Scalable Manufacturing of DOTA-EB-PSMA Precursors for Next-Generation Prostate Cancer Radioligand Therapy
The pharmaceutical landscape for targeted prostate cancer treatment is undergoing a significant transformation with the advent of advanced radioligand therapies (RLT), specifically those utilizing PSMA-targeting agents. A pivotal development in this domain is documented in patent CN115947775B, which discloses a novel and highly efficient method for preparing Compound (I), also known as DOTA-EB-PSMA. This compound serves as a critical precursor for radiopharmaceuticals that combine a tumor-targeting molecule with a radioisotope, enabling precise DNA damage within cancer cells while sparing healthy tissue. The technical breakthrough presented in this patent addresses the longstanding challenges of scalability and purity that have hindered the widespread clinical adoption of such therapies. By re-engineering the synthetic route, the inventors have achieved a process that not only meets the rigorous quality standards of bulk drugs but also facilitates commercial-scale production. For R&D Directors and Supply Chain Heads, this represents a viable pathway to secure a reliable radiopharmaceutical intermediate supplier capable of delivering high-purity materials consistently. The implications of this technology extend beyond mere chemical synthesis; it offers a strategic advantage in cost reduction in radioligand therapy manufacturing by streamlining purification bottlenecks that traditionally plague complex peptide-conjugate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN115947775B, the synthesis of DOTA-EB-PSMA and similar radiopharmaceutical precursors was fraught with inefficiencies that made industrial scale-up nearly impossible. The conventional methods, often referenced in earlier literature and patents such as CN111741751A, relied heavily on repetitive preparative high-performance liquid chromatography (HPLC) for the purification of every single intermediate. This dependency created a severe bottleneck, as each HPLC purification step typically required 4 to 5 days to complete, leading to excessively long production cycles. Furthermore, the repeated exposure of sensitive intermediates to chromatographic conditions often resulted in decomposition, significantly lowering the overall yield and compromising the economic viability of the process. The quality standards achieved were also suboptimal, with most intermediates failing to exceed 90% purity and total impurities often surpassing the 2% limit required for therapeutic applications. These technical barriers meant that prior art methods were suitable only for small-scale research samples, lacking the robustness needed for commercial scale-up of complex radiopharmaceutical precursors.
The Novel Approach
The methodology introduced in patent CN115947775B fundamentally restructures the synthesis pathway to overcome these historical limitations through a strategic reduction in purification complexity. Instead of subjecting every intermediate to resource-intensive preparative HPLC, the new process limits high-efficiency liquid phase separation to only two critical stages: the purification of Compound 2 and the final Compound (I). The intermediates Compound 3 and Compound 4 are purified using crystallization and slurrying techniques, which are far more amenable to large-scale manufacturing. This hybrid approach drastically simplifies the workflow, reducing the production time by at least 8 to 10 days per batch compared to the prior art. The visual representation of this streamlined route highlights the logical progression from EB-Lys-Boc to the final DOTA-EB-PSMA structure, emphasizing the points where purification strategies diverge from traditional norms. 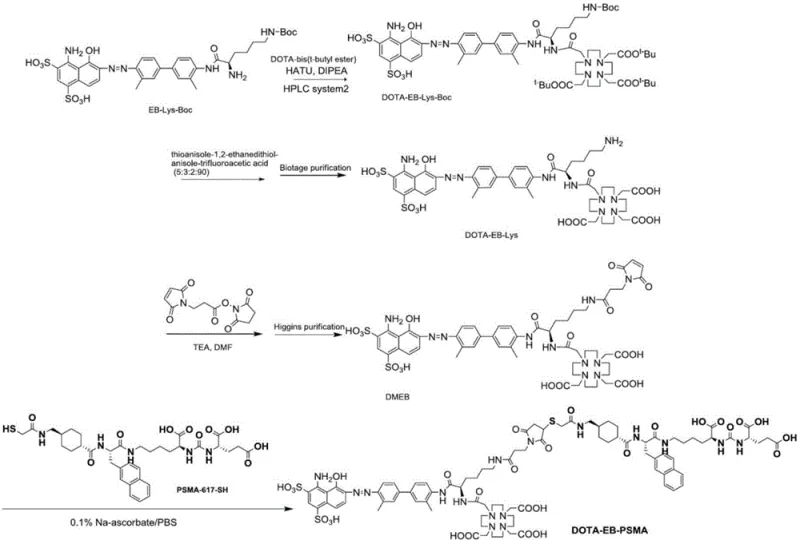 This optimization not only enhances production efficiency but also ensures that the purity of the final product meets the stringent requirements for clinical use, with total impurities controlled well below the 2% threshold. For procurement managers, this translates to a more reliable supply chain with reduced lead time for high-purity prostate cancer imaging agents, mitigating the risks associated with production delays and material shortages.
This optimization not only enhances production efficiency but also ensures that the purity of the final product meets the stringent requirements for clinical use, with total impurities controlled well below the 2% threshold. For procurement managers, this translates to a more reliable supply chain with reduced lead time for high-purity prostate cancer imaging agents, mitigating the risks associated with production delays and material shortages.
Mechanistic Insights into HATU-Mediated Condensation and Conjugation
The chemical robustness of the new synthesis route relies on precise control over reaction conditions and reagent stoichiometry to ensure high fidelity in bond formation. The initial condensation reaction between Compound 1 and purified DOTA-tri-tert-butyl ester is catalyzed by HATU (2-(7-Azabenzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate) in the presence of DIPEA as a base. This coupling mechanism is critical for forming the amide bond that links the Evans Blue derivative to the DOTA chelator, a structural motif essential for subsequent radiolabeling with isotopes like Lutetium-177. The reaction is conducted at controlled temperatures between 0°C and 25°C to minimize side reactions and racemization, ensuring the stereochemical integrity of the chiral centers within the molecule. Following this, the de-protection of the Boc group using trifluoroacetic acid and triisopropylsilane must be carefully managed to prevent acid-induced degradation of the sensitive azo-dye moiety. The subsequent conjugation steps involve reacting the amine-functionalized intermediate with 3-maleimidopropionic acid hydroxysuccinimide ester, which introduces the maleimide group necessary for thiol conjugation. This step is performed in anhydrous DMF under inert gas protection to prevent hydrolysis of the activated ester, demonstrating the high level of process control required for commercial scale-up of complex polymer additives and pharmaceutical intermediates.
Impurity control is a paramount concern in the synthesis of radiopharmaceutical precursors, as even trace contaminants can affect the biodistribution and safety profile of the final drug. The new process addresses this by implementing a rigorous purification strategy for Compound 2 using reverse-phase preparative HPLC with a specific gradient elution profile involving ammonium acetate and trifluoroacetic acid. This ensures that the key intermediate reaches a purity of 99.64% before proceeding to subsequent steps, thereby preventing the carryover of impurities that could complicate downstream purification. The final conjugation reaction between Compound 4 and Compound 5 (PSMA-617-SH) is conducted in an aqueous buffer system containing L-ascorbic acid, which acts as an antioxidant to protect the thiol group from oxidation. 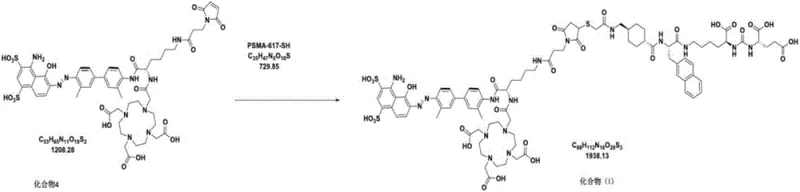 This final step is purified using a specialized HPLC method with a triethylamine-phosphoric acid buffer system, which effectively separates the target Compound (I) from unreacted starting materials and byproducts. The result is a final product with a purity of 99.36% and single impurities below 0.5%, meeting the highest quality specifications for API intermediates. This level of purity is essential for R&D Directors who need to ensure that the material is suitable for clinical trials and regulatory submission without the need for extensive re-processing.
This final step is purified using a specialized HPLC method with a triethylamine-phosphoric acid buffer system, which effectively separates the target Compound (I) from unreacted starting materials and byproducts. The result is a final product with a purity of 99.36% and single impurities below 0.5%, meeting the highest quality specifications for API intermediates. This level of purity is essential for R&D Directors who need to ensure that the material is suitable for clinical trials and regulatory submission without the need for extensive re-processing.
How to Synthesize DOTA-EB-PSMA Efficiently
The implementation of this synthesis route requires a detailed understanding of the specific reaction parameters and purification techniques outlined in the patent to ensure reproducibility and quality. The process begins with the purification of the DOTA-tri-tert-butyl ester starting material, which is critical for the success of the first condensation step. Operators must strictly adhere to the specified molar ratios of reagents, such as the 1.0:2.0-4.0 ratio of Compound 1 to DOTA-tri-tert-butyl ester, to maximize conversion while minimizing waste. The purification of intermediates via crystallization requires precise control of solvent composition and temperature, utilizing solvents like methyl tert-butyl ether to induce precipitation of the product while leaving impurities in solution. Detailed standardized synthesis steps are essential for maintaining consistency across batches, particularly when scaling from laboratory to pilot plant operations. The following guide outlines the critical operational parameters for each stage of the synthesis.
- Condense Compound 1 with purified DOTA-tri-tert-butyl ester using HATU and DIPEA to form Compound 2, followed by preparative HPLC purification.
- Remove the Boc protecting group from Compound 2 using trifluoroacetic acid and triisopropylsilane to yield Compound 3, purified via crystallization.
- React Compound 3 with 3-maleimidopropionic acid hydroxysuccinimide ester to form Compound 4, purified by slurrying and filtration.
- Conjugate Compound 4 with Compound 5 (PSMA-617-SH) in aqueous buffer with L-ascorbic acid, followed by final HPLC purification to obtain Compound (I).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the technical improvements in the synthesis of DOTA-EB-PSMA offer substantial benefits for procurement and supply chain management, primarily through the optimization of resource utilization and production throughput. The reduction in the number of preparative HPLC steps from four to two represents a significant decrease in the consumption of expensive chromatography resins, organic solvents, and operational labor hours. This streamlining directly contributes to cost reduction in radioligand therapy manufacturing by lowering the variable costs associated with each production batch. Furthermore, the replacement of HPLC purification with crystallization for intermediates Compound 3 and Compound 4 enhances the scalability of the process, as crystallization is a unit operation that is easily scaled from grams to kilograms without the linear increase in cost and time seen with chromatography. This scalability ensures that the supply chain can respond flexibly to fluctuating demand for prostate cancer imaging agents without the risk of bottlenecks that typically constrain peptide-based manufacturing.
- Cost Reduction in Manufacturing: The elimination of two preparative HPLC purification steps significantly lowers the operational expenditure associated with solvent purchase, waste disposal, and column maintenance. By relying on crystallization and slurrying for intermediate purification, the process reduces the dependency on high-cost consumables and specialized equipment, leading to substantial cost savings over the lifecycle of the product. This economic efficiency makes the commercial production of high-purity OLED material and pharmaceutical intermediates more viable, allowing for competitive pricing strategies in the global market. The qualitative improvement in process efficiency ensures that the economic benefits are no longer difficult to guarantee, as was the case with prior art methods.
- Enhanced Supply Chain Reliability: The shortened production cycle, which saves at least 8 to 10 days per batch, directly improves the agility of the supply chain, enabling faster response times to customer orders and reducing inventory holding costs. The robustness of the crystallization steps ensures that the process is less susceptible to the variability and downtime often associated with complex chromatographic separations. This reliability is crucial for reducing lead time for high-purity prostate cancer imaging agents, ensuring that clinical trials and commercial launches are not delayed by material shortages. A reliable radiopharmaceutical intermediate supplier must be able to demonstrate such process stability to gain the trust of multinational pharmaceutical partners.
- Scalability and Environmental Compliance: The shift towards crystallization-based purification reduces the volume of organic waste generated per kilogram of product, aligning the manufacturing process with stricter environmental regulations and sustainability goals. The ability to perform bulk production in batches meets the growing demand for radioligand therapies without compromising on quality or safety standards. This scalability supports the commercial scale-up of complex radiopharmaceutical precursors, ensuring a continuous supply of materials needed for the expanding field of targeted cancer therapy. The process design inherently supports green chemistry principles by minimizing solvent usage and maximizing atom economy in the coupling reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of DOTA-EB-PSMA, based on the specific data and claims presented in patent CN115947775B. These answers are designed to provide clarity on the quality standards, process capabilities, and regulatory compliance of the material. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this precursor into their drug development pipelines. The information provided reflects the current state of the art in radiopharmaceutical manufacturing and highlights the competitive advantages of the new method.
Q: How does the new synthesis method improve purity compared to prior art?
A: The new method reduces the number of preparative HPLC purifications from four to two, utilizing crystallization for intermediates. This minimizes decomposition risks and ensures total impurities remain below 2%, meeting bulk drug quality standards.
Q: What are the scalability advantages of this process for supply chains?
A: By replacing multiple HPLC steps with crystallization and slurrying for intermediates Compound 3 and 4, the process eliminates significant bottlenecks, allowing for batch production and reducing lead time by at least 8 to 10 days per cycle.
Q: Is the final compound suitable for clinical radioligand therapy?
A: Yes, the final Compound (I) achieves a purity of 99.36% with single impurities below 0.5%, satisfying the stringent quality requirements for radiopharmaceutical precursors used in targeted prostate cancer treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DOTA-EB-PSMA Supplier
The technical potential of the DOTA-EB-PSMA synthesis route described in CN115947775B is best realized through partnership with an experienced CDMO capable of executing complex chemical pathways with precision and consistency. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. Our facility is equipped with stringent purity specifications and rigorous QC labs that are fully capable of validating the 99.36% purity and impurity profiles required for this critical radiopharmaceutical intermediate. We understand the critical nature of supply continuity in the pharmaceutical industry and have structured our operations to guarantee the availability of high-purity PSMA targeting agent materials for our global clients.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements and timelines. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of adopting this streamlined manufacturing process for your supply chain. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver on the promises of this innovative patent. Let us collaborate to advance the availability of life-saving radioligand therapies through superior chemical manufacturing and supply chain excellence.
