Advanced Catalytic Synthesis of Alpha-Hydroxy Phosphates Using Novel Ytterbium Complexes
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the demand for more efficient and environmentally benign synthetic routes, particularly for critical intermediates like alpha-hydroxy phosphates. These compounds serve as indispensable building blocks in the development of antiviral agents, anticancer therapeutics, and advanced enzyme inhibitors, while also finding extensive utility in polymer science and nanotechnology. A groundbreaking advancement in this field is detailed in Chinese Patent CN108250236B, which discloses a highly active preparation method for alpha-hydroxy phosphates utilizing a novel bis-beta-diimino divalent ytterbium complex. This technology represents a paradigm shift from conventional catalytic systems, offering a robust pathway that operates under mild, solvent-free conditions with exceptional atom economy. For global pharmaceutical and fine chemical manufacturers, this innovation presents a compelling opportunity to enhance process efficiency while adhering to increasingly stringent environmental regulations. The core of this breakthrough lies in the unique electronic and steric properties of the divalent ytterbium center coordinated by bulky beta-diimino ligands, which facilitates the Pudovik reaction with unprecedented activity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-hydroxy phosphates via the addition of diethyl phosphite to carbonyl compounds, known as the Pudovik reaction, has relied on a diverse array of catalytic systems ranging from inorganic acids and bases to various metal-organic complexes. However, many of these traditional methodologies suffer from significant drawbacks that hinder their scalability and economic viability in modern industrial settings. Conventional rare earth catalysts, such as trifluorosulfonic acid rare earth compounds or rare earth amides, often necessitate the use of volatile organic solvents and may require elevated temperatures or prolonged reaction times to achieve acceptable conversion rates. Furthermore, the reliance on trivalent rare earth species can sometimes lead to issues with catalyst stability and recovery, complicating the downstream purification processes. The presence of residual metals in the final product is a critical concern for pharmaceutical applications, often mandating expensive and time-consuming metal scavenging steps. Additionally, the use of stoichiometric amounts of reagents or high catalyst loadings in older protocols contributes to increased raw material costs and generates substantial chemical waste, conflicting with the principles of green chemistry that are now central to sustainable manufacturing strategies.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN108250236B introduces a sophisticated catalytic system based on a bis-beta-diimino divalent ytterbium complex, specifically designated as Yb(L2-Me)2(THF). This novel approach fundamentally alters the reaction landscape by enabling the synthesis to proceed under completely solvent-free conditions at room temperature, thereby eliminating the need for energy-intensive heating or cooling cycles. The catalyst exhibits remarkable activity, requiring only a minute loading of 0.2 to 1 mol% relative to the ketone substrate to drive the reaction to completion within a short timeframe of 10 to 25 minutes. This dramatic reduction in reaction time and catalyst quantity not only accelerates throughput but also minimizes the introduction of impurities into the reaction matrix. The versatility of this system is further evidenced by its broad substrate scope, effectively accommodating both aromatic ketones with varying electronic properties—such as those bearing fluoro, chloro, nitro, or methyl groups—and aliphatic ketones like acetone. 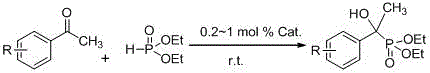
Mechanistic Insights into Bis-Beta-Diimino Divalent Ytterbium Catalysis
The exceptional performance of this catalytic system can be attributed to the unique coordination environment surrounding the divalent ytterbium ion. The beta-diimino ligand framework, characterized by its ability to finely tune steric hindrance and charge effects, stabilizes the divalent state of the ytterbium, which possesses a larger ionic radius and distinct reducibility compared to its trivalent counterparts. This electronic configuration allows the metal center to interact effectively with the phosphorus-hydrogen bond of the diethyl phosphite, facilitating the formation of a reactive intermediate that readily attacks the carbonyl carbon of the ketone. The specific structure of the catalyst, featuring 2-methylphenyl groups on the nitrogen atoms, provides an optimal balance of steric protection and electronic donation, preventing catalyst deactivation while maintaining high turnover frequencies. 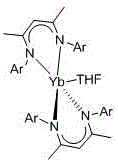
From an impurity control perspective, the mechanism offers distinct advantages over traditional acid or base-catalyzed routes. The mild, neutral nature of the rare earth catalysis minimizes side reactions such as aldol condensations or dehydration of the product, which are common pitfalls in harsher acidic or basic environments. The rapid reaction kinetics at room temperature further suppress the formation of thermal degradation byproducts, ensuring a cleaner crude reaction profile. This inherent selectivity simplifies the purification workflow, often allowing for simple washing procedures with non-polar solvents like n-hexane to isolate high-purity products, rather than requiring complex chromatographic separations. For R&D teams focused on process robustness, this mechanistic clarity translates to a more predictable and controllable manufacturing process with reduced risk of batch-to-batch variability.
How to Synthesize Alpha-Hydroxy Phosphate Efficiently
The implementation of this catalytic technology into a production environment is streamlined by its straightforward operational protocol, which eliminates the complexities associated with solvent handling and rigorous temperature control. The process begins with the preparation of the reaction mixture under an inert atmosphere, typically nitrogen, to protect the sensitive divalent ytterbium species from oxidation. The specific sequence of addition is critical; the catalyst is first mixed with diethyl phosphite to allow for pre-coordination before the introduction of the ketone substrate. This optimized feeding order has been shown to significantly enhance reaction yields, as demonstrated in experimental data where yields reached up to 99% for specific substrates like p-fluoroacetophenone. Following the reaction period, the workup involves a simple quenching step with water and extraction, leveraging the solubility differences to isolate the target alpha-hydroxy phosphate.
- Under a nitrogen atmosphere in a glove box, add the bis-beta-diimino divalent ytterbium complex catalyst to a reaction vessel.
- Add diethyl phosphite to the catalyst and stir at room temperature for 8 to 12 minutes to ensure proper mixing and activation.
- Add the ketone substrate (aromatic or aliphatic) to the mixture and react at room temperature for 10 to 25 minutes, followed by standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ytterbium-catalyzed process offers tangible strategic benefits that extend beyond mere technical performance. The elimination of organic solvents from the reaction phase represents a major cost reduction in fine chemical manufacturing, as it removes the expenses related to solvent procurement, storage, recycling infrastructure, and hazardous waste disposal. This solvent-free approach also inherently reduces the volumetric footprint of the reaction, allowing existing reactor capacity to produce significantly more product per batch without capital investment in larger vessels. Furthermore, the mild reaction conditions mitigate safety risks associated with exothermic runaway reactions or the handling of flammable solvents at elevated temperatures, contributing to a safer operational environment and potentially lower insurance premiums.
- Cost Reduction in Manufacturing: The drastic reduction in catalyst loading to the 0.2-1 mol% range directly lowers the raw material cost per kilogram of product, especially considering the specialized nature of rare earth complexes. By avoiding the use of stoichiometric reagents and expensive solvent systems, the overall variable cost of production is significantly compressed. The simplified post-treatment procedure, which often requires only washing and evaporation rather than column chromatography, reduces labor hours and consumable costs associated with purification media. These cumulative efficiencies create a leaner cost structure that enhances competitiveness in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the catalyst across a wide range of substrates ensures consistent supply continuity even when feedstock specifications vary slightly. The short reaction time of 10 to 25 minutes dramatically increases asset utilization rates, enabling faster turnaround times and more responsive fulfillment of customer orders. Since the process does not rely on specialized high-pressure or high-temperature equipment, it can be easily scaled or transferred between different manufacturing sites with minimal retrofitting, thereby diversifying supply sources and mitigating geopolitical or logistical risks.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of solvent-related heat transfer limitations, which are often the bottleneck in scaling exothermic reactions. The green chemistry attributes of the method, including atom economy and waste reduction, align perfectly with modern environmental compliance standards, reducing the regulatory burden and facilitating smoother permitting processes. This sustainability profile is increasingly valued by downstream customers who are under pressure to reduce the carbon footprint of their own supply chains, making this technology a key differentiator in B2B negotiations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production lines or new product development pipelines.
Q: What are the primary advantages of this ytterbium catalyst over traditional methods?
A: Unlike traditional systems that often require harsh conditions or expensive triflate salts, this bis-beta-diimino divalent ytterbium complex operates under solvent-free conditions at room temperature with extremely low catalyst loading (0.2-1 mol%), significantly simplifying post-treatment and reducing waste.
Q: Does this catalytic system support a wide range of ketone substrates?
A: Yes, the patent data demonstrates excellent substrate scope, successfully catalyzing reactions with various aromatic ketones containing electron-withdrawing or donating groups (such as fluoro, chloro, nitro, and methyl substituents) as well as aliphatic ketones like acetone and heterocyclic ketones.
Q: How does the solvent-free nature of this process impact manufacturing costs?
A: Eliminating the need for organic solvents removes the costs associated with solvent purchase, recovery, and disposal. Furthermore, the mild reaction conditions reduce energy consumption for heating or cooling, leading to substantial operational expenditure savings in large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the bis-beta-diimino divalent ytterbium complex in reshaping the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-scale operations. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha-hydroxy phosphate delivered meets the exacting standards required for drug substance manufacturing. We are committed to leveraging our technical expertise to optimize this solvent-free route for maximum efficiency and yield.
We invite forward-thinking partners to collaborate with us to explore the full commercial potential of this innovative synthesis method. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive value and resilience in your supply chain.
