Advanced Metal-Free Synthesis of 3-Nitropyrrole for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and safe methodologies for synthesizing critical heterocyclic building blocks. A significant breakthrough in this domain is detailed in patent CN111875531B, which discloses a novel method for preparing 3-nitropyrrole based on sodium peroxodisulfate. This technology represents a paradigm shift from traditional, hazardous nitration protocols to a greener, metal-free oxidative system. By utilizing pyrrole as the starting material and employing sodium nitrite in conjunction with peroxodisulfate oxidants, this process achieves direct functionalization at the C3 position with exceptional efficiency. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition: it eliminates the need for expensive transition metal catalysts and dangerous strong acids, thereby streamlining the supply chain for high-purity pharmaceutical intermediates. The ability to produce 3-nitropyrrole—a key precursor for DNA sequencing bases and various alkaloids—through such a benign pathway underscores its potential for widespread adoption in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitropyrroles has been plagued by significant safety and operational challenges that hinder efficient commercial production. Traditional routes often rely on the use of highly corrosive and oxidizing reagents, such as concentrated nitric acid or reactive acyl chlorides, which pose severe risks of thermal runaway and equipment corrosion. Furthermore, existing technologies frequently necessitate multi-step sequences involving the installation and subsequent removal of amino protecting groups to prevent over-oxidation or polymerization of the sensitive pyrrole ring. These additional steps not only extend the overall lead time but also drastically reduce the comprehensive yield due to material losses during isolation and purification phases. The generation of substantial acidic waste streams from mixed-acid nitration further complicates environmental compliance, imposing heavy burdens on wastewater treatment facilities and increasing the total cost of ownership for manufacturers striving to meet modern green chemistry standards.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN111875531B introduces a streamlined, one-pot strategy that fundamentally simplifies the production landscape. This approach leverages the oxidative power of peroxodisulfates (such as sodium, potassium, or ammonium peroxodisulfate) to activate sodium nitrite, generating the necessary nitro-species in situ under mild thermal conditions ranging from 60-120°C. The elegance of this system lies in its directness; it bypasses the need for amino protection entirely, allowing for the direct conversion of pyrrole to 3-nitropyrrole in a single reaction vessel. This reduction in synthetic complexity translates directly into operational efficiency, as the process demonstrates stable and controllable kinetics suitable for scale-up. Moreover, the use of solid, commercially available oxidants and nitrite salts enhances workplace safety by eliminating the handling of volatile, fuming liquids, thus creating a more secure environment for plant operators and reducing the regulatory hurdles associated with hazardous material storage.

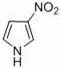
Mechanistic Insights into Peroxodisulfate-Mediated Nitration
From a mechanistic perspective, this reaction likely proceeds through a radical-mediated pathway initiated by the thermal decomposition of the peroxodisulfate anion. Upon heating, the peroxodisulfate generates sulfate radical anions, which are potent single-electron oxidants capable of activating the nitrite ion to form nitrogen dioxide radicals or related nitro-species. These electrophilic species then attack the electron-rich pyrrole ring, preferentially at the C3 position due to electronic stabilization of the intermediate sigma-complex. The absence of metal catalysts is particularly advantageous for pharmaceutical applications, as it removes the risk of heavy metal contamination in the final API, thereby alleviating the need for costly and time-consuming scavenging steps typically required to meet strict ppm limits. This metal-free nature ensures that the impurity profile of the resulting 3-nitropyrrole is cleaner and easier to manage, facilitating a more straightforward downstream purification process via standard column chromatography or crystallization techniques.
Furthermore, the regioselectivity observed in this transformation is a critical factor for R&D teams focusing on impurity control. The reaction conditions are tuned to favor mono-nitration, minimizing the formation of di-nitro byproducts or polymeric tars that often plague pyrrole chemistry. The patent data indicates that by carefully selecting the solvent system—ranging from polar aprotic solvents like DMF and DMSO to less polar options like toluene and chlorobenzene—the reaction environment can be optimized to stabilize the transition state and suppress side reactions. This tunability allows process chemists to maximize the yield, which has been reported to reach impressive levels between 92% and 98% across various examples. Such high efficiency suggests that the activation energy barrier for the desired pathway is significantly lower than that of competing degradation pathways, ensuring a robust process window that can tolerate minor variations in industrial settings without compromising product quality.
How to Synthesize 3-Nitropyrrole Efficiently
To implement this synthesis effectively, process engineers should focus on the precise stoichiometric balance between the pyrrole substrate and the oxidant system. The standard protocol involves dissolving pyrrole and sodium nitrite in a chosen solvent, followed by the addition of the peroxodisulfate salt. The mixture is then heated to the specified temperature range, with reaction progress monitored via thin-layer chromatography to ensure complete conversion before workup. Detailed standardized synthesis steps see the guide below.
- Combine pyrrole, sodium nitrite, and a peroxodisulfate oxidant (such as sodium, potassium, or ammonium peroxodisulfate) in a suitable organic solvent like acetonitrile or THF.
- Heat the reaction mixture to a temperature range of 60-120°C and maintain until thin-layer chromatography indicates complete consumption of the starting material.
- Upon completion, isolate the crude product and purify via column chromatography using a petroleum ether and ethyl acetate eluent system to obtain high-purity 3-nitropyrrole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this peroxodisulfate-based nitration method offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio; by replacing specialized, hazardous nitrating agents with commodity chemicals like sodium nitrite and common persulfates, companies can mitigate supply chain volatility and reduce procurement costs significantly. The elimination of protection-deprotection sequences means fewer unit operations, less solvent consumption, and reduced labor hours, all of which contribute to a leaner manufacturing cost structure. Additionally, the enhanced safety profile reduces insurance premiums and regulatory compliance costs associated with handling dangerous goods, making this route economically superior for long-term production contracts.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the removal of expensive catalytic systems and the reduction of synthetic steps. By avoiding the use of precious metal catalysts, manufacturers eliminate the capital expenditure associated with catalyst recovery systems and the ongoing cost of catalyst replenishment. Furthermore, the one-step nature of the reaction minimizes solvent usage and energy consumption compared to multi-step alternatives, leading to substantial operational savings. The high yields reported in the patent data imply less raw material waste per kilogram of product, directly improving the gross margin for high-purity pharmaceutical intermediates produced at scale.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved because the key reagents—pyrrole, sodium nitrite, and peroxodisulfates—are bulk commodities with established global supply chains. Unlike specialized reagents that may suffer from single-source bottlenecks or long lead times, these materials are readily available from multiple vendors, ensuring continuity of supply even during market fluctuations. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by sensitivity to minor environmental variations, allowing for more predictable delivery timelines to downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, operating at moderate temperatures that do not require extreme cooling or high-pressure equipment, thus lowering the barrier for technology transfer from lab to plant. From an environmental standpoint, the avoidance of strong mineral acids reduces the load on effluent treatment plants, aligning with increasingly stringent global environmental regulations. This green chemistry profile not only future-proofs the manufacturing site against regulatory changes but also enhances the brand reputation of the supplier as a sustainable partner in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitration technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational benefits for potential adopters. Understanding these nuances is essential for making informed decisions about process integration and supplier selection.
Q: What are the safety advantages of this nitration method compared to traditional mixed acid nitration?
A: This method replaces hazardous concentrated nitric acid and acyl chlorides with solid sodium nitrite and peroxodisulfates, significantly reducing the risk of explosive runaway reactions and corrosive handling issues.
Q: Does this process require amino protection groups for the pyrrole ring?
A: No, the patented process achieves direct regioselective nitration at the C3 position without the need for prior amino protection, thereby simplifying the synthetic route and improving atom economy.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the reaction operates under mild thermal conditions (60-120°C) with stable and controllable kinetics, making it highly adaptable for kilogram-to-ton scale manufacturing in standard reactor vessels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitropyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our team of expert process chemists has extensively evaluated the peroxodisulfate-mediated nitration route and possesses the technical capability to execute this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our 3-nitropyrrole meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener synthesis route for your supply chain. We encourage you to contact us today to discuss your requirements,索取 specific COA data, and review our comprehensive route feasibility assessments to ensure the successful and timely execution of your next campaign.
