Advanced Iridium-Catalyzed Synthesis of Chiral 3-Allylindole Compounds for Scalable Pharmaceutical Manufacturing
Advanced Iridium-Catalyzed Synthesis of Chiral 3-Allylindole Compounds for Scalable Pharmaceutical Manufacturing
The landscape of organic synthesis for life science applications is constantly evolving, driven by the demand for higher purity and more efficient access to complex chiral scaffolds. Patent CN112876401A introduces a groundbreaking methodology for the synthesis of chiral 3-allylindole compounds, a structural motif that serves as the core skeleton for numerous natural alkaloids and bioactive pharmaceutical ingredients. This innovation addresses critical bottlenecks in current manufacturing by utilizing a sophisticated dual-catalytic system comprising chiral iridium, a specialized chiral ligand, and cuprous trifluoromethanesulfonate. By enabling a direct, one-step transformation of indoles and vinylcyclopropanes under mild thermal conditions, this technology offers a pathway to high-value intermediates with exceptional atom economy and environmental compatibility, positioning it as a vital asset for modern fine chemical supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral 3-allylindole frameworks has been plagued by significant synthetic challenges that hinder efficient commercial production. Traditional routes often rely on multi-step sequences involving tedious protection and de-protection strategies, which inherently lower the overall yield and increase waste generation. Furthermore, many established methods require drastic reaction conditions, such as strong Lewis acids or cryogenic temperatures, which pose safety risks and complicate process control in a manufacturing setting. These conventional approaches frequently suffer from poor enantioselectivity, necessitating costly and time-consuming chiral resolution steps to isolate the desired optical isomer. The cumulative effect of these inefficiencies results in elevated production costs and extended lead times, creating a substantial burden for procurement teams managing the supply of critical pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112876401A presents a streamlined, catalytic solution that fundamentally reshapes the synthetic landscape for these compounds. The novel approach leverages a highly active iridium-based catalytic system to facilitate a direct asymmetric allylic alkylation, effectively merging bond formation and stereocontrol into a single operational step. As illustrated in the reaction scheme below, the process proceeds smoothly in tetrahydrofuran at a moderate temperature of 50°C, eliminating the need for extreme thermal inputs or hazardous reagents. This methodological shift not only simplifies the operational workflow but also ensures high atom economy, as the vinylcyclopropane reactant is efficiently incorporated into the final product structure with minimal byproduct formation.
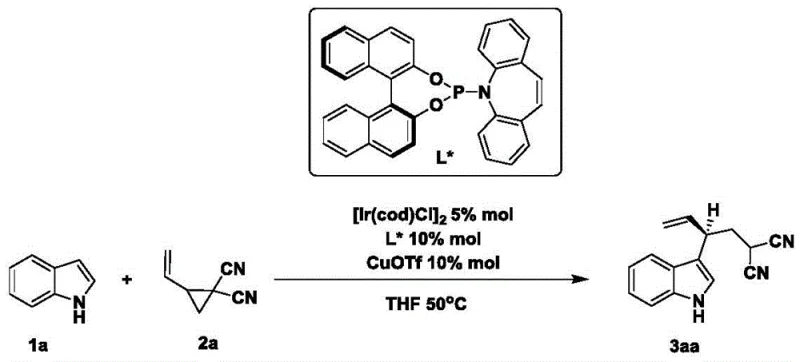
Mechanistic Insights into Iridium-Catalyzed Asymmetric Allylic Alkylation
The success of this synthetic route hinges on the precise orchestration of the catalytic cycle involving the iridium precursor [Ir(cod)Cl]2 and the chiral phosphoramidite ligand L*. Upon activation by the copper co-catalyst (CuOTf), the iridium center generates a cationic species capable of oxidative addition into the strained carbon-carbon bond of the vinylcyclopropane. This step is critical as it forms a reactive pi-allyl iridium intermediate, which serves as the electrophilic partner in the subsequent nucleophilic attack by the indole. The chiral environment created by the bulky binaphthyl backbone of the ligand L* exerts strict stereochemical control over this attack, ensuring that the new carbon-carbon bond is formed with high facial selectivity. This mechanistic precision is what allows the process to achieve enantiomeric ratios as high as 97:3, effectively bypassing the need for downstream chiral purification.
Furthermore, the inclusion of CuOTf plays a pivotal role in modulating the reactivity of the indole nucleophile and stabilizing the transition state. The copper salt likely assists in the generation of the active catalytic species and may also coordinate with the nitrogen of the indole or the nitrile/ester groups on the cyclopropane, enhancing the regioselectivity for C3-alkylation over N-alkylation. This dual-metal synergy ensures that the reaction proceeds with excellent chemoselectivity, tolerating a wide array of functional groups on the indole ring without requiring additional protecting groups. The robustness of this mechanism against electronic variations—whether electron-rich methoxy groups or electron-deficient halogens—is a testament to the versatility of the catalytic system, making it a reliable tool for synthesizing diverse libraries of chiral building blocks.
How to Synthesize Chiral 3-Allylindole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and stereoselectivity. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent catalyst deactivation by oxygen or moisture. The standardized protocol involves dissolving the indole substrate and the vinylcyclopropane derivative in anhydrous tetrahydrofuran, followed by the sequential addition of the iridium precatalyst, the chiral ligand, and the copper salt. The detailed standardized synthesis steps are provided in the guide below, outlining the exact molar ratios and workup procedures required to replicate the high performance reported in the patent data.
- Combine indole substrate and vinylcyclopropane derivative in tetrahydrofuran (THF) solvent under inert atmosphere.
- Add catalytic amounts of [Ir(cod)Cl]2, chiral ligand L*, and CuOTf to the reaction mixture.
- Stir the reaction at 50°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iridium-catalyzed technology translates into tangible strategic advantages regarding cost stability and supply reliability. By condensing what was traditionally a multi-step synthesis into a single pot operation, the process drastically reduces the consumption of solvents, reagents, and labor hours associated with intermediate isolation and purification. The mild reaction conditions (50°C) and the use of common solvents like THF further lower the energy footprint and infrastructure requirements, allowing for production in standard stainless steel reactors without the need for specialized cryogenic equipment. These factors collectively contribute to a significantly reduced cost of goods sold (COGS), enabling more competitive pricing for downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of protecting group manipulations and chiral resolution steps represents a major driver for cost optimization in this process. Traditional routes often incur heavy expenses due to the loss of material during resolution (maximum 50% theoretical yield) and the cost of chiral stationary phases; this catalytic method circumvents those losses entirely by delivering high enantiomeric excess directly. Additionally, the high atom economy means that a greater proportion of the raw material mass ends up in the final product, reducing waste disposal costs and improving overall material efficiency. This lean manufacturing approach ensures that the production of high-purity chiral 3-allylindoles remains economically viable even at large scales.
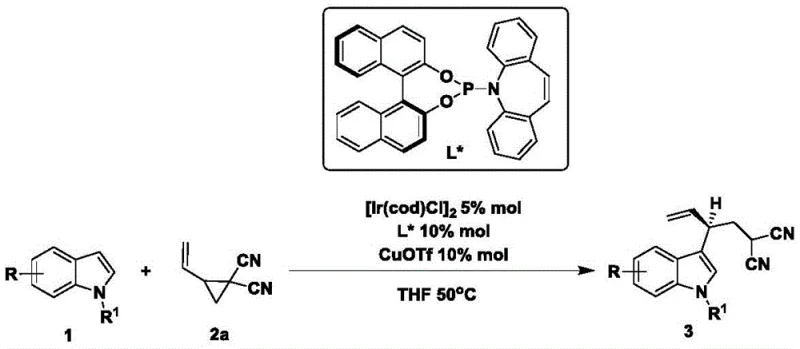
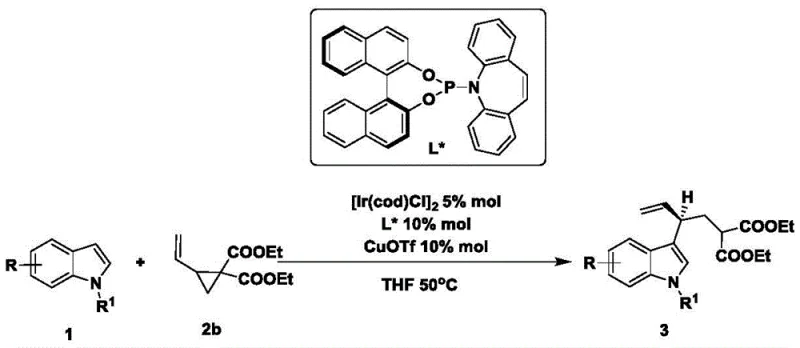
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against substrate variation ensures a stable supply of diverse intermediates regardless of specific structural requirements. As demonstrated by the broad substrate scope, the process tolerates various substituents on the indole ring, including halogens and alkyl groups, which are common in drug candidates. This flexibility means that supply chain disruptions caused by the unavailability of highly specialized, custom-synthesized precursors are minimized, as the method can adapt to commercially available starting materials. Consequently, lead times for high-purity pharmaceutical intermediates can be shortened, providing a buffer against market volatility and ensuring continuous availability for clinical and commercial programs.
- Scalability and Environmental Compliance: The simplicity of the post-reaction workup, involving basic filtration and silica gel chromatography, facilitates easy scale-up from gram to kilogram quantities without complex engineering changes. The process avoids the use of toxic heavy metals in stoichiometric amounts, relying instead on catalytic quantities of iridium and copper, which aligns with increasingly stringent environmental regulations regarding heavy metal residues in APIs. The environmentally friendly nature of the process, combined with its high yield and selectivity, supports sustainable manufacturing practices and reduces the regulatory burden associated with impurity profiling and solvent recovery, making it an ideal candidate for green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific target molecules and for procurement teams assessing the long-term viability of the supply chain.
Q: What are the advantages of this iridium-catalyzed method over traditional indole alkylation?
A: This method operates under mild conditions (50°C) in a single step, avoiding the harsh reagents and multi-step sequences typical of conventional approaches, while delivering superior enantioselectivity (up to 97:3 er).
Q: Is this process suitable for large-scale production of pharmaceutical intermediates?
A: Yes, the process utilizes readily available starting materials like indoles and vinylcyclopropanes, employs a robust catalytic system, and involves simple post-treatment workups, making it highly amenable to industrial scale-up.
Q: What types of substituents are tolerated on the indole ring?
A: The catalytic system demonstrates broad substrate scope, successfully accommodating electron-donating groups (e.g., methyl, methoxy) and electron-withdrawing groups (e.g., fluoro, bromo, chloro) at various positions on the indole nucleus.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3-Allylindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iridium-catalyzed synthesis for the production of next-generation pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, guaranteeing that every batch of chiral 3-allylindole meets the exacting standards required for global regulatory submissions. We are committed to leveraging this innovative chemistry to deliver high-quality intermediates that accelerate our partners' drug development timelines.
We invite you to collaborate with us to explore how this technology can optimize your specific project requirements. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating the economic benefits of switching to this catalytic route. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary candidates, and let us help you secure a reliable and cost-effective supply of these critical chiral building blocks.
