Scalable Water-Based Synthesis of N,N'-Dicyclohexylthiourea for Pharmaceutical Applications
Scalable Water-Based Synthesis of N,N'-Dicyclohexylthiourea for Pharmaceutical Applications
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, safer, and more cost-effective processes, particularly for critical pharmaceutical intermediates. A pivotal development in this domain is detailed in patent CN102731357B, which outlines a novel preparation method for high-purity N,N'-dicyclohexylthiourea. This compound serves as a vital precursor in the synthesis of biological polypeptide condensing agents, making its production efficiency paramount for downstream API manufacturing. The disclosed technology fundamentally reimagines the synthesis pathway by replacing hazardous aromatic hydrocarbon solvents with water, thereby addressing long-standing safety and environmental concerns inherent in legacy production methods. By leveraging a precise aqueous phase reaction followed by a specialized vacuum thermal decomposition step, this methodology achieves exceptional yields exceeding 99% and purity levels greater than 99.5%. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages of this water-based route is essential for optimizing supply chain resilience and reducing overall production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N,N'-dicyclohexylthiourea has relied heavily on the reaction of cyclohexylamine with carbon disulfide in the presence of organic solvents such as benzene, toluene, or xylene. While these aromatic hydrocarbon-based methods have supported industrial-scale output, they are fraught with significant operational drawbacks that modern manufacturing standards can no longer tolerate. The primary concern is safety; the solvents used are highly flammable and volatile, creating substantial explosion risks within the plant environment and necessitating expensive explosion-proof infrastructure. Furthermore, the toxicity of these solvents poses severe health risks to operators and complicates regulatory compliance regarding worker safety and environmental emissions. From an economic perspective, the loss of solvent during the production cycle—through evaporation or retention in the product matrix—drives up raw material costs significantly. Alternative methods attempting to use water with catalysts like zinc oxide or aluminum oxide have failed to achieve industrial viability due to poor suitability for large-scale operations, while other aqueous approaches involving strong bases like sodium hydroxide require complex extraction and purification steps that lower overall yield to around 90% and generate excessive wastewater.
The Novel Approach
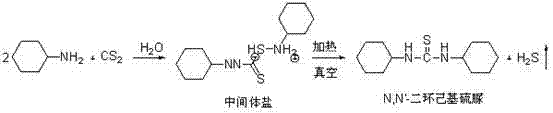
The innovative process described in the patent data circumvents these issues by utilizing water as the sole reaction medium, effectively eliminating the need for any organic solvents throughout the synthesis. This approach not only removes the fire and explosion hazards associated with aromatic hydrocarbons but also drastically simplifies the post-reaction workup. The reaction proceeds smoothly at mild temperatures between 0°C and 40°C, where cyclohexylamine and carbon disulfide react to form an intermediate salt which precipitates out of the aqueous solution. This precipitation allows for easy separation via simple filtration, avoiding the energy-intensive distillation or extraction steps required in organic solvent systems. Crucially, the mother liquor generated during filtration is not discarded but is recycled directly back into the reactor for subsequent batches, creating a closed-loop system that approaches zero liquid discharge. This circular economy model within the reactor significantly reduces raw material consumption and wastewater treatment burdens, positioning this method as a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Aqueous Thiourea Formation
The core of this technological breakthrough lies in the controlled formation and subsequent decomposition of the dithiocarbamate intermediate salt within an aqueous environment. In the first stage, cyclohexylamine is mixed with water in a specific mass ratio ranging from 2.5:1 to 3.8:1, creating a homogeneous phase that facilitates the nucleophilic attack on carbon disulfide. As carbon disulfide is added dropwise over a period of 1 to 3 hours, it reacts with the amine to form the corresponding dithiocarbamic acid salt. The temperature is strictly maintained between 0°C and 40°C during this addition and the subsequent 1 to 3-hour holding period to ensure optimal crystal formation of the intermediate salt while minimizing side reactions. The stoichiometry is carefully balanced, with the mass ratio of carbon disulfide to cyclohexylamine kept between 0.3:1 and 0.45:1, ensuring that the amine is not in such vast excess that it complicates downstream purification, yet sufficient to drive the reaction to completion.
Following the filtration of the intermediate salt, the process enters a critical dehydration and cyclization phase driven by vacuum thermal treatment. The wet filter cake, containing the intermediate salt, is subjected to a vacuum environment with a tightness ranging from -0.03 MPa to -0.1 MPa. Under these reduced pressure conditions, the temperature is raised to between 50°C and 100°C. This specific combination of heat and vacuum facilitates the elimination of hydrogen sulfide (H2S) gas from the intermediate structure, driving the equilibrium towards the formation of the stable thiourea linkage. The continuous removal of H2S gas prevents the reverse reaction and ensures that the final product is free from sulfur-containing impurities that could compromise its efficacy as a coupling agent. This low-temperature vacuum stripping technique is far more energy-efficient than traditional high-temperature drying and guarantees the high removal efficiency of volatile byproducts, resulting in the observed purity of greater than 99.5%.
How to Synthesize N,N'-Dicyclohexylthiourea Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the aqueous system. The process begins with the charging of water and cyclohexylamine into a reactor equipped with efficient stirring and temperature control capabilities. Operators must adhere strictly to the dropping rate of carbon disulfide to manage the exotherm and ensure uniform particle size of the precipitating salt. Once the reaction is complete, the slurry is filtered, and the solid cake is transferred to a drying vessel capable of sustaining high vacuum. The drying cycle is not merely for moisture removal but is the actual chemical conversion step where the thiourea ring closes. Detailed standard operating procedures regarding the specific ramp rates for temperature and vacuum application are critical for reproducibility. For a comprehensive breakdown of the exact operational parameters and equipment specifications required for this synthesis, please refer to the standardized guide below.
- Mix water and cyclohexylamine, then add carbon disulfide dropwise at 0-40°C while maintaining a mass ratio of water to amine between 2.5: 1 and 3.8:1.
- Filter the resulting intermediate salt precipitate to separate it from the mother liquor, which can be recycled for subsequent batches.
- Subject the filtered intermediate salt to vacuum heating at 50-100°C under negative pressure (-0.03 to -0.1 MPa) to remove hydrogen sulfide and obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this water-based synthesis protocol offers tangible strategic advantages that extend beyond simple regulatory compliance. The elimination of organic solvents fundamentally alters the cost structure of the manufacturing process by removing the need for solvent recovery units, explosion-proof electrical installations, and complex VOC abatement systems. This simplification of the plant infrastructure translates directly into lower capital expenditure (CAPEX) and reduced operational expenditure (OPEX). Furthermore, the ability to recycle the mother liquor indefinitely means that the consumption of water and auxiliary chemicals is minimized, leading to substantial cost savings in raw material procurement. The robustness of the process, demonstrated by consistent yields above 99% across multiple cycles, ensures a predictable supply output, mitigating the risk of production shortfalls that can disrupt downstream API synthesis schedules.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete substitution of expensive and hazardous organic solvents with water. In traditional processes, the cost of purchasing, recovering, and disposing of aromatic solvents constitutes a major portion of the variable costs. By eliminating this requirement, the process removes the associated energy costs of distillation and the regulatory costs of hazardous waste disposal. Additionally, the high yield (>99%) implies that nearly all input raw materials are converted into saleable product, minimizing waste generation and maximizing atom economy. This efficiency allows for a more competitive pricing structure for the final high-purity pharmaceutical intermediates, providing a distinct market advantage.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the volatility of solvent markets and strict transportation regulations regarding hazardous chemicals. Since this process relies on water and readily available bulk chemicals like cyclohexylamine and carbon disulfide, it is less susceptible to supply disruptions. The simplicity of the operation also means that the process can be easily scaled or replicated across different manufacturing sites without requiring specialized solvent-handling expertise. The recycling of the mother liquor further insulates the supply chain from fluctuations in water quality or availability, as the system becomes self-sustaining regarding the reaction medium, ensuring consistent delivery timelines for clients.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this aqueous method inherently mitigates them. The absence of flammable solvents makes the scale-up from pilot plant to commercial production significantly safer and easier to permit. Environmentally, the "zero discharge" characteristic of the mother liquor recycling aligns perfectly with increasingly stringent global environmental regulations. The low-temperature vacuum dehydration step also reduces the carbon footprint of the manufacturing process by lowering energy consumption compared to high-temperature reflux or distillation methods. This sustainability profile enhances the brand value of the final product for eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this material into their supply chains, we have compiled answers to common inquiries based on the patent specifications. These responses address critical concerns regarding product quality, process safety, and environmental impact, providing a clear picture of the operational benefits. Understanding these technical nuances is vital for making informed sourcing decisions that align with long-term corporate sustainability and quality goals.
Q: How does this water-based method improve safety compared to traditional aromatic solvent routes?
A: Traditional methods utilize flammable and toxic aromatic hydrocarbons like benzene or toluene, posing significant fire and health risks. This patented aqueous process eliminates organic solvents entirely, removing explosion hazards and reducing operator exposure to volatile organic compounds (VOCs).
Q: What purity levels can be achieved with the vacuum dehydration technique?
A: By employing low-temperature vacuum technology to strip hydrogen sulfide from the intermediate salt, the process consistently achieves product purity greater than 99.5%, meeting stringent requirements for peptide coupling agents without complex recrystallization steps.
Q: Is the mother liquor waste generated during filtration hazardous?
A: No, the process is designed for zero liquid discharge. The mother liquor remaining after filtration is not treated as waste but is directly recycled back into the reactor for the next batch, significantly reducing wastewater treatment costs and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-Dicyclohexylthiourea Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN102731357B requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our facilities are equipped with the necessary corrosion-resistant reactors and high-efficiency vacuum systems required to execute this aqueous synthesis safely and effectively. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of N,N'-dicyclohexylthiourea meets the >99.5% purity threshold essential for high-value peptide coupling applications.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this green synthesis technology, we can help you optimize your bill of materials while enhancing your supply chain's sustainability profile. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
