Advanced Synthesis of Quinolin-2-ylmethoxyphenylacetic Acid Derivatives for Pharmaceutical Applications
Advanced Synthesis of Quinolin-2-ylmethoxyphenylacetic Acid Derivatives for Pharmaceutical Applications
The pharmaceutical landscape continuously demands novel scaffolds capable of modulating specific enzymatic pathways with high potency and selectivity. Patent CN1087337A introduces a significant advancement in the field of anti-inflammatory agents through the development of substituted quinolin-2-ylmethoxyphenylacetic acid derivatives. These compounds are identified as potent inhibitors of arachidonic acid metabolism, specifically targeting the 5-lipoxygenase pathway, which is critical in the biosynthesis of leukotrienes. For R&D directors and procurement specialists in the fine chemical sector, understanding the synthetic accessibility and structural versatility of these molecules is paramount. The patent outlines a robust methodology that allows for extensive diversification at multiple positions on the phenylacetic acid core, enabling the rapid generation of structure-activity relationship (SAR) data. This technical insight report delves into the mechanistic nuances and commercial implications of this synthesis route, positioning it as a viable candidate for scale-up in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing complex phenylacetic acid derivatives often suffer from regioselectivity issues and harsh reaction conditions that compromise overall yield and purity. Conventional etherification strategies frequently require aggressive alkylating agents or elevated temperatures that can lead to decomposition of sensitive functional groups, particularly when dealing with heterocyclic systems like quinoline. Furthermore, introducing substituents at the alpha-position of the acetic acid chain traditionally involves multi-step sequences with poor atom economy, often requiring protecting group strategies that add unnecessary complexity and cost to the manufacturing process. The inability to easily introduce diverse alkenyl or alkynyl moieties without resorting to low-yielding substitution reactions has historically limited the exploration of chemical space around this pharmacophore. These limitations result in extended lead times for process development and increased costs associated with waste disposal and purification, posing significant challenges for supply chain heads aiming for efficient commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology described in CN1087337A overcomes these hurdles through a modular synthetic strategy centered on the etherification of substituted phenols with 2-halogenated methylquinolines. This approach utilizes mild conditions, typically employing potassium carbonate in dimethylformamide (DMF) at approximately 100°C, which ensures high conversion rates while preserving the integrity of the quinoline ring system. A key innovation lies in the subsequent alpha-alkylation step, where strong bases like potassium tert-butoxide facilitate the direct introduction of cycloalkyl or alkyl groups with impressive yields, as demonstrated by the 80.6% yield in Example 2. Moreover, the integration of palladium(0)-catalyzed cross-coupling reactions allows for the seamless incorporation of alkenyl and alkynyl side chains, vastly expanding the library of accessible derivatives without the need for pre-functionalized starting materials. This streamlined workflow significantly reduces the number of isolation steps and enhances the overall process mass intensity, offering a clear pathway for cost reduction in pharmaceutical intermediates manufacturing.
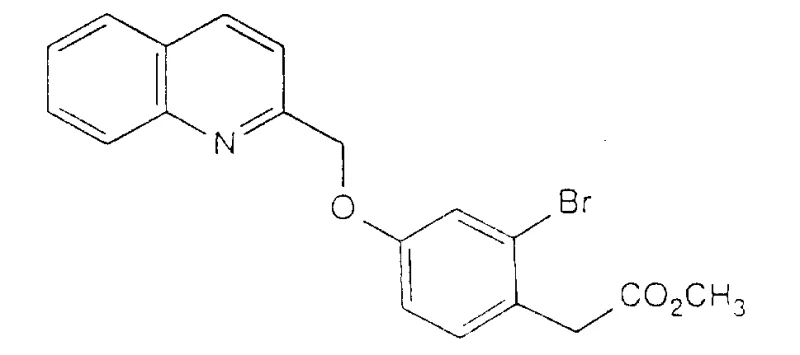
Mechanistic Insights into Etherification and Palladium-Catalyzed Coupling
The core of this synthetic route relies on a nucleophilic aromatic substitution mechanism during the initial etherification stage. The phenolic oxygen, activated by a base such as potassium carbonate, attacks the electrophilic methylene carbon of the 2-chloromethylquinoline hydrochloride. This SN2-type displacement is highly efficient in polar aprotic solvents like DMF, which stabilize the transition state and solvate the resulting halide salt. The choice of base and solvent is critical; the patent data indicates that using inorganic carbonates avoids the side reactions often seen with stronger alkoxides, ensuring a clean formation of the quinolin-2-ylmethoxy linkage. Following this, the alpha-proton of the acetate ester is deprotonated by a strong base like potassium tert-butoxide to generate a stabilized enolate. This nucleophilic species then reacts with alkyl halides, such as bromocyclopentane or bromocycloheptane, to install the desired alpha-substituent. The steric bulk of the base and the solvent environment are tuned to favor mono-alkylation, preventing over-alkylation and ensuring high regioselectivity for the target mono-substituted product.
For more complex substitutions, the process employs a palladium-catalyzed cross-coupling mechanism, specifically utilizing tetrakis(triphenylphosphine)palladium(0). In this catalytic cycle, the palladium center undergoes oxidative addition with the aryl bromide intermediate, forming an aryl-palladium complex. Subsequent transmetallation with organotin reagents, such as allyltributyltin or vinyltributyltin, transfers the organic group to the palladium center. Reductive elimination then releases the coupled product and regenerates the active Pd(0) catalyst. This mechanism is particularly valuable for introducing unsaturated side chains that are difficult to access via direct alkylation. The patent highlights that this reaction proceeds effectively in toluene under argon atmosphere, minimizing oxidation of the catalyst. The ability to perform this transformation on the ester intermediate allows for late-stage diversification, which is a crucial advantage for process chemists aiming to optimize the impurity profile and biological activity of the final active pharmaceutical ingredient.
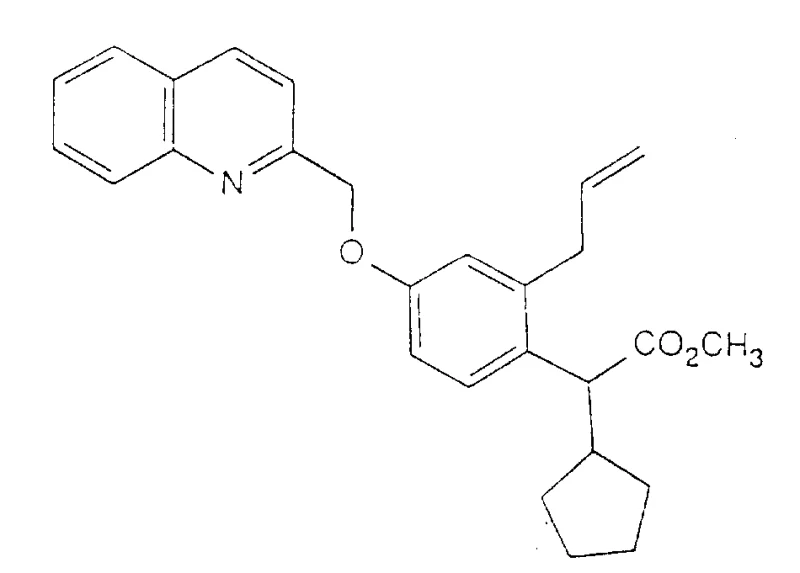
How to Synthesize Quinolin-2-ylmethoxyphenylacetic Acid Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to ensure reproducibility and safety on a commercial scale. The process begins with the preparation of the key ether scaffold, followed by sequential functionalization of the side chain. Operators must maintain strict temperature control during the exothermic alkylation steps and ensure an inert atmosphere for the palladium-catalyzed couplings to prevent catalyst deactivation. The detailed standardized synthesis steps provided below outline the specific stoichiometry, solvent choices, and workup procedures validated in the patent examples to achieve optimal yields and purity specifications.
- Perform etherification of substituted phenols with 2-halogenated methylquinoline in an inert solvent like DMF using a base such as potassium carbonate at elevated temperatures.
- Conduct alpha-alkylation of the resulting ester using alkyl halides and a strong base like potassium tert-butoxide to introduce cycloalkyl or alkyl groups at the alpha position.
- Optionally utilize palladium(0)-catalyzed cross-coupling reactions with organotin compounds to introduce alkenyl or alkynyl substituents, followed by hydrolysis or amidation to finalize the active acid or sulfonamide derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route detailed in CN1087337A offers substantial strategic advantages over legacy methods. The reliance on commercially available starting materials, such as substituted phenols and quinoline derivatives, mitigates the risk of raw material shortages and price volatility. The high yields reported in the patent examples, such as the 80.6% yield for the alpha-alkylation step and 87.7% for the palladium coupling, indicate a robust process that minimizes material loss and maximizes throughput. This efficiency translates directly into lower production costs per kilogram, making the final API intermediate more competitive in the global market. Furthermore, the modularity of the synthesis allows for flexible manufacturing schedules; different derivatives can be produced on the same equipment train by simply swapping the alkylating agent or organotin reagent, thereby enhancing asset utilization and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process eliminates the need for complex protecting group strategies and reduces the total number of synthetic steps required to reach the final scaffold. By utilizing direct alkylation and efficient etherification, the consumption of reagents and solvents is significantly optimized. The high conversion rates observed in the examples suggest that downstream purification costs, such as chromatography or recrystallization, can be minimized, leading to substantial cost savings in the overall manufacturing budget. Additionally, the use of standard heterogeneous catalysts that can potentially be recovered further contributes to economic efficiency.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are commodity chemicals or readily accessible fine chemicals, ensuring a stable and continuous supply chain. Unlike routes dependent on exotic or custom-synthesized building blocks, this method leverages established supply networks for phenols, quinolines, and common alkyl halides. This reduces the risk of supply disruptions and allows for better forecasting and inventory management. The scalability of the reactions, demonstrated from gram to multi-gram scales in the patent, provides confidence that the process can be seamlessly transferred to pilot and commercial plants without significant re-engineering.
- Scalability and Environmental Compliance: The reaction conditions are generally mild and operate at atmospheric pressure, reducing the engineering controls required for high-pressure reactors. The solvents used, such as DMF, toluene, and methanol, are well-understood in industrial settings with established recovery and recycling protocols, facilitating compliance with environmental regulations. The high atom economy of the coupling reactions and the ability to recycle catalysts contribute to a greener manufacturing footprint. This alignment with sustainability goals is increasingly important for multinational corporations seeking to reduce their environmental impact while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these quinoline derivatives. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on reaction scope and potential limitations.
Q: What is the primary therapeutic application of these quinoline derivatives?
A: According to patent CN1087337A, these compounds act as inhibitors of arachidonic acid metabolism, specifically 5-lipoxygenase, making them potent candidates for treating inflammatory diseases like asthma, rheumatism, and psoriasis.
Q: How is the alpha-position functionalized in this synthesis route?
A: The alpha-position is functionalized through alkylation using strong bases like potassium tert-butoxide in DMF, or via palladium-catalyzed cross-coupling reactions using organotin reagents for more complex side chains.
Q: What are the typical reaction conditions for the key etherification step?
A: The etherification is typically conducted in polar aprotic solvents such as DMF at temperatures around 100°C, using inorganic bases like potassium carbonate to facilitate the nucleophilic substitution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolin-2-ylmethoxyphenylacetic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation anti-inflammatory therapies. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1087337A can be executed with precision and consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific analogs or large-scale supply of the core scaffold, our facility is designed to accommodate your evolving project needs with flexibility and speed.
We invite you to collaborate with us to accelerate your drug development timeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain objectives and drive your project forward efficiently.
