Scalable Synthesis of Heterocyclic Phenylbutenamide Derivatives for Immunological Therapies
Introduction to Novel Immunomodulatory Intermediates
The pharmaceutical landscape for treating immunological disorders such as arthritis and leukotriene-mediated diseases has evolved significantly with the discovery of advanced phenylbutenamide derivatives. Patent CN87105515A discloses a groundbreaking class of compounds characterized by a unique structural motif where a heterocyclic ring links the amide functionality to a substituted phenyl group. This architectural shift from simple phenyl substitutions to complex heterocyclic systems represents a critical advancement in drug design, offering improved binding affinity and metabolic stability. The core structure, defined by Formula (I), allows for extensive diversification through various substituents on both the phenyl ring and the heterocyclic linker, enabling fine-tuning of pharmacokinetic properties.
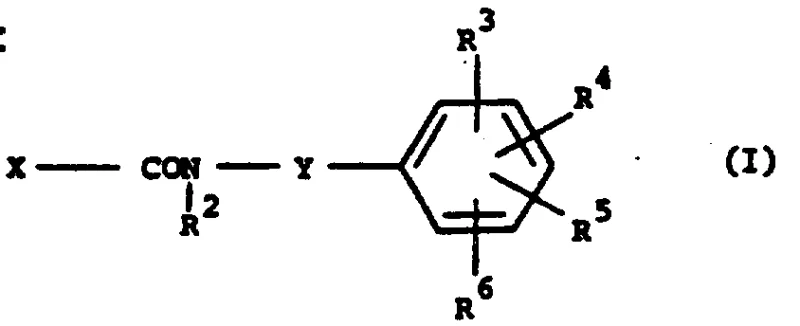
These compounds function as potent inhibitors of T-cell proliferation and graft-versus-host responses, mechanisms central to autoimmune pathologies. Furthermore, their ability to inhibit 5-lipoxygenase products positions them as valuable therapeutic agents for asthma and hypersensitivity reactions. For research and development teams seeking reliable pharmaceutical intermediate suppliers, understanding the synthetic accessibility and structural versatility of these molecules is paramount. The patent outlines multiple preparation methods, providing a robust foundation for developing scalable manufacturing processes that meet the stringent purity requirements of modern API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, such as British Patent 1571990, described phenylbutenamide compounds where the benzene ring was optionally substituted with simple alkyl groups like methyl or ethyl. While these earlier compounds demonstrated some pharmaceutical activity, their structural simplicity often limited their potency and selectivity. Direct substitution on the phenyl ring without an intervening heterocyclic spacer can lead to rapid metabolic clearance or off-target interactions, reducing therapeutic efficacy. Additionally, traditional synthesis routes for similar amides often relied on harsh conditions or produced complex mixtures of byproducts that were difficult to separate, complicating the purification process and increasing overall production costs.
The Novel Approach
The present invention overcomes these limitations by introducing a 5- or 6-membered heterocyclic ring (Y), such as thiophene, furan, or thiazole, between the amide nitrogen and the phenyl ring. This structural innovation not only enhances biological activity but also offers superior synthetic flexibility. The patent details several distinct pathways to access these scaffolds, with the reaction of heterocyclic isocyanates with cyanoacetone salts emerging as a particularly efficient strategy. This approach allows for the modular assembly of the molecule, where the heterocyclic component and the phenyl component can be optimized independently before coupling. Such modularity is essential for cost reduction in API manufacturing, as it enables the parallel optimization of sub-units and simplifies the final coupling step.
Mechanistic Insights into Isocyanate-Mediated Amide Formation
The preferred synthetic pathway (Process A) involves the nucleophilic attack of a cyanoacetone enolate on an electrophilic isocyanate carbon. This reaction is typically conducted in an inert organic solvent like tetrahydrofuran at temperatures ranging from -30°C to 100°C. The mechanism proceeds through the formation of a tetrahedral intermediate which subsequently rearranges to form the stable amide bond. One of the key advantages of this mechanism is the high atom economy and the avoidance of toxic coupling reagents often used in peptide synthesis. The resulting product exists in a tautomeric equilibrium between the enol and keto forms, as well as Z and E geometric isomers, with the Z-enol form generally predominating.
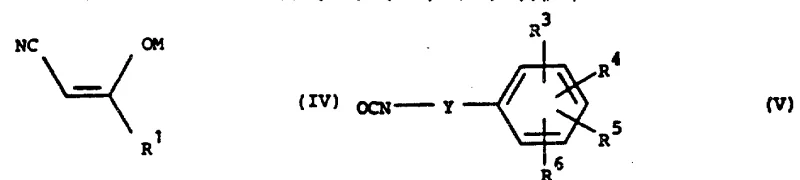
Impurity control in this process is managed by carefully regulating the reaction temperature and the stoichiometry of the base used to generate the enolate. Excess base can lead to over-reaction or degradation of the sensitive isocyanate functionality, while insufficient activation results in low conversion rates. The patent specifies that the crude salt formed can be converted to the free hydroxy compound by treatment with aqueous mineral acid, a step that also helps in precipitating the product from the reaction mixture. This straightforward workup procedure minimizes the need for chromatographic purification, which is a significant benefit for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Phenylbutenamide Derivatives Efficiently
The synthesis of these high-value immunomodulatory intermediates requires precise control over reaction parameters to ensure high yield and purity. The process begins with the preparation of the heterocyclic isocyanate, often derived from the corresponding carboxylic acid via a Curtius rearrangement involving an acyl azide intermediate. This step must be handled with care due to the potential hazards associated with azides, but it provides a highly reactive species ideal for the subsequent coupling. The detailed standardized synthesis steps for generating the final butenamide structure are outlined in the guide below, ensuring reproducibility across different manufacturing scales.
- Preparation of the heterocyclic isocyanate intermediate by converting the corresponding carboxylic acid to an acyl azide, followed by thermal rearrangement.
- Generation of the nucleophilic enolate species by reacting cyanoacetone or its derivative with a strong base such as sodium hydride or sodium ethoxide in an inert solvent.
- Coupling the enolate with the isocyanate at controlled temperatures (-30°C to 100°C) to form the target butenamide backbone, followed by acidification to isolate the free hydroxy compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers tangible benefits in terms of cost efficiency and supply reliability. The reliance on widely available starting materials, such as substituted benzenes and common heterocycles, mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Furthermore, the robustness of the isocyanate coupling reaction means that it can be performed in standard stainless steel reactors without the need for specialized equipment, facilitating easier technology transfer between sites.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, which are often required for alternative cross-coupling strategies to build the heterocyclic-phenyl bond, leads to significant cost savings. By utilizing classical organic transformations like the Curtius rearrangement and enolate coupling, manufacturers can avoid the costly removal of trace metal residues, a critical step in meeting regulatory standards for pharmaceutical ingredients. This streamlined process reduces both material costs and waste disposal expenses.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the stocking of key intermediates, such as the heterocyclic acids and cyanoacetone derivatives, which have long shelf lives. This inventory flexibility ensures that production can be ramped up quickly in response to market demand without long lead times for custom synthesis. The use of common solvents like THF and ethanol further simplifies logistics, as these are readily available from multiple global suppliers.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to older methods that might utilize heavy metals or chlorinated solvents in excess. The final acidification step produces inorganic salts as byproducts, which are easily treated in standard wastewater facilities. This environmental profile aligns with modern green chemistry initiatives, reducing the regulatory burden and enhancing the sustainability credentials of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the production and application of these phenylbutenamide derivatives. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and scope of the technology.
Q: What distinguishes these phenylbutenamide derivatives from prior art compounds?
A: Unlike earlier phenylbutenamides substituted directly on the benzene ring, these novel compounds incorporate a 5- or 6-membered heterocyclic linker (such as thiophene or furan) between the amide group and the phenyl ring. This structural modification significantly enhances their efficacy in inhibiting T-cell proliferation and leukotriene formation, making them superior candidates for treating arthritis and allergic conditions.
Q: Which synthetic route is most suitable for large-scale manufacturing?
A: The reaction between a metal salt of a cyanoacetone derivative and a heterocyclic isocyanate (Process A) is identified as the optimal route for industrial scale-up. This method avoids the use of expensive transition metal catalysts required for other pathways and utilizes readily available starting materials, ensuring high throughput and simplified purification protocols.
Q: How is the stereochemistry of the butenamide double bond controlled?
A: The synthesis typically yields a mixture of Z and E isomers, with the Z-form predominating under standard conditions. While the keto form serves as a synthetic intermediate, the final products can be separated into pure geometric isomers using conventional crystallization techniques, allowing for the isolation of the specific stereoisomer with the desired pharmacological profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylbutenamide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation immunotherapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of phenylbutenamide derivative meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain for these vital compounds. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your drug development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
