Revolutionizing Organocatalysis: Scalable Synthesis of High-Strain Sulfur-Phosphorus Derivatives for Industrial Applications
Revolutionizing Organocatalysis: Scalable Synthesis of High-Strain Sulfur-Phosphorus Derivatives for Industrial Applications
The landscape of organocatalysis is undergoing a significant transformation with the disclosure of patent CN108383876B, which introduces a novel class of sulfur-substituted quaternary pentavalent phosphorus derivatives. Unlike traditional nitrogen-based organocatalysts such as imines and amines that have dominated the field, these new phosphorus-containing compounds offer unique electronic properties derived from their highly strained four-membered ring structures. The integration of a sulfur atom adjacent to the phosphorus center fundamentally alters the electron density, creating a highly active catalytic site capable of facilitating complex transformations like the intramolecular Wittig reaction. For R&D directors and process chemists, this represents a pivotal shift towards more versatile and efficient catalytic systems that can access chemical space previously difficult to navigate with standard reagents. The structural novelty lies in the specific arrangement of substituents around the phosphetane oxide core, which balances stability with the necessary reactivity for catalytic turnover.
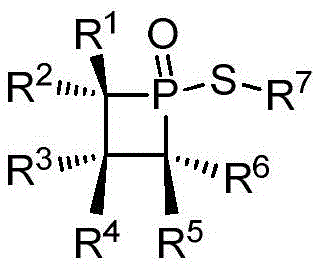
This technological breakthrough is not merely academic; it addresses critical pain points in the synthesis of high-value fine chemicals and pharmaceutical intermediates. The patent outlines a robust synthetic pathway that begins with readily available pentaalkyl propylene derivatives, reacting them with trivalent phosphorus sources under Lewis acid catalysis. This approach bypasses the need for expensive transition metal catalysts often associated with similar coupling reactions, thereby simplifying the impurity profile and reducing the burden on downstream purification processes. By establishing a reliable supply chain for these specialized organophosphorus catalysts, manufacturers can significantly enhance the efficiency of their production lines. The ability to source such high-performance catalysts from a reliable organophosphorus catalyst supplier ensures continuity in manufacturing schedules and mitigates the risks associated with supply chain disruptions common with rare-earth or precious metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organocatalysts has been heavily skewed towards nitrogen-containing species, which, while effective for many transformations, often struggle with specific redox cycles required for phosphorus-mediated chemistry. Conventional methods for generating active phosphorus species frequently involve harsh conditions, unstable intermediates, or the use of stoichiometric amounts of phosphine reagents that generate substantial waste. Furthermore, traditional phosphine oxides often lack the necessary lability in their P=O bonds to participate efficiently in catalytic cycles without demanding excessive energy input or aggressive reducing agents. This limitation restricts the scope of reactions that can be performed economically on a large scale. Additionally, the reliance on transition metals for similar transformations introduces concerns regarding heavy metal contamination in the final API, necessitating costly and time-consuming purification steps to meet stringent regulatory limits. These factors collectively contribute to higher operational expenditures and longer lead times for high-purity intermediates.
The Novel Approach
The methodology described in CN108383876B offers a paradigm shift by leveraging the inherent ring strain of a four-membered phosphorus heterocycle combined with the electronic influence of a sulfur substituent. As illustrated in the general synthesis scheme, the process initiates with the reaction of a polysubstituted propylene derivative with phosphorus trichloride in the presence of aluminum chloride. This Lewis acid-catalyzed step efficiently constructs the strained ring system, which is subsequently hydrolyzed to form a stable phosphine oxide intermediate. The final step involves a nucleophilic substitution with a thiol compound to install the sulfur moiety, completing the catalyst architecture. This route is characterized by its linearity, high atom economy, and the use of commodity chemicals, making it highly attractive for cost reduction in fine chemical manufacturing. The mild reaction conditions, typically ranging from 0°C to 60°C, further underscore the safety and scalability of this novel approach compared to high-temperature or high-pressure alternatives.
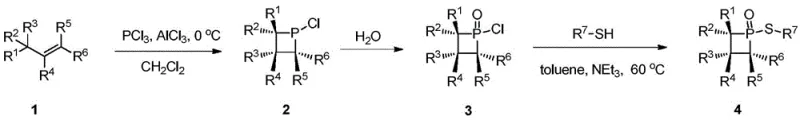
Mechanistic Insights into Sulfur-Substituted Phosphetane Oxide Catalysis
The exceptional catalytic activity of these derivatives stems from a sophisticated interplay between steric strain and electronic modulation. The four-membered phosphetane ring possesses significant angle strain, which destabilizes the ground state of the molecule and lowers the activation energy for bond cleavage. Crucially, the sulfur atom attached to the phosphorus center possesses two pairs of lone electrons that interact with the phosphorus d-orbitals. This interaction weakens the phosphorus-oxygen double bond (P=O), rendering it more susceptible to reduction. In the context of a catalytic cycle, such as the intramolecular Wittig reaction, this lability allows for the facile interconversion between the pentavalent phosphorus oxide (P=O) and the trivalent phosphine (P:) species. This redox flexibility is the engine of the catalytic cycle, enabling the catalyst to activate substrates, facilitate bond formation, and regenerate without being consumed. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters for specific substrate classes.
Furthermore, the substituent groups (R1 through R7) on the ring provide a tunable handle for modifying the catalyst's solubility, steric bulk, and electronic properties. For instance, varying the aryl or alkyl groups on the sulfur atom can fine-tune the nucleophilicity of the catalyst or its compatibility with different solvent systems. This modularity allows chemists to tailor the catalyst for specific applications, whether it be in non-polar hydrocarbon solvents or more polar media. The ability to control the impurity profile is also enhanced by the specificity of the Lewis acid-mediated cyclization, which tends to produce fewer side products compared to radical-based cyclization methods. This precision in molecular construction translates directly to higher purity specifications in the final product, reducing the need for extensive chromatographic purification and aligning with the industry's push towards greener, more sustainable synthesis protocols.
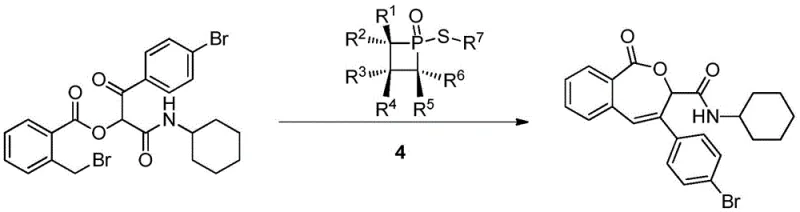
How to Synthesize 1-((4-chlorophenyl)thio)-2,2,3,4,4-pentamethylphosphetane 1-oxide Efficiently
The synthesis of these high-value catalysts follows a streamlined three-step protocol that is amenable to both laboratory discovery and pilot plant operations. The process begins with the careful addition of Lewis acid and phosphorus trichloride to a solution of the alkene precursor at controlled low temperatures to manage exotherms. Following the formation of the chlorophosphonium salt, a controlled hydrolysis step yields the oxidized ring intermediate. Finally, the installation of the sulfur group is achieved through a base-mediated substitution reaction in toluene. This sequence ensures high yields and reproducibility, as evidenced by the experimental data provided in the patent documentation. For detailed operational parameters, safety guidelines, and precise stoichiometric ratios required for GMP-compliant production, please refer to the standardized synthesis protocol below.
- React pentaalkyl propylene with PCl3 and AlCl3 in dichloromethane at 0°C to form the quaternary phosphorus chloride salt intermediate.
- Quench the reaction mixture with water under nitrogen protection, separate the organic phase, and isolate the white crystalline phosphine oxide intermediate.
- Perform nucleophilic substitution by reacting the intermediate with a thiol compound and triethylamine in toluene at 60°C to yield the final sulfur-substituted catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this technology offers substantial strategic advantages rooted in raw material availability and process simplicity. The synthesis relies on bulk commodities such as phosphorus trichloride, aluminum chloride, and substituted alkenes, all of which are produced on a massive global scale with stable pricing dynamics. This contrasts sharply with catalysts dependent on scarce precious metals like palladium or rhodium, whose prices are subject to volatile geopolitical fluctuations. By shifting to an organophosphorus system, companies can achieve significant cost reduction in manufacturing by eliminating the expense of noble metals and the associated recovery processes. Moreover, the absence of heavy metals simplifies the regulatory compliance landscape, particularly for pharmaceutical applications where residual metal limits are strictly enforced. This reduction in regulatory burden accelerates time-to-market for new drug candidates and reduces the overall cost of goods sold.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive, commodity-grade reagents directly lowers the bill of materials. The process operates at mild temperatures (0°C to 60°C), which significantly reduces energy consumption compared to high-temperature pyrolysis or reflux conditions often required in traditional heterocycle synthesis. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove by-products, thereby reducing solvent usage and waste disposal costs associated with purification. These factors combine to create a leaner, more cost-effective production model that enhances overall profit margins without compromising on quality.
- Enhanced Supply Chain Reliability: Sourcing strategies are strengthened by the reliance on widely available chemical feedstocks rather than specialized, single-source reagents. The synthetic route is robust and tolerant to minor variations in reaction conditions, ensuring consistent batch-to-bquality even when scaling up. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream customers. By diversifying the supply base away from critical raw materials prone to shortages, procurement managers can mitigate supply chain risks and ensure a steady flow of high-purity intermediates to support uninterrupted manufacturing operations.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, utilizing standard unit operations such as liquid-liquid extraction, filtration, and distillation that are commonplace in modern chemical plants. The reaction generates minimal hazardous waste, and the solvents used (dichloromethane, toluene) are easily recoverable and recyclable, supporting sustainability goals. The absence of toxic heavy metals in the catalyst structure simplifies effluent treatment and reduces the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating these catalysts into existing workflows and assess the potential return on investment. For further technical discussions or custom synthesis requests, our team is prepared to provide detailed route feasibility assessments.
Q: What is the primary advantage of the sulfur-substituted four-membered ring structure in this catalyst?
A: The four-membered ring introduces significant ring strain, while the sulfur atom's lone pair electrons modify the electronic structure of the phosphorus center. This combination drastically lowers the bond energy of the P=O double bond, facilitating easier reduction and conversion between P(V) and P(III) states, which is critical for catalytic cycling.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent demonstrates that the synthesis utilizes conventional reagents like PCl3, AlCl3, and substituted alkenes under mild conditions (0°C to 60°C). The steps involve standard unit operations such as stirring, quenching, and filtration, indicating high operability and feasibility for commercial scale-up without requiring exotic equipment.
Q: What specific reactions does this new catalyst facilitate?
A: The primary application demonstrated is the catalysis of intramolecular Wittig reactions. The catalyst acts as an efficient oxaphosphide reagent, playing an irreplaceable role in specific phosphorus-mediated transformations where traditional nitrogen-based organocatalysts may be ineffective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfur-Substituted Phosphorus Derivatives Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications for complex organophosphorus compounds, guaranteeing that every batch meets the highest international standards. We understand the critical nature of catalyst performance in your synthesis and are committed to delivering materials that consistently drive high yields and selectivity in your target reactions. Our expertise in handling sensitive phosphorus chemistry ensures safe and efficient production, minimizing risks and maximizing output for your commercial campaigns.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic technology can be integrated into your specific process requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this organophosphorus system. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your project needs. Together, we can accelerate your development timelines and secure a competitive advantage in the global market through superior chemical innovation and reliable supply chain execution.
