reduction in raw material consumption and processing time. By cutting the synthetic sequence from nine steps to essentially two major operational stages, manufacturers save substantially on solvent volumes, energy consumption for heating and cooling, and labor hours. The avoidance of expensive protecting group reagents and the associated deprotection chemicals further reduces the direct material costs. Additionally, the higher overall yield resulting from fewer isolation steps means that less starting material is required to produce the same amount of final product, directly improving the margin profile for the API manufacturer. This efficiency makes the production of Galantamine intermediates more economically viable in regions with stricter environmental regulations and higher operational costs.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly enhanced by the use of commodity chemicals and readily available starting materials. The key building blocks, such as vanillic acid derivatives and tyramine analogues, are produced by multiple global suppliers, reducing the risk of single-source dependency. The robustness of the direct coupling chemistry also means that the process is less sensitive to minor variations in reagent quality, leading to more consistent batch-to-batch performance. This reliability is crucial for maintaining continuous API production schedules and avoiding costly delays caused by failed batches or extended purification times. The simplified workflow also facilitates easier technology transfer between different manufacturing sites, providing greater flexibility in sourcing and production planning.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns well with green chemistry principles by minimizing waste generation and avoiding the use of heavy metals or highly toxic reagents. The reduction in step count inherently reduces the E-factor (mass of waste per mass of product), making the process more sustainable and easier to permit in environmentally sensitive jurisdictions. The scalability of the reaction is proven by the use of standard unit operations such as stirred tank reactors and filtration units, which are ubiquitous in the fine chemical industry. This ensures that the process can be scaled from kilogram to multi-ton quantities without the need for specialized reactor configurations or exotic catalysts, securing a stable long-term supply of this critical pharmaceutical intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology. Understanding these details is vital for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: How does this new process eliminate the difficult-to-remove impurity C3?
A: The conventional method utilizes benzyl protecting groups which, upon removal with hydrobromic acid, can migrate to the phenol ring forming impurity C3. This novel process avoids benzyl protection entirely by using unprotected starting materials, thereby eliminating the formation pathway for this specific migration impurity.
Q: What are the scalability advantages of the direct coupling method?
A: The process reduces the synthetic sequence from nine steps to essentially two main operational stages (activation and coupling). This reduction minimizes unit operations, solvent usage, and processing time, making it highly suitable for commercial scale-up from pilot plants to multi-ton production without the yield losses associated with multiple isolation steps.
Q: Is the starting material 2-bromo-5-hydroxy-4-methoxybenzoic acid commercially viable?
A: Yes, the starting acid (B13) can be prepared efficiently from readily available aldehydes via oxidation with sodium chlorite, a cost-effective and scalable oxidant. This ensures a reliable supply chain for the key building block without relying on exotic or expensive reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl-N-(2-(4-Hydroxyphenyl)Ethyl)-2-Bromo-5-Hydroxy-4-Methoxybenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team specializes in the process development and scale-up of complex pharmaceutical intermediates, including the Galantamine precursor detailed in patent CN101674835A. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of intermediate meets the highest international standards for API synthesis. We understand the critical nature of neurological drug supply chains and are committed to delivering products that facilitate the uninterrupted manufacture of life-saving medications.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. Whether you are looking for a Customized Cost-Saving Analysis or need to evaluate the technical feasibility of this route for your existing infrastructure, our experts are ready to assist. Please contact us to request specific COA data, route feasibility assessments, and comprehensive quotations tailored to your volume needs. Let us collaborate to drive efficiency and quality in the production of next-generation Alzheimer's therapeutics.
