Advanced Synthesis of L-3-Hydroxy-4-Methoxy-5-Methylphenylalanine Intermediates for Oncology Drugs
Advanced Synthesis of L-3-Hydroxy-4-Methoxy-5-Methylphenylalanine Intermediates for Oncology Drugs
The development of potent antitumor agents often hinges on the availability of high-purity chiral intermediates, a challenge vividly illustrated in the synthesis of tetrahydroisoquinoline alkaloids. Patent CN101585745B introduces a groundbreaking methodology for the preparation of amino-protected L-3-hydroxy-4-methoxy-5-methyl-phenylalaninol and its corresponding acid derivatives. These compounds serve as the critical chiral building blocks for synthesizing highly active anticancer agents such as Ecteinascidin 743 (Trabectedin), Saframycin A, and Cribrostatin IV. The strategic importance of these molecules cannot be overstated, as Ecteinascidin 743 has demonstrated antitumor activity exceeding that of widely used clinical drugs like taxol and cis-platin by one to three orders of magnitude. 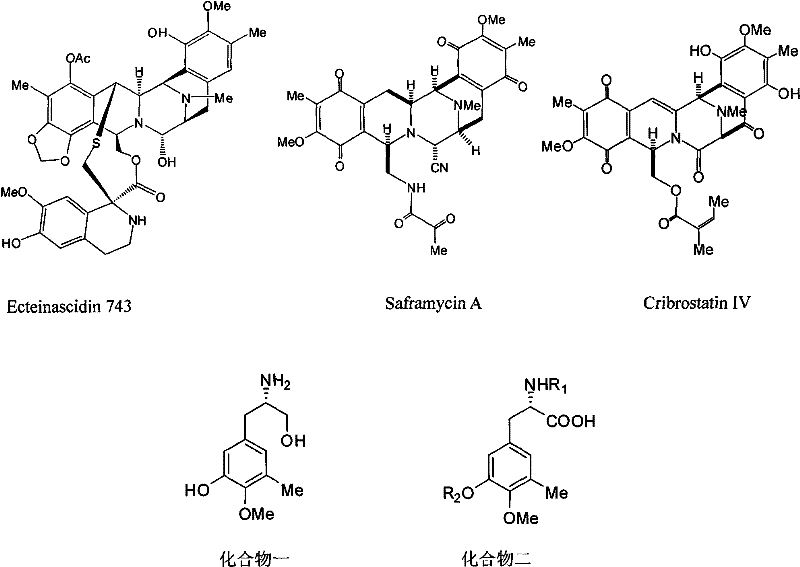
This patented technology addresses the severe bottlenecks found in traditional synthetic routes, which have historically plagued the supply chain for these vital oncology intermediates. By leveraging a concise five-step sequence starting from readily available L-tyrosine derivatives, the invention achieves a total recovery rate of approximately 70%, a significant improvement over prior art. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this route offers a compelling value proposition through its use of inexpensive reagents, mild reaction conditions, and operational simplicity. The shift from complex asymmetric alkylations to a streamlined formylation and oxidation strategy represents a paradigm shift in the cost reduction in API manufacturing for this class of compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted phenylalanine derivatives has been fraught with inefficiencies that hinder commercial viability. Early approaches, such as those reported by Williams et al., relied on the enantioselective alkylation of chiral piperazinone derivatives with benzyl bromides. As depicted in the reaction schemes of the prior art, these methods involve multi-step linear sequences that are not only tedious but also require expensive reagents and stringent anhydrous, oxygen-free conditions. 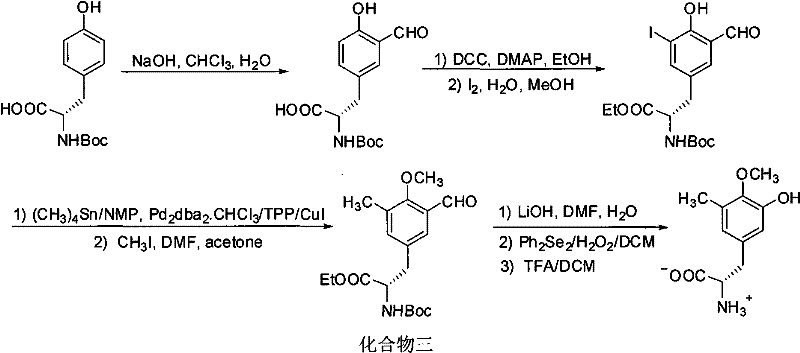
Furthermore, subsequent improvements by Zhu et al. utilizing N-diphenylmethyl Padil tert-butyl ester enantioselective alkylation introduced new complexities. While effective on a small scale, these reactions often suffer from poor stirring at low temperatures due to the high solid content in the reaction system, leading to extended feeding times and a decline in product enantiomeric excess (ee value). Perhaps most critically, the route described by Schmidt et al., which starts from natural chiral source L-N-Boc-tyrosine, relies on the Reimer-Tiemann reaction to introduce the formyl group. This specific step is a major yield bottleneck, typically affording only 25% to 30% conversion. Additionally, the subsequent Dakin oxidation to convert the aldehyde to the phenol proceeds with a mediocre yield of about 50%, drastically inflating the cost of goods and limiting the scalability required for high-purity oncology intermediates.
The Novel Approach
The novel approach detailed in patent CN101585745B circumvents these historical pitfalls by reimagining the synthetic logic entirely. Instead of relying on difficult asymmetric alkylations or low-yielding formylations, the new method initiates with a cheap compound four (an L-tyrosine derivative) and proceeds through a highly efficient one-pot esterification and methylation to generate compound five. The core innovation lies in the selective formylation of the aromatic ring using dichloromethyl ether in the presence of a Lewis acid, which avoids the harsh conditions and poor selectivity of the Reimer-Tiemann reaction. This is followed by a Baeyer-Villiger oxidation to install the hydroxyl group, a transformation that is significantly more robust and higher yielding than the traditional Dakin oxidation. This strategic pivot results in a succinct five-step route that is not only economically superior but also technically more robust for the commercial scale-up of complex amino acid derivatives.
Mechanistic Insights into Baeyer-Villiger Oxidation and Selective Formylation
The mechanistic elegance of this new route is anchored in two key transformations: the Lewis acid-catalyzed selective formylation and the Baeyer-Villiger oxidation. In the formylation step, the use of titanium tetrachloride or tin tetrachloride as a Lewis acid activates dichloromethyl ether, generating an electrophilic species that attacks the electron-rich aromatic ring of the phenylalanine derivative. This reaction is conducted in methylene chloride at temperatures ranging from -78°C to 40°C, allowing for precise control over regioselectivity. Unlike the Reimer-Tiemann reaction, which generates a mixture of ortho- and para-isomers and suffers from polymerization side reactions, this method delivers the desired 3-formyl-4-methoxy-5-methyl substitution pattern with high fidelity. The preservation of the chiral center during this electrophilic aromatic substitution is critical, ensuring that the optical purity of the starting L-tyrosine is maintained throughout the synthesis.
Following formylation, the conversion of the aldehyde to the phenol via Baeyer-Villiger oxidation represents a masterstroke in process chemistry. Traditionally, converting an aromatic aldehyde to a phenol involves the Dakin reaction, which requires strong basic conditions and hydrogen peroxide, often leading to racemization or degradation of sensitive functional groups. In contrast, the patented method employs metachloroperbenzoic acid (mCPBA) in methylene chloride at mild temperatures (-20°C to 60°C). This mechanism involves the nucleophilic attack of the peracid on the carbonyl carbon, followed by a rearrangement that inserts an oxygen atom into the C-H bond. This oxidative insertion is highly chemoselective and tolerates the ester and carbamate protecting groups present on the molecule. Subsequent reduction of the resulting formate ester with LiBH4 or hydrolysis with LiOH cleanly reveals the target phenol functionality. This mechanistic pathway effectively eliminates the formation of tarry byproducts common in Dakin oxidations, thereby simplifying downstream purification and enhancing the overall impurity profile of the final API intermediate.
How to Synthesize L-3-Hydroxy-4-Methoxy-5-Methylphenylalanine Efficiently
The synthesis of these critical intermediates is now accessible through a standardized protocol that balances yield, purity, and operational safety. The process begins with the protection and modification of the tyrosine scaffold, followed by the crucial formylation and oxidation steps described above. The detailed experimental procedures provided in the patent demonstrate that high yields (e.g., 92% for the esterification/reduction sequence and 89% for the formylation) are reproducible under standard laboratory conditions. For process chemists looking to implement this technology, the key lies in maintaining strict temperature control during the Lewis acid catalysis and ensuring the stoichiometry of the oxidant is optimized to prevent over-oxidation. The detailed standardized synthesis steps are outlined below to guide your process development team.
- Perform one-pot carboxyl esterification and phenolic hydroxyl methylation on the starting tyrosine derivative, followed by benzyloxy reduction to obtain the methyl ester intermediate.
- Conduct selective aromatic ring formylation using dichloromethyl ether and a Lewis acid catalyst such as titanium tetrachloride to generate the aldehyde intermediate.
- Execute Baeyer-Villiger oxidation on the aromatic aldehyde followed by ester reduction or hydrolysis to yield the final protected phenylalaninol or phenylalanine acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond mere technical feasibility. The primary driver of value is the drastic simplification of the supply chain for raw materials. By utilizing L-tyrosine derivatives as the starting point, manufacturers can bypass the need for exotic chiral pool precursors or expensive chiral catalysts that characterize older methods. This shift to commodity-grade starting materials inherently stabilizes the supply chain, reducing the risk of shortages that often plague the production of specialized oncology intermediates. Furthermore, the reduction in step count from lengthy multi-stage sequences to a concise five-step process directly correlates with reduced labor costs, lower solvent consumption, and decreased facility occupancy time, all of which contribute to significant cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The elimination of low-yielding steps such as the Reimer-Tiemann reaction (which previously yielded only 25-30%) fundamentally alters the cost structure of the final product. By improving the overall yield to approximately 70%, the amount of starting material required per kilogram of finished goods is drastically reduced. Additionally, the replacement of expensive reagents and complex chiral auxiliaries with common Lewis acids and peracids lowers the direct material costs. The avoidance of cryogenic conditions for extended periods and the ability to perform reactions in common solvents like methylene chloride and acetone further reduces utility and waste disposal expenses, creating a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent delivery timelines, a critical factor for pharmaceutical companies managing tight clinical trial schedules. The use of stable intermediates like the N-Cbz protected esters allows for potential stockpiling at various stages of the synthesis, providing buffer capacity against demand fluctuations. Moreover, the operational simplicity of the method, which avoids difficult-to-handle pyrophoric reagents or extremely sensitive organometallics, minimizes the risk of batch failures due to operator error or equipment malfunction. This reliability makes the supplier a more dependable partner for long-term contracts and just-in-time delivery models essential for modern pharmaceutical logistics.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns well with green chemistry principles. The Baeyer-Villiger oxidation generates fewer hazardous byproducts compared to heavy metal-catalyzed oxidations or the harsh basic waste streams of the Dakin reaction. The ability to run reactions at near-ambient temperatures and pressures facilitates easier scale-up from pilot plant to multi-ton commercial production without the need for specialized high-pressure or cryogenic reactors. This scalability ensures that the supply can grow in tandem with the clinical and commercial success of the downstream drug, while the cleaner reaction profile simplifies the environmental permitting process and reduces the burden on wastewater treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy for your technical due diligence. Understanding these nuances is essential for evaluating the feasibility of integrating this intermediate into your broader drug substance manufacturing strategy.
Q: What is the primary advantage of this synthesis route over the Reimer-Tiemann method?
A: The patented route replaces the low-yielding Reimer-Tiemann formylation (25-30% yield) and Dakin oxidation with a highly efficient selective formylation and Baeyer-Villiger oxidation sequence, boosting overall yield to approximately 70%.
Q: Is this process suitable for large-scale manufacturing of Ecteinascidin 743 intermediates?
A: Yes, the method utilizes cheap raw materials like L-tyrosine derivatives, avoids expensive chiral auxiliaries, and employs mild reaction conditions that are easily scalable for industrial production.
Q: What protecting groups are compatible with this synthetic strategy?
A: The process is optimized for Cbz (carbobenzoxy) protection but is adaptable to various carbalkoxy, acyl, or alkylsulfonyl groups ranging from C1 to C16, offering flexibility for downstream coupling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-3-Hydroxy-4-Methoxy-5-Methylphenylalanine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation oncology therapies depends on the secure supply of high-quality chiral intermediates. Our technical team has thoroughly analyzed the route described in patent CN101585745B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and state-of-the-art manufacturing facilities capable of meeting the stringent purity specifications required for GMP-grade API production. Our commitment to quality ensures that every batch of L-3-hydroxy-4-methoxy-5-methylphenylalanine delivered meets the exacting standards necessary for the synthesis of life-saving medicines like Trabectedin.
We invite you to collaborate with us to optimize your supply chain for these critical building blocks. By leveraging our expertise in process optimization and scale-up, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to accelerate your drug development timeline and ensure a uninterrupted supply of high-purity oncology intermediates for your global operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
