Advanced Synthesis of 5-Chloro-2-Aminobenzenesulfonamide for Commercial Pharmaceutical Manufacturing
Introduction to Next-Generation Sulfonamide Synthesis
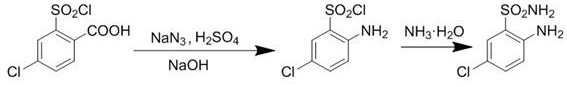
The pharmaceutical industry constantly seeks robust pathways for critical intermediates, and the preparation method disclosed in patent CN114920670B represents a significant leap forward for the production of 5-chloro-2-aminobenzenesulfonamide. This compound serves as a pivotal building block for benzothiadiazine derivatives, which are extensively utilized across various therapeutic areas. The patented technology addresses long-standing inefficiencies in traditional manufacturing by introducing a streamlined two-step sequence that converts 4-chloro-2-(chlorosulfonyl) benzoic acid directly into the target amine functionality. By leveraging sodium azide chemistry, this method circumvents the need for complex catalytic hydrogenation or corrosive sulfonation processes that have historically plagued this chemical space. For R&D directors and process chemists, this innovation offers a tangible route to higher yields and superior purity profiles without compromising on operational safety or equipment simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-chloro-2-aminobenzenesulfonamide has been fraught with significant technical and economic hurdles that hinder efficient commercial production. Traditional routes often rely on the hydrolysis of 5-chlorobenzothiadiazine, a pathway characterized by exorbitant raw material costs and poor economic viability for large-scale operations. Alternative methods involving the chlorination of o-aminobenzenesulfonamide suffer from prolonged reaction times and disappointingly low yields, creating bottlenecks in supply chains. Furthermore, processes utilizing aluminum trichloride catalysis with p-chloroaniline require harsh reaction conditions that demand specialized corrosion-resistant equipment, driving up capital expenditure. Perhaps most critically, the reduction of nitro precursors using iron generates massive quantities of iron mud, a hazardous waste stream that is notoriously difficult to treat and dispose of, posing severe environmental compliance challenges for modern manufacturing facilities.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a clever transformation of the carboxyl group into an amino group via an azide intermediate. This strategy begins with the reaction of 4-chloro-2-(chlorosulfonyl) benzoic acid with sodium azide in a polar aprotic solvent system, followed by a carefully controlled acid-base workup to isolate the key intermediate, 2-amino-5-chlorobenzenesulfonyl chloride. The second step involves a mild ammonolysis reaction where the sulfonyl chloride moiety is converted to the sulfonamide using ammonia water. This two-step sequence is remarkable for its operational simplicity, proceeding under normal atmospheric pressure without the need for high-temperature or high-pressure reactors. The elimination of transition metal catalysts and the avoidance of heavy metal waste streams fundamentally alter the cost structure and environmental footprint of the synthesis.
Mechanistic Insights into Azide-Mediated Carboxyl Conversion
The core chemical innovation lies in the first step, which effectively mimics a Curtius-type rearrangement logic to install the amine functionality ortho to the sulfonyl chloride group. When 4-chloro-2-(chlorosulfonyl) benzoic acid reacts with sodium azide, it forms an acyl azide intermediate in situ, which subsequently undergoes thermal rearrangement to an isocyanate species before hydrolyzing to the primary amine. The patent specifies precise molar ratios, such as maintaining the ratio of starting acid to sodium azide between 1.03 and 1.12, ensuring complete conversion while minimizing excess reagent costs. The subsequent addition of sodium nitrite and sulfuric acid, followed by neutralization with sodium hydroxide, facilitates the isolation of the intermediate sulfonyl chloride with high fidelity. This mechanistic pathway is superior because it avoids the radical mechanisms associated with nitro reductions, thereby reducing the formation of complex side products that are difficult to separate.
Impurity control is further enhanced in the second step through the use of specific recrystallization techniques. The patent highlights an alcohol-water purification method where the crude product is dissolved in ethanol and precipitated by the addition of deionized water. This solvent engineering approach exploits the differential solubility of the target sulfonamide versus potential by-products, such as unreacted sulfonyl chloride or hydrolysis products. By optimizing the volume ratio of ethanol to water, typically between 1:5 and 1:10, the process ensures the precipitation of a white, high-purity solid. This physical purification step is critical for meeting the stringent purity specifications required for pharmaceutical intermediates, consistently achieving purity levels above 98% and often exceeding 99% in optimized embodiments.
How to Synthesize 5-Chloro-2-Aminobenzenesulfonamide Efficiently
Implementing this synthesis requires careful attention to temperature control and reagent addition rates to maximize safety and yield. The process begins with the dissolution of the benzoic acid derivative in solvents like acetone or DMF, followed by the controlled addition of sodium azide at temperatures ranging from 50°C to 80°C. Detailed standard operating procedures regarding the specific stoichiometry, reaction times, and workup protocols are essential for reproducibility. For a comprehensive guide on executing this synthesis with optimal parameters, please refer to the standardized protocol below.
- React 4-chloro-2-(chlorosulfonyl) benzoic acid with sodium azide in a solvent like acetone or DMF at 50-80°C, followed by acidification and basification to isolate 2-amino-5-chlorobenzenesulfonyl chloride.
- Treat the intermediate sulfonyl chloride with ammonia water or pyrrolidine ammonium dithioformate to effect aminolysis.
- Purify the crude product via alcohol-water recrystallization to achieve purity levels exceeding 98%.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative benefits that directly impact the bottom line and operational resilience. The shift away from expensive and scarce starting materials like 5-chlorobenzothiadiazine to widely available commodity chemicals like 4-chloro-2-(chlorosulfonyl) benzoic acid drastically reduces raw material volatility. This stability in sourcing ensures that production schedules are not disrupted by supply shortages, a common issue with specialized heterocyclic precursors. Furthermore, the simplified equipment requirements mean that manufacturing can be conducted in standard glass-lined or stainless steel reactors without the need for exotic alloys or high-pressure vessels, significantly lowering the barrier to entry for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts, such as nickel used in traditional hydrogenation routes, removes the costly and time-consuming step of heavy metal scavenging and removal. This not only saves on the cost of the catalyst itself but also reduces the consumption of auxiliary materials like activated carbon or specialized resins needed for purification. Additionally, the high yield reported in the patent examples implies a more efficient use of raw materials, meaning less waste is generated per kilogram of product, which translates to substantial cost savings in waste disposal and raw material procurement.
- Enhanced Supply Chain Reliability: The reliance on atmospheric pressure operations and moderate temperatures significantly reduces the risk of unplanned shutdowns due to equipment failure or safety incidents. Unlike high-pressure hydrogenation processes that require rigorous safety monitoring and specialized infrastructure, this azide-based route can be scaled up with greater confidence and speed. The use of common solvents like ethanol and acetone further ensures that supply chains remain robust, as these materials are globally sourced and less prone to logistical bottlenecks compared to specialized reagents.
- Scalability and Environmental Compliance: One of the most significant advantages is the drastic reduction in hazardous waste generation, particularly the avoidance of iron mud associated with nitro reductions. This simplifies the environmental compliance burden, reducing the costs associated with wastewater treatment and solid waste disposal. The process is inherently greener, aligning with modern sustainability goals and regulatory pressures, which facilitates smoother permitting and long-term operational continuity for manufacturing sites aiming to minimize their ecological footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on yield expectations, purity profiles, and scalability factors. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this new method for commercial production.
Q: What are the primary advantages of the azide-based route over traditional nitro reduction?
A: Unlike traditional nitro reduction which generates difficult-to-treat iron mud and requires harsh conditions, the azide-based route described in CN114920670B operates under atmospheric pressure with simple equipment, significantly simplifying waste treatment and purification.
Q: How is high purity (>99%) achieved in this synthesis?
A: High purity is secured through a specific alcohol-water recrystallization process in the final step, which effectively removes impurities and by-products, yielding a white powder suitable for sensitive pharmaceutical applications.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes readily available raw materials like 4-chloro-2-(chlorosulfonyl) benzoic acid and avoids high-pressure reactors, making it highly suitable for large-scale commercial manufacturing with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-2-Aminobenzenesulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for high-value pharmaceutical intermediates like 5-chloro-2-aminobenzenesulfonamide. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for downstream drug synthesis. Our facility is equipped to handle the specific solvent systems and reaction conditions outlined in this patent, allowing us to deliver product that matches the high-quality benchmarks set by the latest intellectual property.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how switching to this advanced synthesis route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can support your long-term strategic goals in the pharmaceutical sector.
