Advanced One-Step Diazotization Strategy for High-Purity Uridine Commercial Manufacturing
Advanced One-Step Diazotization Strategy for High-Purity Uridine Commercial Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for producing critical nucleoside intermediates. A recent technological breakthrough disclosed in patent CN113735926A presents a highly efficient synthesis process for uridine, a pivotal compound used in the treatment of liver diseases, cardiovascular conditions, and as a key precursor for antiviral agents like fluorouracil. This innovation shifts the paradigm from traditional, labor-intensive biological fermentation or multi-step chemical syntheses to a streamlined, one-step diazotization reaction performed in an aqueous medium. By leveraging cytidine as a readily available starting material, this method achieves exceptional conversion rates exceeding 90% and yields surpassing 80%, while maintaining a product purity of greater than 99.5%. For global procurement leaders and R&D directors, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates, reducing dependency on complex bioprocesses and enhancing overall manufacturing efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of uridine has been dominated by biological fermentation methods or complex chemical synthesis routes starting from uracil. The fermentation approach, while biologically elegant, is plagued by inherent limitations such as lengthy production cycles, complex downstream purification requirements to remove biological impurities, and significant batch-to-batch variability that complicates quality control. Alternatively, the traditional chemical synthesis from uracil involves a tedious sequence of acetylation, condensation, hydrolysis, alcoholysis, and crystallization steps. Each of these stages introduces potential yield losses, requires hazardous reagents, and generates substantial waste streams, thereby inflating the cost of goods sold (COGS) and extending the lead time for high-purity pharmaceutical intermediates. These inefficiencies create bottlenecks in the supply chain, making it difficult for manufacturers to respond agilely to market demands for nucleoside-based therapeutics.
The Novel Approach
In stark contrast, the novel process described in the patent utilizes a direct deamination strategy, converting cytidine directly into uridine through a controlled diazotization reaction. This method eliminates the need for protecting group manipulations and multi-step functional group transformations that characterize older chemical routes. By conducting the reaction in an aqueous solution at moderate temperatures ranging from 10 to 60°C, the process ensures safety and operational simplicity. The use of inexpensive and abundant reagents like sodium nitrite and common mineral acids further drives down raw material costs. Furthermore, the post-treatment involves a straightforward pH adjustment and crystallization using common alcohols, which simplifies isolation and significantly reduces solvent consumption compared to traditional organic synthesis. This streamlined workflow not only accelerates the synthesis cycle but also enhances the environmental profile of the manufacturing process, aligning with modern green chemistry principles.
Mechanistic Insights into Cytidine Diazotization and Hydrolysis
The core of this innovative synthesis lies in the precise mechanistic execution of the diazotization of the exocyclic amine group on the cytosine base of the cytidine molecule. Under acidic conditions, the nitrite reagent generates nitrous acid in situ, which reacts with the primary amine to form an unstable diazonium salt intermediate. This intermediate is highly reactive and rapidly undergoes hydrolysis in the aqueous medium, replacing the diazonium group with a hydroxyl group that subsequently tautomerizes to the ketone form found in the uracil base. The critical challenge in this transformation is preserving the integrity of the N-glycosidic bond connecting the base to the ribose sugar, which is susceptible to acid-catalyzed hydrolysis. The patent specifies a tightly controlled pH range of 3.5 to 4.5 during the addition of reagents, which is essential to balance the rate of diazotization against the stability of the glycosidic linkage, preventing the formation of free uracil and ribose by-products.
Following the formation of the uridine species in the aqueous solution, the process employs a sophisticated purification mechanism to ensure the high purity required for API applications. The reaction mixture is first concentrated under reduced pressure at a controlled pH of 1 to 5 to remove volatile by-products and excess acid gases. Subsequently, the pH is adjusted to a near-neutral range of 5 to 7 before final concentration. This pH swing is crucial for maximizing the recovery of the product while minimizing degradation. The final crystallization step utilizes alcoholic solvents such as ethanol or methanol, which selectively precipitate the uridine while keeping impurities in solution. Data indicates that ethanol yields superior results in terms of both recovery and purity compared to other solvents, highlighting the importance of solvent selection in the crystallization thermodynamics. This rigorous control over reaction and purification parameters ensures a consistent impurity profile, which is vital for regulatory compliance in pharmaceutical manufacturing.
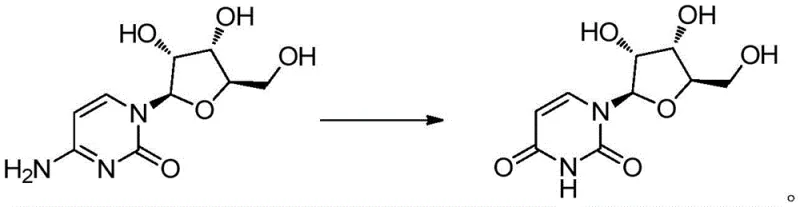
How to Synthesize Uridine Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent literature. The process begins with the preparation of two distinct feed streams: an acid salt solution of cytidine and an aqueous solution of the nitrite reagent. These streams are introduced simultaneously into a reactor containing water, maintained at a temperature between 10 and 60°C. The dropwise addition is controlled over a period of 1 to 5 hours to manage the exotherm and maintain the critical pH window. Following the addition, the mixture is held at temperature for 1 to 10 hours to ensure complete conversion, as monitored by HPLC. The detailed standardized operating procedures, including specific molar ratios, agitation speeds, and filtration protocols necessary for GMP-compliant production, are outlined in the technical guide below.
- Prepare cytidine acid salt solution and nitrite reagent solution separately, maintaining specific molar ratios for optimal reaction kinetics.
- Add both solutions dropwise into a temperature-controlled reactor (10-60°C), maintaining pH between 3.5 and 4.5 to facilitate diazotization without degrading the glycosidic bond.
- Post-reaction, adjust pH to 1-5 for concentration, neutralize to pH 5-7, and induce crystallization using alcoholic solvents like ethanol or methanol to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this diazotization-based synthesis route offers transformative benefits that extend beyond simple yield improvements. The shift from fermentation or multi-step organic synthesis to this direct aqueous conversion fundamentally alters the cost structure and risk profile of uridine production. By utilizing cytidine, a commodity chemical produced in large volumes with stable pricing, manufacturers can decouple their supply from the volatility associated with specialized fermentation substrates or complex protected sugars. The elimination of multiple reaction steps reduces the requirement for intermediate isolation, drying, and quality testing, which translates into substantial operational cost savings and a reduced carbon footprint. Furthermore, the use of water as the primary reaction solvent minimizes the need for expensive, hazardous organic solvents during the reaction phase, lowering waste disposal costs and enhancing workplace safety standards.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the direct deamination reaction and the use of low-cost inorganic reagents. Unlike traditional routes that require expensive protecting groups and anhydrous conditions, this method operates in water, drastically reducing solvent procurement and recycling costs. The high conversion rate means that less raw material is wasted, and the simplified downstream processing reduces energy consumption associated with distillation and drying. These factors combine to lower the overall cost of goods, allowing for more competitive pricing strategies in the global market for nucleoside intermediates without compromising on margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the reliance on widely available starting materials. Cytidine and sodium nitrite are bulk chemicals with established supply chains, reducing the risk of shortages that often plague specialty fermentation broths or custom-synthesized intermediates. The robustness of the chemical process, which tolerates a broad range of operating conditions while maintaining high purity, ensures consistent output even during scale-up. This reliability allows for better production planning and shorter lead times, enabling pharmaceutical companies to maintain leaner inventories and respond more quickly to fluctuations in demand for antiviral and oncology drugs.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the aqueous nature of the reaction simplifies the transition from laboratory to commercial scale. Water has excellent heat transfer properties, making temperature control easier in large reactors compared to viscous fermentation broths or organic solvent mixtures. The process generates minimal hazardous waste, primarily consisting of saline solutions that are easier to treat than complex organic waste streams. This alignment with green chemistry principles facilitates regulatory approval and reduces the burden of environmental compliance, making it an attractive option for manufacturers looking to expand capacity in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this uridine synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, aiming to clarify the feasibility and advantages for potential adopters. Understanding these nuances is critical for R&D teams evaluating process transfer and for procurement specialists assessing the long-term viability of this supply source.
Q: What are the primary advantages of this diazotization method over biological fermentation?
A: Unlike fermentation, which suffers from complex downstream processing and biological variability, this chemical route offers a significantly shorter synthesis cycle, higher conversion rates exceeding 90%, and easier scalability for industrial production.
Q: How does the process ensure high purity suitable for API manufacturing?
A: The process utilizes a specific post-treatment purification involving pH-controlled concentration and alcoholic crystallization, which effectively removes impurities and by-products, consistently achieving purity levels greater than 99.5%.
Q: Is the raw material cytidine readily available for large-scale production?
A: Yes, cytidine is currently produced in large batches domestically with sufficient supply and low cost, making this synthetic route highly viable for commercial scale-up and ensuring supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Uridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient nucleoside synthesis in the modern pharmaceutical landscape. As a leading CDMO and fine chemical manufacturer, we possess the technical expertise and infrastructure to translate innovative patent technologies like the cytidine diazotization route into commercial reality. Our facilities are equipped to handle diverse synthetic pathways, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our team understands the critical nature of supply continuity for API manufacturers and is dedicated to providing a stable, high-quality source of uridine.
We invite global partners to collaborate with us to leverage this advanced synthesis technology for their specific product pipelines. Whether you require custom synthesis services or bulk supply of uridine, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out to our technical procurement team to obtain specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project goals and quality standards.
